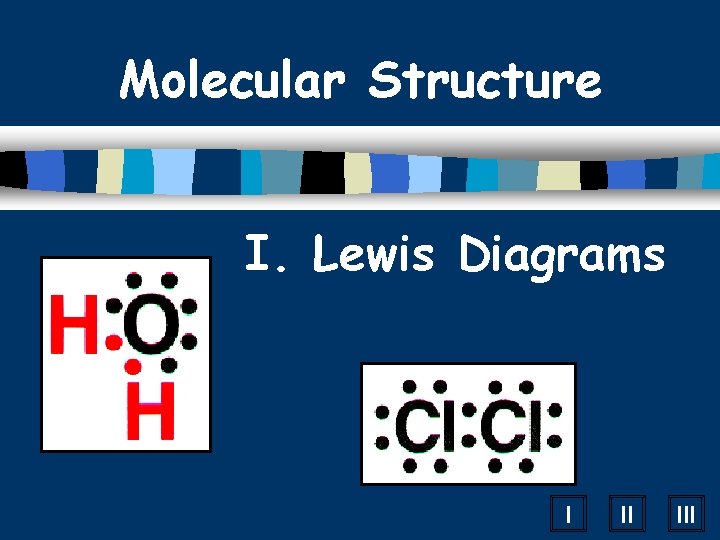
Molecular Structure I Lewis Diagrams I II III












- Slides: 33

Molecular Structure I. Lewis Diagrams I II III

A. Octet Rule n Remember… · Most atoms form bonds in order to have 8 valence electrons.

A. Octet Rule n Exceptions: · Hydrogen 2 valence e· Groups 1, 2, 3 get 2, 4, 6 valence e· Expanded octet more than 8 valence e- (e. g. S, P, Xe) · Radicals odd # of valence e-

B. Drawing Lewis Diagrams n Find total # of valence e-. n Arrange atoms - singular atom is usually in the middle. n Form bonds between atoms (2 e-). n Distribute remaining e- to give each atom an octet (recall exceptions). n If there aren’t enough e- to go around, form double or triple bonds.

