Molecular Vibrations Chain of Approximations Schrdinger Dirac BornOppenheimer

Molecular Vibrations
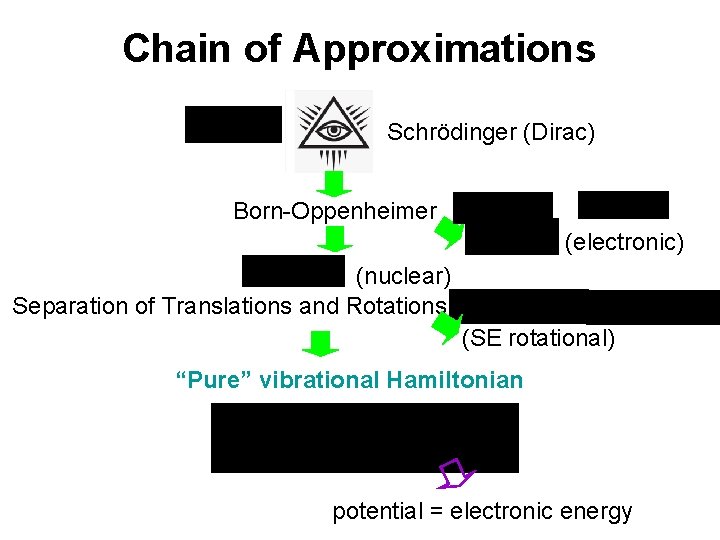
Chain of Approximations Schrödinger (Dirac) Born-Oppenheimer (electronic) (nuclear) Separation of Translations and Rotations (SE rotational) “Pure” vibrational Hamiltonian potential = electronic energy
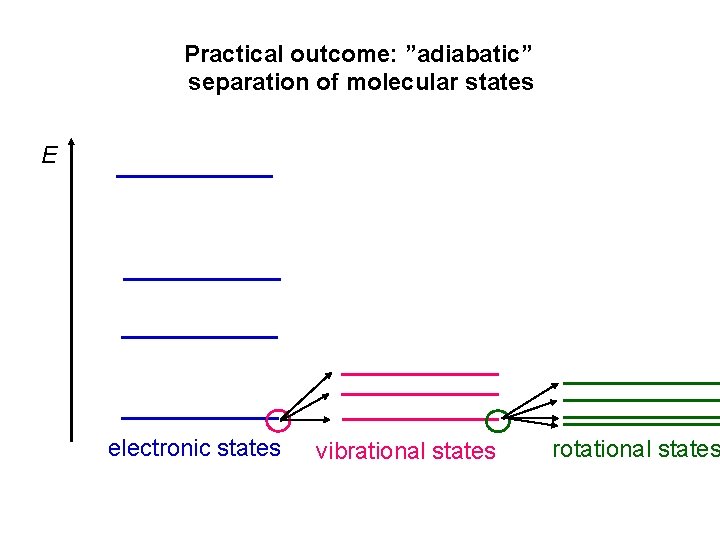
Practical outcome: ”adiabatic” separation of molecular states E electronic states vibrational states rotational states
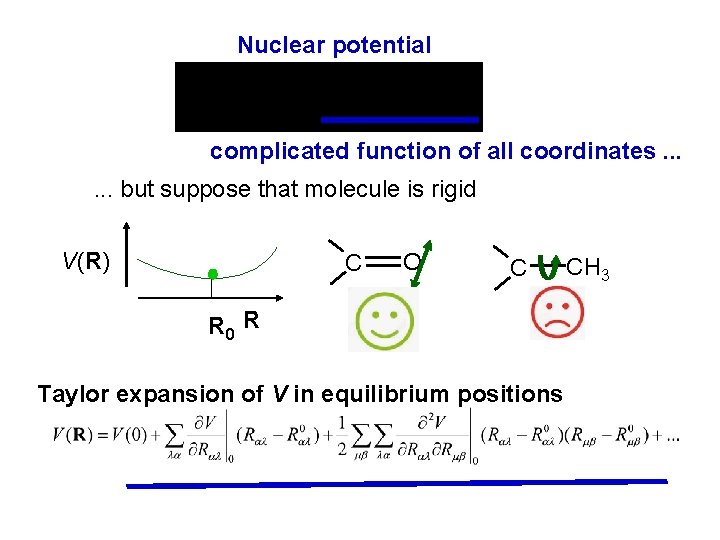
Nuclear potential complicated function of all coordinates. . . but suppose that molecule is rigid V(R) C O C R 0 R Taylor expansion of V in equilibrium positions CH 3

