Molecules in Magnetic Fields Svetlana Berdyugina Kiepenheuer Institut

Molecules in Magnetic Fields Svetlana Berdyugina Kiepenheuer Institut für Sonnenphysik, Freiburg, Germany Hot Molecules in Exoplanets and Inner Disks

Content 1. Coupling of angular momenta 2. Atomic Zeeman effect 3. Molecular Zeeman effect 4. Molecular Paschen-Back effect

Coupling of angular momenta • A moving charge creates a magnetic field. • The magnetic field associated with the orbital motion of the electron can be idealized as that of an infinitesimal dipole with a magnetic moment: • Analogously, another magnetic moment is associated with the electron internal angular momentum (spin): J • Coupling of angular momenta is the interaction of their magnetic moments • LS-coupling leads to the total angular momentum with the quantum number J: S L
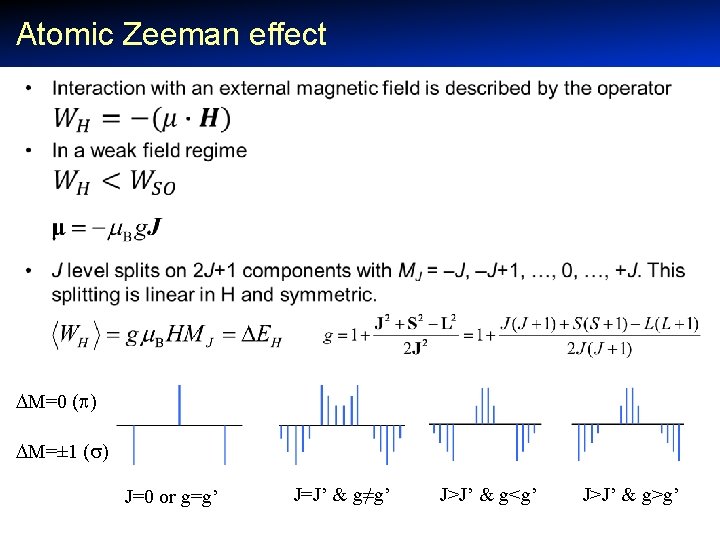
Atomic Zeeman effect • M=0 ( ) M=± 1 ( ) J=0 or g=g’ J=J’ & g≠g’ J>J’ & g<g’ J>J’ & g>g’