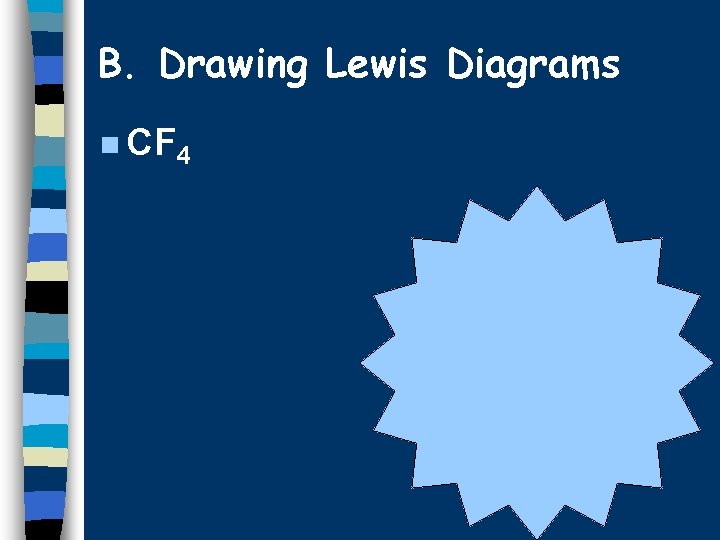
B. Drawing Lewis Diagrams n CF 4

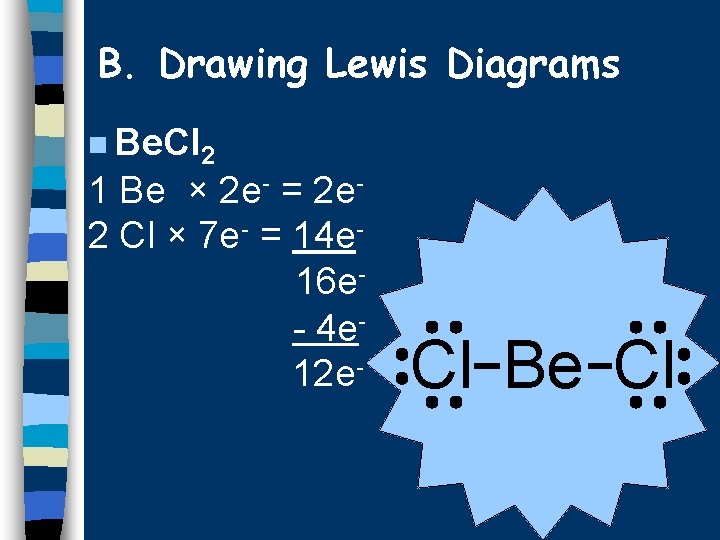
B. Drawing Lewis Diagrams n Be. Cl 2 1 Be × 2 e- = 2 e 2 Cl × 7 e- = 14 e 16 e- 4 e 12 e- Cl Be Cl

B. Drawing Lewis Diagrams n CO 2

C. Polyatomic Ions n To find total # of valence e-: · Add 1 e- for each negative charge. · Subtract 1 e- for each positive charge. n Place brackets around the ion and label the charge.

C. Polyatomic Ions n Cl. O 4 -

C. Polyatomic Ions n NH 4+

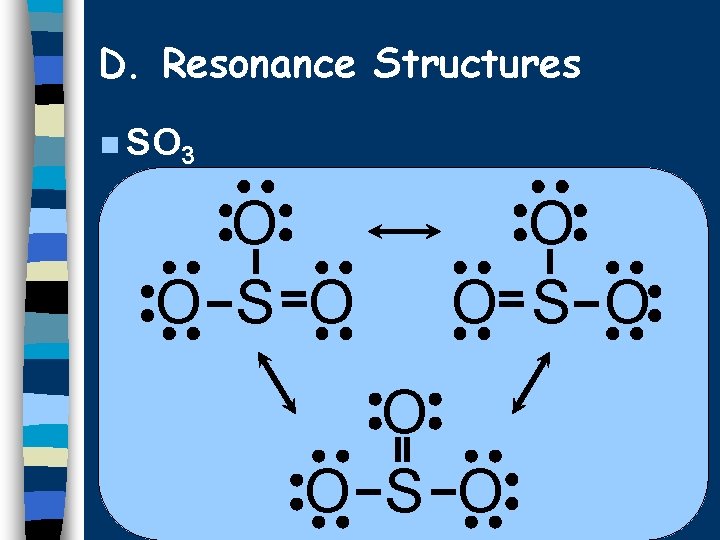
D. Resonance Structures Molecules that can’t be correctly represented by a single Lewis diagram. n Actual structure is an average of all the possibilities. n Show possible structures separated by a double-headed arrow. n

D. Resonance Structures n SO 3 O O S O

Molecular Structure II. Molecular Geometry I II III

A. VSEPR Theory n Valence Shell Electron Pair Repulsion Theory n Electron pairs orient themselves in order to minimize repulsive forces.

A. VSEPR Theory n Types of e- Pairs · Bonding pairs - form bonds · Lone pairs - nonbonding e- Lone pairs repel more strongly than bonding pairs!!!

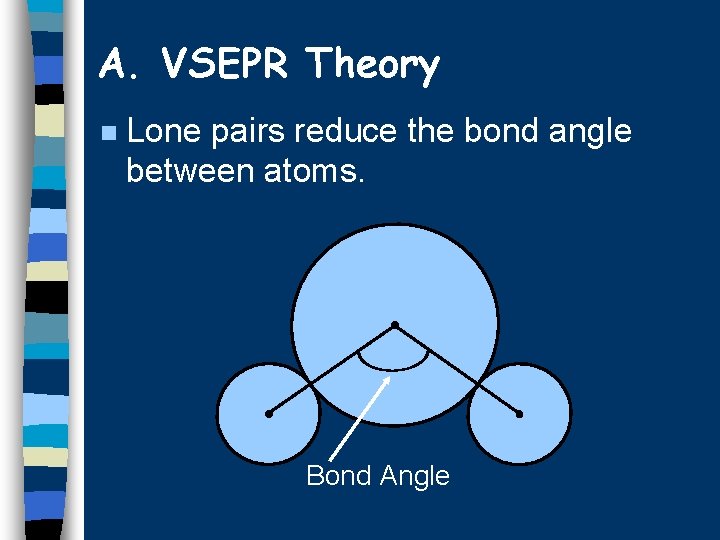
A. VSEPR Theory n Lone pairs reduce the bond angle between atoms. Bond Angle

