Molecular symmetry analysis of lowenergy torsional and vibrational

Molecular symmetry analysis of low-energy torsional and vibrational states in the S 0 and S 1 states of p-xylene to interpret the REMPI spectrum of p-xylene P. Groner Department of Chemistry, University of Missouri - Kansas City, MO, USA A. M. Gardner, W. D. Tuttle, T. G. Wright School of Chemistry, University of Nottingham, University Park, Nottingham, UK International Symposium on Molecular Spectroscopy, June 19 -23, 2017 1

p-Xylene has * * Two methyl internal rotors CH 3 -C 6 H 4 -CH 3 Molecular frame with D 2 h symmetry No permanent dipole moment → No pure rotational spectrum Very low barriers to internal rotation (vibronic GS toluene V 6 = 4. 8386 cm-1 ) VV Ilyushin et al. , J. Mol. Spectrosc. 259 (2010) 26 -38 * Molecular symmetry group D 3 × C 2 (order 72) for 12 C 81 H 10 P Groner, JR Durig, J. Chem. Phys. 66 (1977) 1856 -1874 * Electronic spectroscopy S 1 ← S 0 PJ Breen et al. , J. Chem. Phys. 87 (1987) 1917 -1926 6/15/2016 e-mail with questions from Tim Wright, U of Nottingham, UK Answers lead to more questions, more answers, and. . . collaboration. AM Gardner, WD Tuttle, P Groner, TG Wright, J. Chem Phys. 146 (2017) 124308 WD Tuttle, AM Gardner, KB O’Regan, W Malewicz, TG Wright, J. Chem Phys. 146 (2017) 124309 International Symposium on Molecular Spectroscopy, June 19 -23, 2017 2
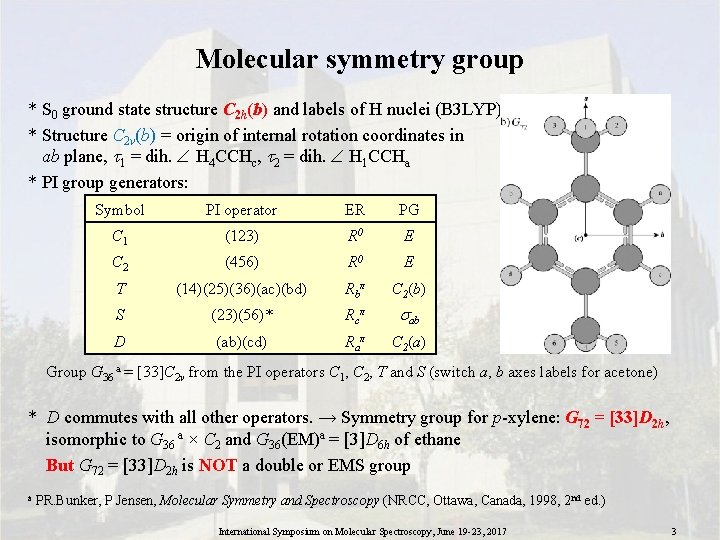
Molecular symmetry group * S 0 ground state structure C 2 h(b) and labels of H nuclei (B 3 LYP) * Structure C 2 v(b) = origin of internal rotation coordinates in ab plane, 1 = dih. H 4 CCHc, 2 = dih. H 1 CCHa * PI group generators: Symbol PI operator ER PG C 1 (123) R 0 E C 2 (456) R 0 E T (14)(25)(36)(ac)(bd) Rbπ C 2(b) S (23)(56)* Rcπ ab D (ab)(cd) Raπ C 2(a) Group G 36 a = [33]C 2 v from the PI operators C 1, C 2, T and S (switch a, b axes labels for acetone) * D commutes with all other operators. → Symmetry group for p-xylene: G 72 = [33]D 2 h, isomorphic to G 36 a × C 2 and G 36(EM)a = [3]D 6 h of ethane But G 72 = [33]D 2 h is NOT a double or EMS group a PR. Bunker, P Jensen, Molecular Symmetry and Spectroscopy (NRCC, Ottawa, Canada, 1998, 2 nd ed. ) International Symposium on Molecular Spectroscopy, June 19 -23, 2017 3