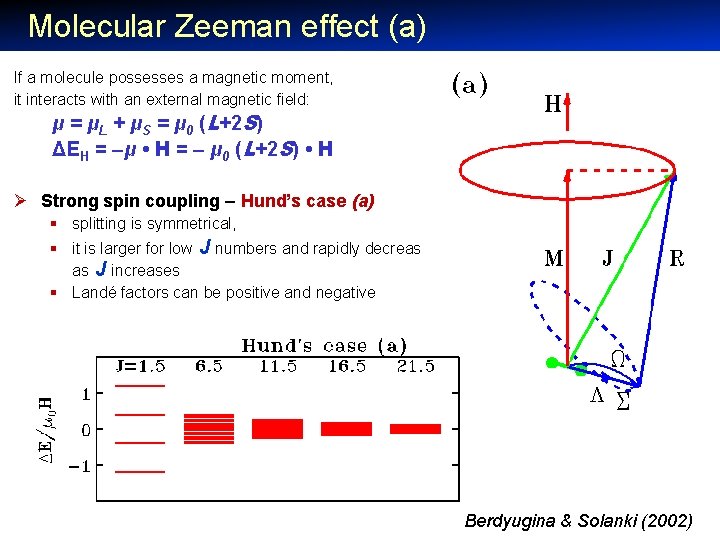
Molecular Zeeman effect (a) If a molecule possesses a magnetic moment, it interacts with an external magnetic field: μ = μL + μS = μ 0 (L+2 S) ΔEH = –μ • H = – μ 0 (L+2 S) • H Ø Strong spin coupling – Hund’s case (a) § splitting is symmetrical, § it is larger for low J numbers and rapidly decreases as J increases § Landé factors can be positive and negative Berdyugina & Solanki (2002)
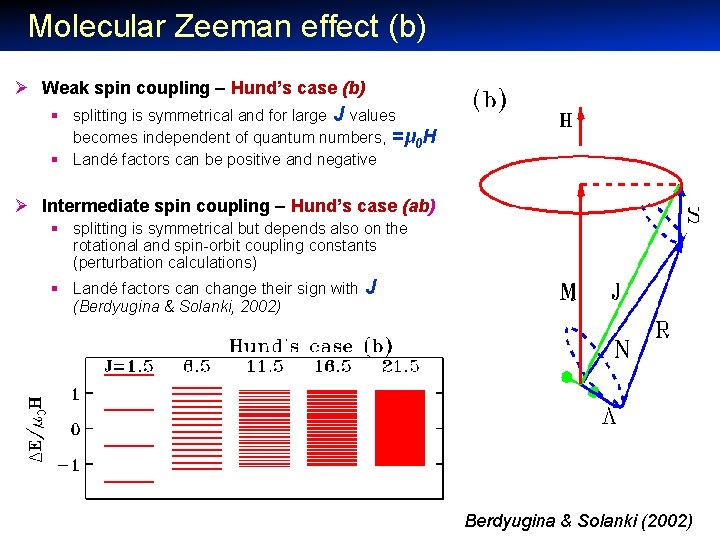
Molecular Zeeman effect (b) Ø Weak spin coupling – Hund’s case (b) § splitting is symmetrical and for large J values becomes independent of quantum numbers, =μ 0 H § Landé factors can be positive and negative Ø Intermediate spin coupling – Hund’s case (ab) § splitting is symmetrical but depends also on the rotational and spin-orbit coupling constants (perturbation calculations) § Landé factors can change their sign with (Berdyugina & Solanki, 2002) J Berdyugina & Solanki (2002)
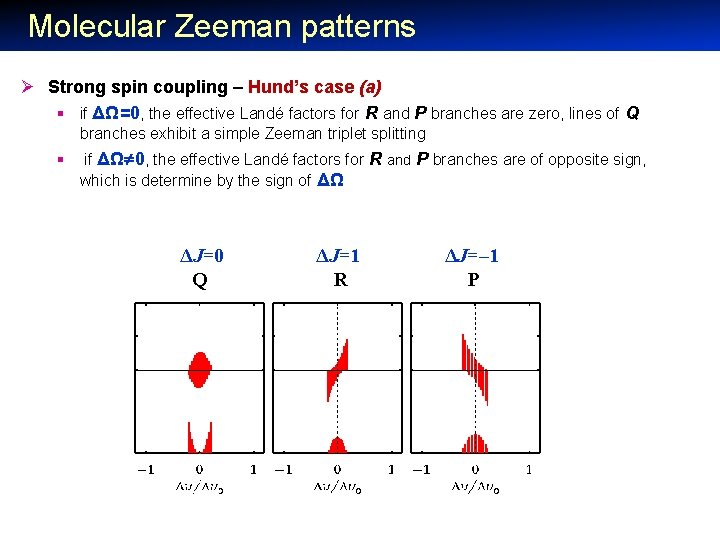