B. Determining Molecular Shape n Draw the Lewis Diagram. n Tally up e- pairs on central atom. · double/triple bonds = ONE pair n Shape is determined by the # of bonding pairs and lone pairs. Know the 8 common shapes & their bond angles!

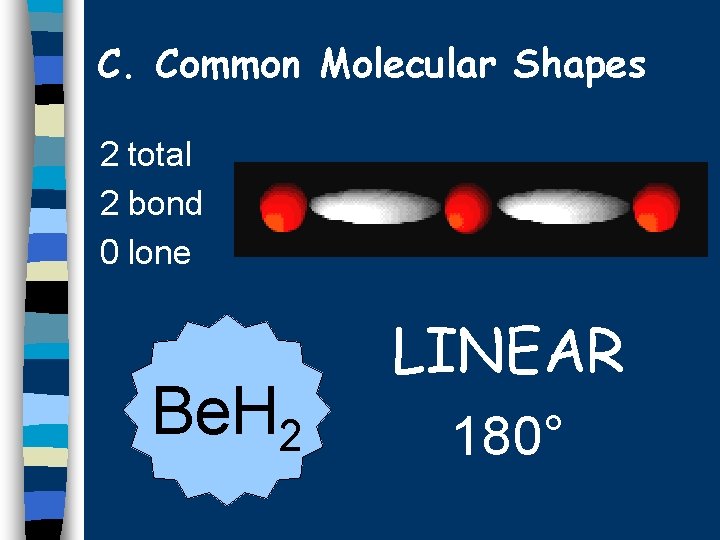
C. Common Molecular Shapes 2 total 2 bond 0 lone Be. H 2 LINEAR 180°

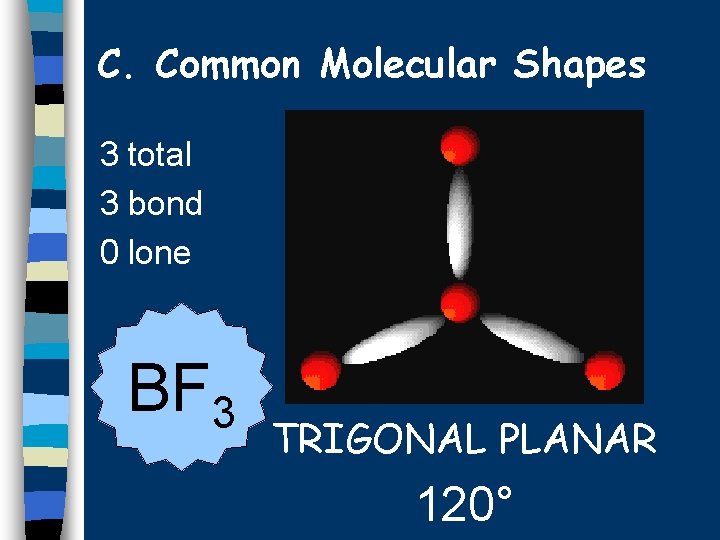
C. Common Molecular Shapes 3 total 3 bond 0 lone BF 3 TRIGONAL PLANAR 120°