The Harmonic Approximation X X ! In matrix notation: (vector of deviations from equilibrium positions)

“Force field” = “second energy derivatives” = “Hessian” - symmetric (Hermitian) matrix - calculation (e. g. Gaussian: FREQ) =0 - needed “coupled-perturbed” HF/KS equations

Usage of Hessian -vibrational spectra – infrared absorption - vibrational circular dichroism - Raman scattering - Raman optical activity - inelastic neutron scattering - optimization control - positive eigenvalues - negative eigenvalue(s)

Solution of the Harmonic Problem by Two Coordinate Transformations 1) mass weighted coordinates x

Solution of the Harmonic Problem by Two Coordinate Transformations 1) mass weighted coordinates x 2) normal mode coordinates Q require . . . coordinate rotation . . . 2. . diagonal matrix

Vibrational Hamiltonian of any Molecule . . . sum of 3 N harmonic oscillators Remark: we can also define a direct transformation between Cartesian and normal mode coordinates via a “big” S-matrix
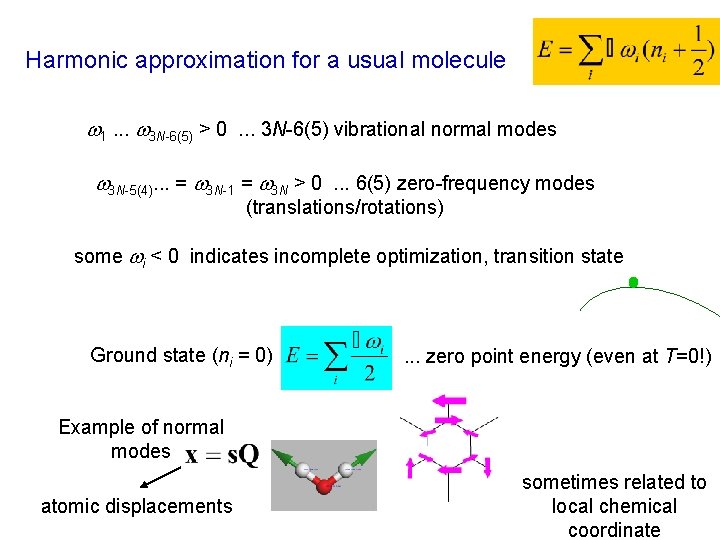
Harmonic approximation for a usual molecule 1. . . 3 N-6(5) > 0. . . 3 N-6(5) vibrational normal modes 3 N-5(4). . . = 3 N-1 = 3 N > 0. . . 6(5) zero-frequency modes (translations/rotations) some i < 0 indicates incomplete optimization, transition state Ground state (ni = 0) . . . zero point energy (even at T=0!) Example of normal modes atomic displacements sometimes related to local chemical coordinate
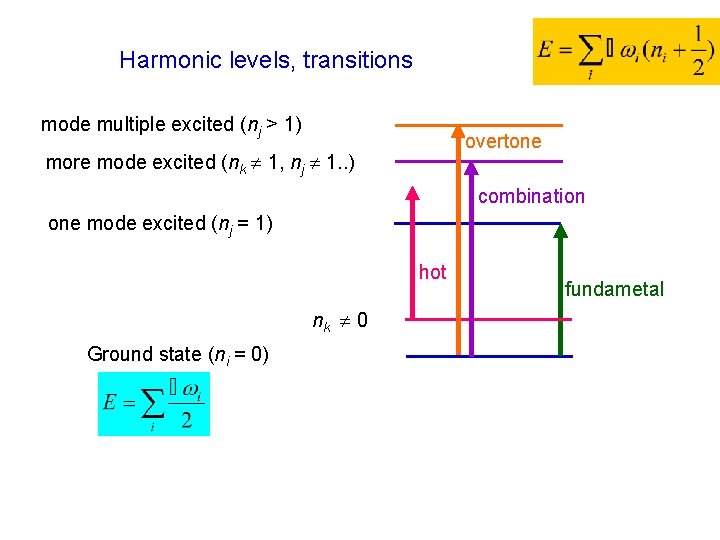
Harmonic levels, transitions mode multiple excited (nj > 1) overtone more mode excited (nk 1, nj 1. . ) combination one mode excited (nj = 1) hot nk 0 Ground state (ni = 0) fundametal