Molecular Zeeman patterns Ø Strong spin coupling – Hund’s case (a) § if ΔΩ=0, the effective Landé factors for R and P branches are zero, lines of Q branches exhibit a simple Zeeman triplet splitting § if ΔΩ 0, the effective Landé factors for R and P branches are of opposite sign, which is determine by the sign of ΔΩ ΔJ=0 Q ΔJ=1 R ΔJ=– 1 P
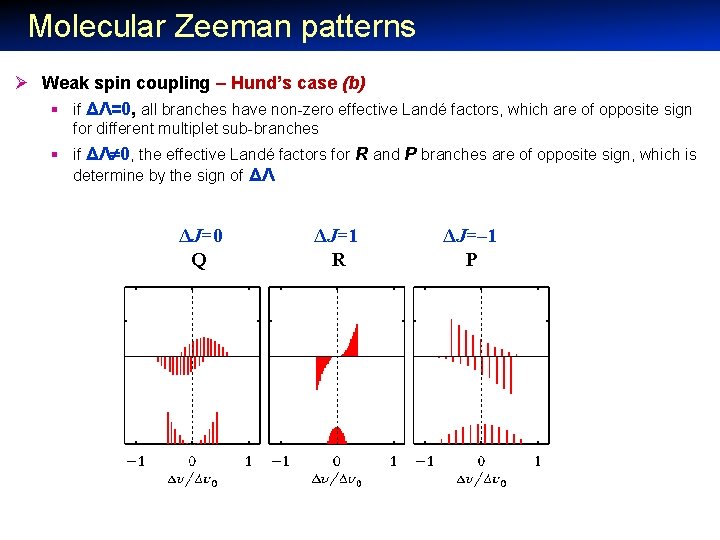
Molecular Zeeman patterns Ø Weak spin coupling – Hund’s case (b) § if ΔΛ=0, all branches have non-zero effective Landé factors, which are of opposite sign for different multiplet sub-branches § if ΔΛ 0, the effective Landé factors for R and P branches are of opposite sign, which is determine by the sign of ΔΛ ΔJ=0 Q ΔJ=1 R ΔJ=– 1 P
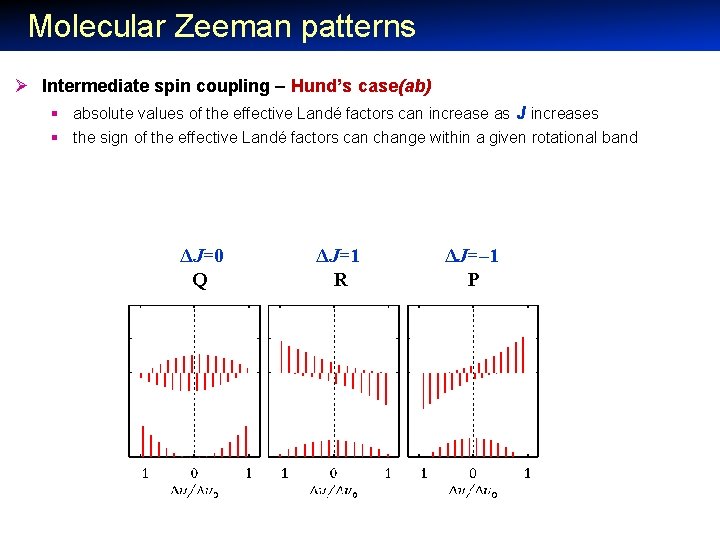
Molecular Zeeman patterns Ø Intermediate spin coupling – Hund’s case(ab) § absolute values of the effective Landé factors can increase as J increases § the sign of the effective Landé factors can change within a given rotational band ΔJ=0 Q ΔJ=1 R ΔJ=– 1 P