Position vectors and coordinate substitutions • Operator C 1 C 2 Order 3 3 → → → 1 → 2 → 1 - 1 2 2 - Xj, l → Xj, l X 1, k → X 1, k-1 X 1, k X 2, k → X 2, k-1 T 2 +π π- - 2 1 X 3 -j, 3 -l X 2, k X 1, k S 2 +π π- π- - 1 - 2 -Xj, l -X 1, -k -X 2, -k International Symposium on Molecular Spectroscopy, June 19 -23, 2017 D 2 +π 1 - π 2 - π Xj, 3 -l X 1, k X 2, k 4
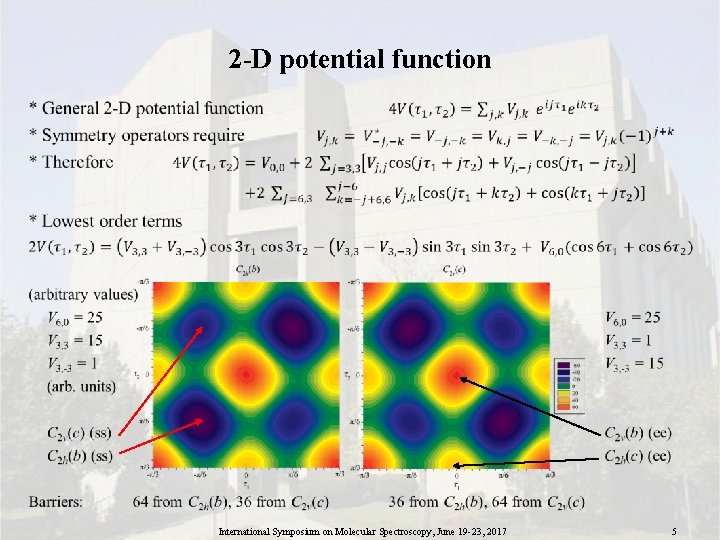
2 -D potential function • International Symposium on Molecular Spectroscopy, June 19 -23, 2017 5
![Character table of G 72 = [33]D 2 h Since G 72 = [33]D Character table of G 72 = [33]D 2 h Since G 72 = [33]D](http://slidetodoc.com/presentation_image_h2/5247c834a53144eedef7852cd5db7472/image-6.jpg)
Character table of G 72 = [33]D 2 h Since G 72 = [33]D 2 h = G 36 × C 2{E, D}, its character table has the form ( 1 2)gtu ' Op D*Op G 36 ( 1 2)utu " G 36 [3, 3]D 2 h #c 00 g++ 00 g+‑ 00 g‑+ 00 g‑‑ 11 g+ 11 g‑ 12 g+ 12 g‑ 01 g * * G 72 A 1' A 3' A 2' A 4' E 3' E 4' E 1' E 2' G' E 1 1 1 2 2 4 C 1 C 2 -1 2 1 1 2 2 -1 -1 -2 C 1 C 2 2 1 1 -1 -1 2 2 -2 C 1 4 1 1 -1 -1 1 T 3 1 1 -1 -1 2 -2 0 0 0 C 1 C 2 T 6 1 1 -1 -1 -1 1 0 0 0 U 3 1 -1 0 0 2 -2 0 C 1 C 2 -1 U 6 1 -1 0 0 -1 1 0 S 9 1 -1 -1 1 0 0 0 ( 1 2)gtu ' 1 2)utu " ee Tb Γ* oo, Jb eo, Ja, tot Tc Ta , tot oe, Jc -G 36 Dipole moment operator in space-fixed axes belongs to 00 g+‑ (A 3') tot = el* vib* tor* rot* sp tot belong to the 1 -D species 00 g‑+ (A 2') or 00 g‑- (A 4') rot = |JKa. Kc. M. > belong to 1 -D species International Symposium on Molecular Spectroscopy, June 19 -23, 2017 6

Symmetrized internal rotor wavefunctions * tor = |m 1 m 2> = exp[i(m 1 1+m 2 2]/2 ; -∞ < m 1, m 2 < +∞ m 1 + m 2 = even → |m 1 m 2> belong to g (or ') species m 1 + m 2 = odd → |m 1 m 2> belong to u (or ") species * Applying the operators E, T, U = ST and S to |m 1 m 2> creates the functions |m 1 m 2 >, |m 2 m 1 >, |-m 2 -m 1 >, |-m 1 -m 2 >, respectively, forming the set {m 1 m 2} Sets contain one (only {00}), two ({m m}and {m -m}) or four functions Both m 1 and m 2 are integer multiples of 3 → symmetrize to functions of 1 -D species One of (m 1, m 2) is an integer multiple of 3→ set belongs to a 4 -D species Otherwise, → sets of 2 functions belong to 2 -D species or → symmetrize to functions of 2 -D species * Symmetrization {m 1 m 2}tu = |m 1 m 2 > + t |m 2 m 1 > + u |-m 2 -m 1 > + tu |-m 1 -m 2 > (T) > 0 → t = +1; (T) < 0 → t = -1 * Alternate notation: m 1 = 3 m + 1, 1 = m 1(mod 3), m 2 = 3 n + 2, 2 = m 2(mod 3), (U) > 0 → u = +1 (U) > 0 → u = -1, m = (m 1 - 1)/3 n = (m 2 - 2)/3 The symmetry numbers 1, 2 = 0, 1, 2 are used also as partial labels 1 2 of the symmetry species International Symposium on Molecular Spectroscopy, June 19 -23, 2017 7
![Transformation of tor and nuclear spin weights [3, 3]D 2 h G 72 NS Transformation of tor and nuclear spin weights [3, 3]D 2 h G 72 NS](http://slidetodoc.com/presentation_image_h2/5247c834a53144eedef7852cd5db7472/image-8.jpg)
Transformation of tor and nuclear spin weights [3, 3]D 2 h G 72 NS a SW a E 00 g++ 00 g+ ‑ 00 g‑ + 00 g‑‑ 11 g+ 11 g‑ 12 g+ 12 g‑ 01 g 00 u++ 00 u+ ‑ 00 u‑ + 00 u‑ ‑ 11 u+ 11 u‑ 12 u+ 12 u‑ 01 u A 1' A 3' A 2' A 4' E 3' E 4' E 1' E 2' G' A 1" A 3" A 2" A 4" E 3" E 4" E 1" E 2" G" 88 0 0 72 24 16 80 48 0 0 48 12 12 48 72 72 88 88 32 48 40 40 160 48 48 24 24 96 1 1 1 1 2 2 2 2 4 Free internal rotor functions b m≠n m+n {3 m, 3 n}++ even {0, 0}, {3 m, -3 m}+, {3 m, 3 m}+ {3 m, 3 n}+‑ even {3 m, 3 m}{3 m, 3 n}‑+ even {3 m, -3 m}+ {3 m, 3 n}‑‑ even 1+ {3 m+1, 3 n+1} even {3 m+1, 3 m+1} 1‑ {3 m+1, 3 n+1} even 2+ {3 m+1, 3 n-1} even {3 m+1, 3 m-1} {3 m+1, 3 n-1}2‑ even {3 m, 3 n+1} odd ++ {3 m, 3 n} odd +‑ {3 m, 3 n} odd ‑+ {3 m, 3 n} odd ‑‑ {3 m, 3 n} odd 1+ {3 m+1, 3 n+1} odd {3 m+1, 3 m+1} {3 m+1, 3 n+1}1 odd {3 m+1, 3 n-1}2+ odd {3 m+1, 3 m-1} 2{3 m+1, 3 n-1} odd {3 m, 3 n+1} even c, d ee Γ* eo, Ja, tot Tb oo, Jb Tc oe, Jc In the vibronic ground state, tor* rot = |m 1 m 2> |JKa. Kc. M> belongs to a g or u species if m 1 + m 2 + Ka = even or odd, respectively. International Symposium on Molecular Spectroscopy, June 19 -23, 2017 8

Internal rotation Hamiltonian • |0, 3> |3, 0> |-3, 0> |0, -3> Function Energy |0, 3> E 03 V 3, -3/4 V 3, 3/4 V 6, 0/4 {0, 3}++ = cos 3 1 + cos 3 2 = {0, 3(+)}+ E 03 + (V 3, -3+V 3, 3)/4 + V 6, 0/4 |3, 0> V 3, -3/4 E 03 V 6, 0/4 V 3, 3/4 {0, 3}-- = cos 3 1 - cos 3 2 = {0, 3(+)}‑ E 03 - (V 3, -3+V 3, 3)/4 + V 6, 0/4 |-3, 0> V 3, 3/4 V 6, 0/4 E 03 V 3, -3/4 {0, 3}+- = sin 3 1 + sin 3 2 = {0, 3(‑)}+ E 03 + (V 3, -3 -V 3, 3)/4 – V 6, 0/4 |0, -3> V 6, 0/4 V 3, 3/4 V 3, -3/4 E 03 {0, 3}-+ = sin 3 1 - sin 3 2 = {0, 3(‑)}‑ E 03 - (V 3, -3 -V 3, 3)/4 – V 6, 0/4 International Symposium on Molecular Spectroscopy, June 19 -23, 2017 9