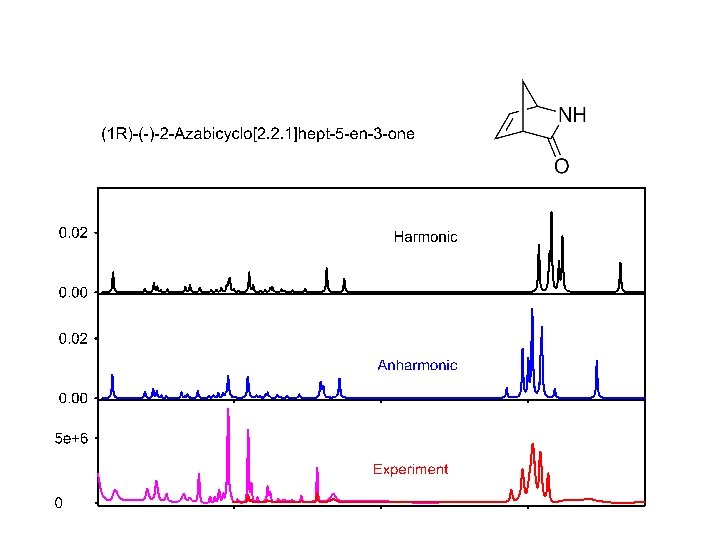
Limits of harmonic approximation - formally works for any molecule - exact solution of the vibrational part of Schrödinger equation - Gaussian: FREQ (VCD, Raman, ROA) - flexible motions (CH 3 rot) unusable - bond stretching – large error - combination modes, overtones – not appropriate
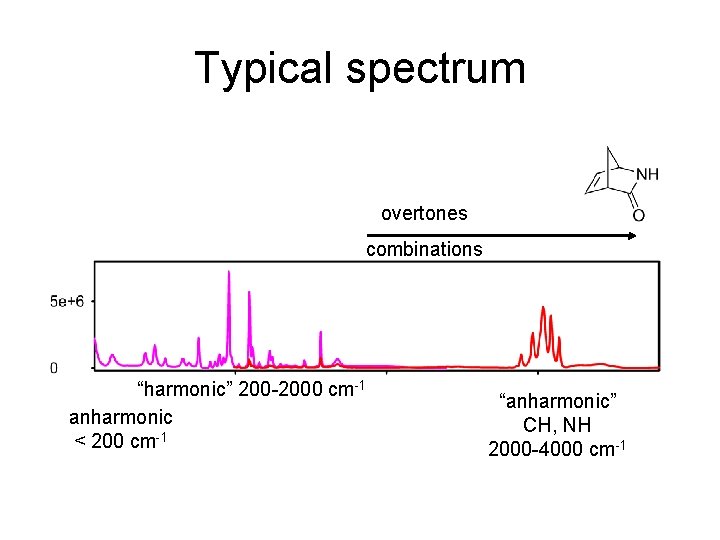
Typical spectrum overtones combinations “harmonic” 200 -2000 cm-1 anharmonic < 200 cm-1 “anharmonic” CH, NH 2000 -4000 cm-1

Calculation beyond the harmonic approximation G 16: AH IR, VCD, Raman, ROA, e. g. Freq=(ROA, Anharm) • Usually cubic and quartic derivatives added: • Corrections based on the perturbation theory
- Slides: 16