Negative Landé Factors Ø The effective Landé factor of an atomic or molecular line: geff = ½ (gu +gl) + ¼ (gu –gl) [Ju(Ju+1) – Jl(Jl+1)], geff < 0 if gu< 0 and gl < 0 Ø The Landé factor of an atomic level (LS coupling): g = 3/2 + [S(S+1) – L(L+1)] / [2 J(J+1)], J=L+S, . . . , |L – S| g < 0 if L(L+1) – S(S+1) > 3 J(J+1), ½(L+1) < S < L for J =L – S, 6 D L=3, S=2. 5 1/2 7 L=4, S=3; 3. 5 F 1, 8 F 1/2 Ø Only few atomic spectral lines exhibit negative Zeeman sptlitting (Stenflo et al. 1984) Ø The Landé factor of a molecular level (Hund’s case a): g = (Λ+Σ)(Λ+2Σ) / J(J+1), J = |Λ+Σ|, |Λ+Σ|+1, |Λ+Σ|+2, . . . g < 0 if –Λ < Σ < ½ Λ , 2Π , 3Π Λ=1, Σ= –½, 0 1/2 1, . . . 3Δ , 2Δ , . . . Λ=2, Σ= – 1, –½, 0, ½ 1 3/2 2 5/2 Ø Negative Landé factors are common in molecular lines (Berdyugina & Solanki, 2002) Berdyugina & Solanki (2002)
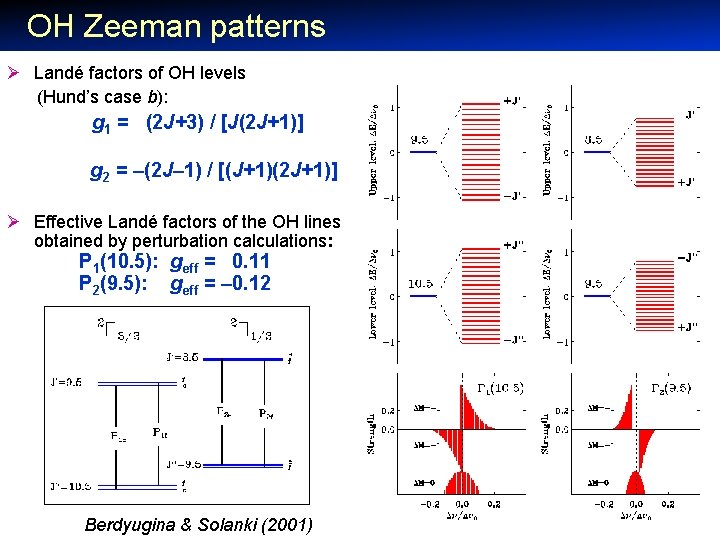