Selection rules for REMPI spectrum * tot = el* vib* tor* rot* sp * One-photon selection rule Let ev = el* vib → tot = ev* tor* rot* sp * = (R ) × ( ) = (R ) × ( ev) × ( tor, ) = a, b, c * ( tor, ) = * × (R ) × ( ev) = (T ) × (ev') × (ev") * S 0 : (ev") = 00 g++ (A 1') S 1 : (ev') = 00 u++ (A 1") → ( ev) = 00 u++ (A 1") * For vibrational ground states in both S 0 and S 1 ( tor, a) = 00 u-- (A 4") a ( 1, 2) = C 3 c(cos 3 1 - cos 3 2) ( tor, b) = 00 g++ (A 1') b ( 1, 2) = C 0 + C 33 c cos 3 1 cos 3 2 + C 33 s sin 3 1 sin 3 2 ( tor, c) = 00 g-+ (A 2') c ( 1, 2) = C 33 sc (sin 3 1 cos 3 2 - cos 3 1 sin 3 2) REMPI spectra of S 1 ← S 0 for toluene and p-xylene look remarkably similar. For toluene, S 1 ← S 0 transition is compatible with b-type electronic transition moment Simulation and fit by: A. L. L. East, et al. , J. Chem. Phys. 112 (2000) 167 -175 International Symposium on Molecular Spectroscopy, June 19 -23, 2017 10
![Discussion & Conclusions * Molecular symmetry group of p-xylene is G 72 = [33]D Discussion & Conclusions * Molecular symmetry group of p-xylene is G 72 = [33]D](http://slidetodoc.com/presentation_image_h2/5247c834a53144eedef7852cd5db7472/image-11.jpg)
Discussion & Conclusions * Molecular symmetry group of p-xylene is G 72 = [33]D 2 h, Even though isomorphic to G 36(EM) = [3]D 6 h of ethane, dimethyl acetylene, it is NOT a double group or EMS group * Symmetrized (basis) functions for asymmetric rotor, free internal rotation Spin statistics Potential function Electric dipole selection rules for rovibronic transitions with 2 methyl internal rotors present * For “Full treatment” of symmetry analysis (and spectroscopy) See: AM Gardner, WD Tuttle, P Groner, TG Wright, J. Chem Phys. 146 (2017) 124308 See talks TD 10 by AM Garner and FC 06 by WD Tuttle for Spectroscopy and application of the symmetry analysis to the REMPI spectrum of p-xylene International Symposium on Molecular Spectroscopy, June 19 -23, 2017 11
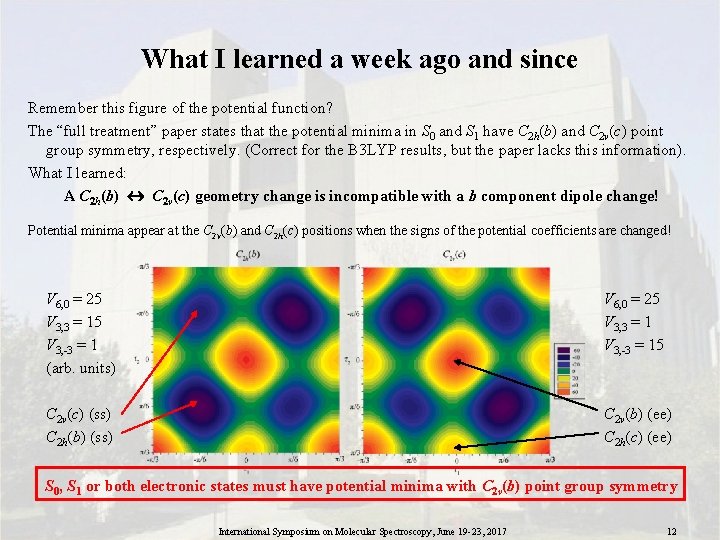