OH Zeeman patterns Ø Landé factors of OH levels (Hund’s case b): g 1 = (2 J+3) / [J(2 J+1)] g 2 = –(2 J– 1) / [(J+1)(2 J+1)] Ø Effective Landé factors of the OH lines obtained by perturbation calculations: P 1(10. 5): geff = 0. 11 P 2(9. 5): geff = – 0. 12 Berdyugina & Solanki (2001)
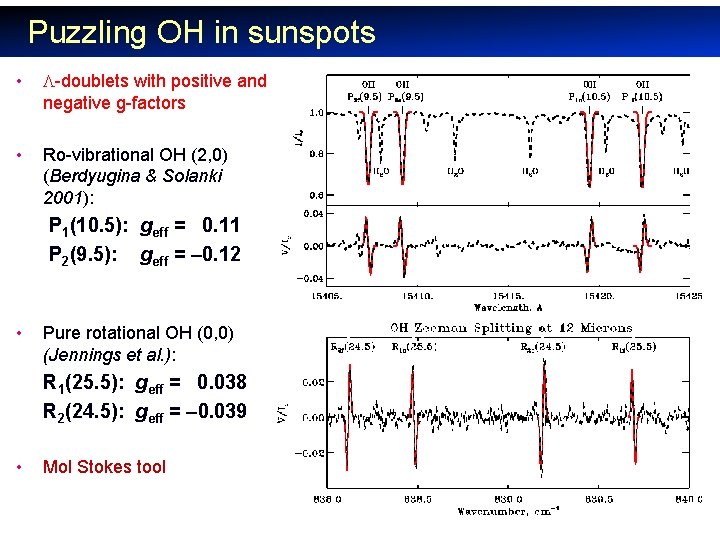
Puzzling OH in sunspots • -doublets with positive and negative g-factors • Ro-vibrational OH (2, 0) (Berdyugina & Solanki 2001): P 1(10. 5): geff = 0. 11 P 2(9. 5): geff = – 0. 12 • Pure rotational OH (0, 0) (Jennings et al. ): R 1(25. 5): geff = 0. 038 R 2(24. 5): geff = – 0. 039 • Mol Stokes tool
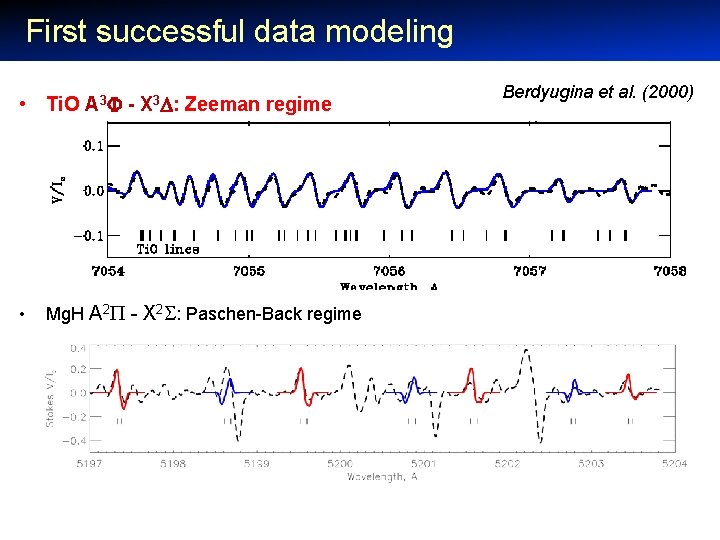
First successful data modeling • Ti. O A 3 - X 3 : Zeeman regime • Mg. H A 2 - X 2 : Paschen-Back regime Berdyugina et al. (2000)
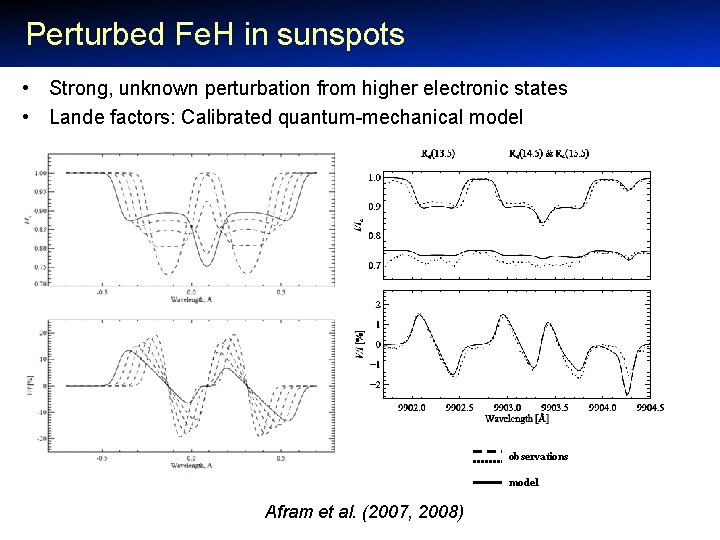
Perturbed Fe. H in sunspots • Strong, unknown perturbation from higher electronic states • Lande factors: Calibrated quantum-mechanical model observations model Afram et al. (2007, 2008)
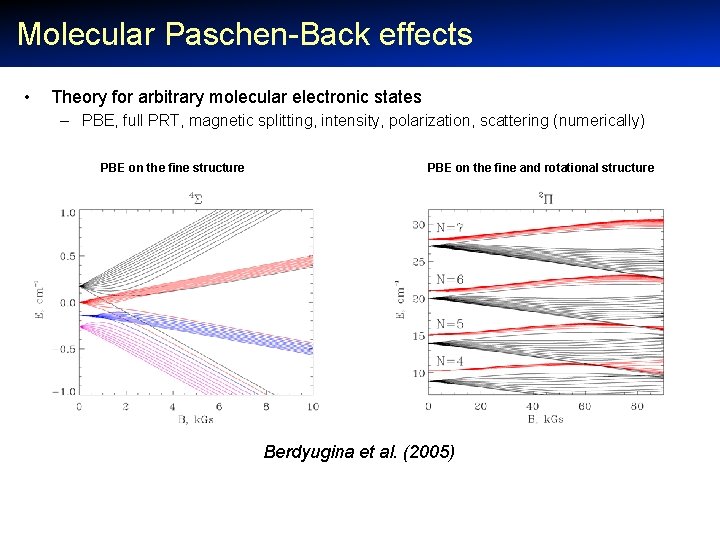
Molecular Paschen-Back effects • Theory for arbitrary molecular electronic states – PBE, full PRT, magnetic splitting, intensity, polarization, scattering (numerically) PBE on the fine structure PBE on the fine and rotational structure Berdyugina et al. (2005)
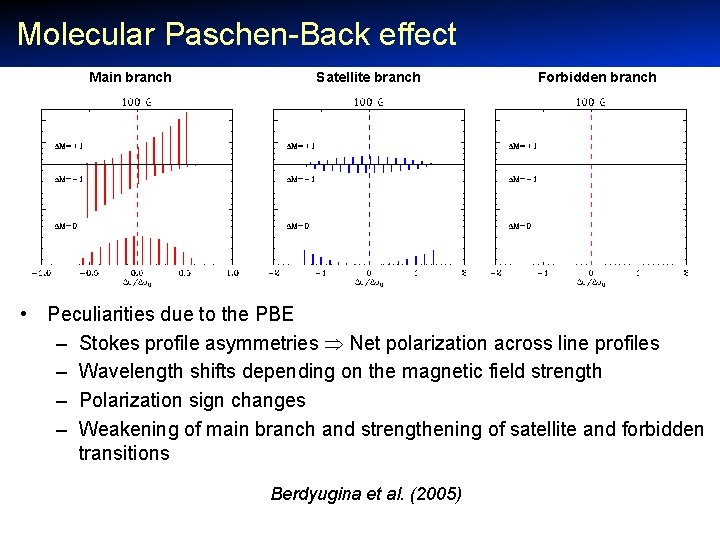
Molecular Paschen-Back effect Main branch Satellite branch Forbidden branch • Peculiarities due to the PBE – Stokes profile asymmetries Net polarization across line profiles – Wavelength shifts depending on the magnetic field strength – Polarization sign changes – Weakening of main branch and strengthening of satellite and forbidden transitions Berdyugina et al. (2005)
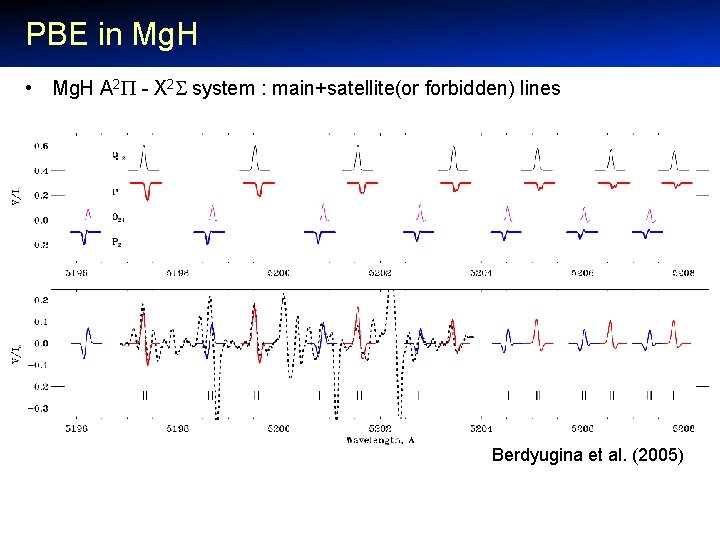
PBE in Mg. H • Mg. H A 2 - X 2 system : main+satellite(or forbidden) lines Berdyugina et al. (2005)
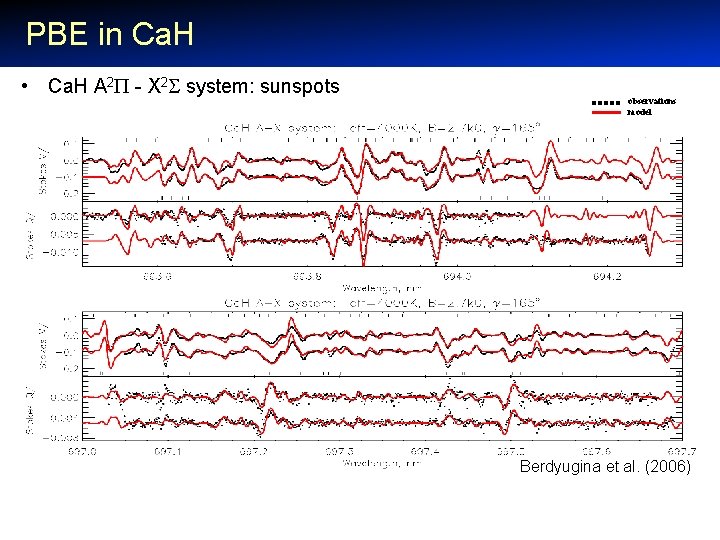
PBE in Ca. H • Ca. H A 2 - X 2 system: sunspots observations model Berdyugina et al. (2006)