What I learned a week ago and since Remember this figure of the potential function? The “full treatment” paper states that the potential minima in S 0 and S 1 have C 2 h(b) and C 2 v(c) point group symmetry, respectively. (Correct for the B 3 LYP results, but the paper lacks this information). What I learned: A C 2 h(b) C 2 v(c) geometry change is incompatible with a b component dipole change! Potential minima appear at the C 2 v(b) and C 2 h(c) positions when the signs of the potential coefficients are changed! V 6, 0 = 25 V 3, 3 = 15 V 3, -3 = 1 (arb. units) V 6, 0 = 25 V 3, 3 = 1 V 3, -3 = 15 C 2 v(c) (ss) C 2 h(b) (ss) C 2 v(b) (ee) C 2 h(c) (ee) S 0, S 1 or both electronic states must have potential minima with C 2 v(b) point group symmetry International Symposium on Molecular Spectroscopy, June 19 -23, 2017 12

Thank you (It’s been a wild ride) International Symposium on Molecular Spectroscopy, June 19 -23, 2017 13

Abstract The electronic transition S 1 S 0 of p-xylene (p. Xyl) has been observed by REMPI and ZEKE spectroscopy. a Its analysis required a detailed investigation of the molecular symmetry of p. Xyl whose methyl groups are almost free internal rotors. The molecular symmetry group of p. Xyl, here called [33]D 2 h, has 72 operators. b It is isomorphic to G 36(EM), c the double group for ethane and dimethyl acetylene even though it is NOT a double group for p. Xyl. Loosely speaking, the group symbol, [33]D 2 h, indicates that is for a molecule with two threefold rotors on a molecular frame with D 2 h point group symmetry. The transformation properties of the (i) free internal rotor basis functions for the torsional coordinates, (ii) the asymmetric rotor basis (Wang) functions for the Eulerian angles, (iii) nuclear spin functions, (iv) potential function, and (v) transitions dipole moment functions were determined. The forms of the torsional potential in the S 0 and S 1 states and the dependence of the first order torsional splittings on the potential coefficients have been obtained. AM Gardner, WD Tuttle, P Groner, TG Wright, J. Chem. Phys. (submitted Dec 2016) b P Groner, JR Durig, J. Chem. Phys. 66 (1977) 1856 -74 c Bunker, Jensen, “Molecular Symmetry and Spectroscopy” (1998, NRC Research Press, Ottawa, 2 nd ed. ) a International Symposium on Molecular Spectroscopy, June 19 -23, 2017 14
- Slides: 14