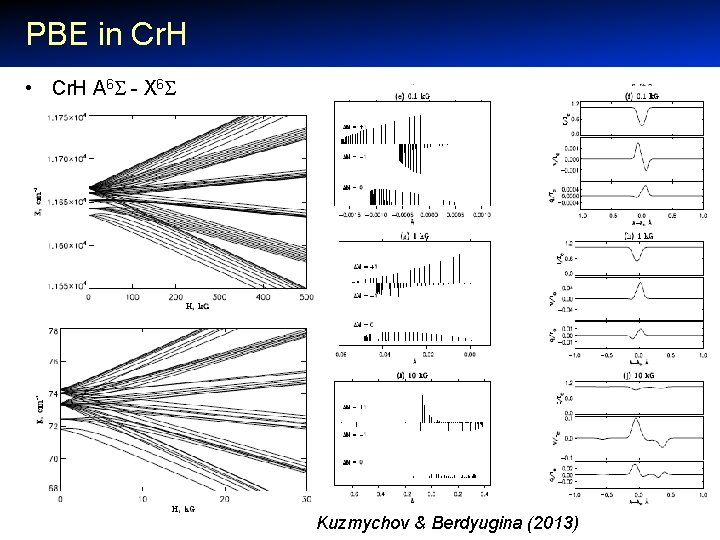
PBE in Cr. H • Cr. H A 6 - X 6 Kuzmychov & Berdyugina (2013)

Conclusions • Molecular lines are magnetically sensitive: • Strong Zeeman signal is expected for transitions with intermediate case spin coupling (e. g. Ti. O, Fe. H) • Negative Landé factors are common for molecular lines • Unusual Stokes V signatures of infrared OH lines is the natural outcome of molecular properties • Stokes polarimetry with the pairs of negative and positive Zeeman-split OH lines offers advantages for assessing instrumental effects (I, Q, U to V crosstalk) or magnetooptic effects in the solar atmosphere § Molecular Pachen-Back effect leads to higher magnetic sensitivity: • • Stokes profile asymmetries Net polarization across line profiles Wavelength shifts depending on the magnetic field strength Polarization sign changes Weakening of main branch and strengthening of satellite and forbidden transitions
- Slides: 20