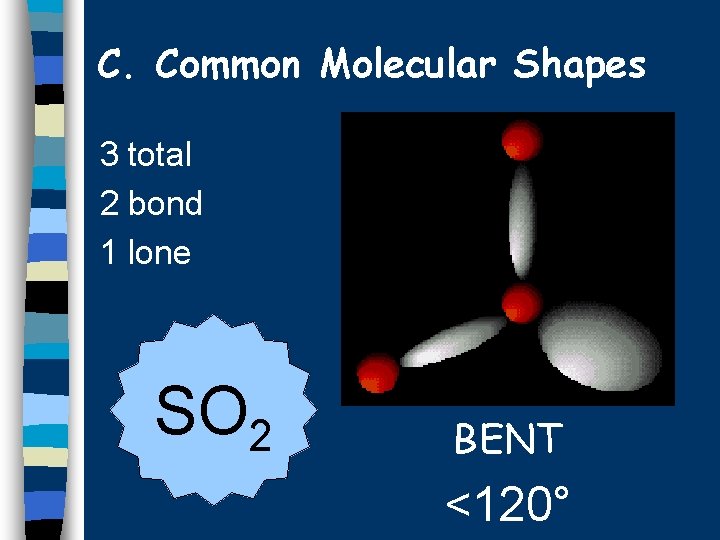
C. Common Molecular Shapes 3 total 2 bond 1 lone SO 2 BENT <120°

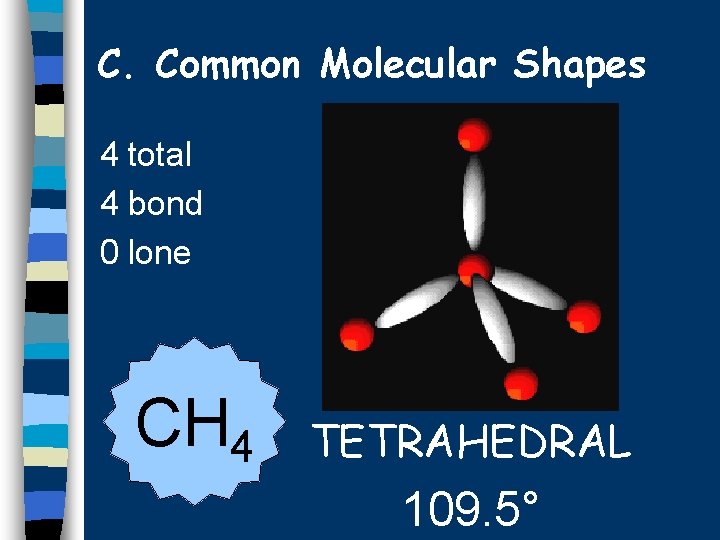
C. Common Molecular Shapes 4 total 4 bond 0 lone CH 4 TETRAHEDRAL 109. 5°

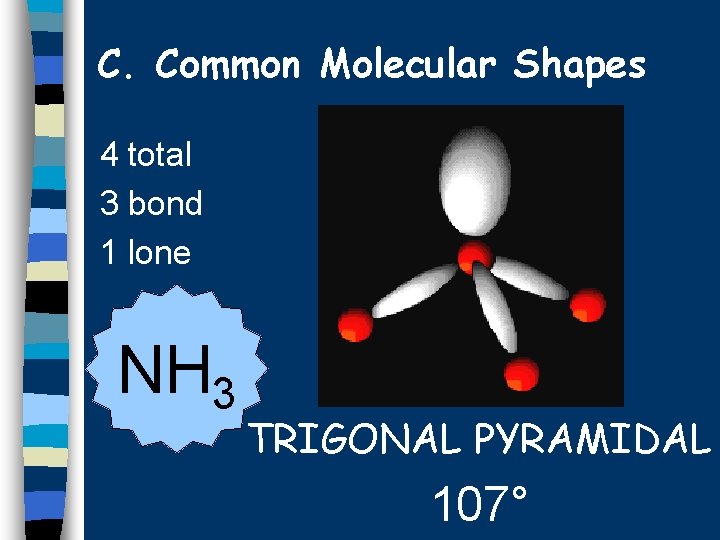
C. Common Molecular Shapes 4 total 3 bond 1 lone NH 3 TRIGONAL PYRAMIDAL 107°

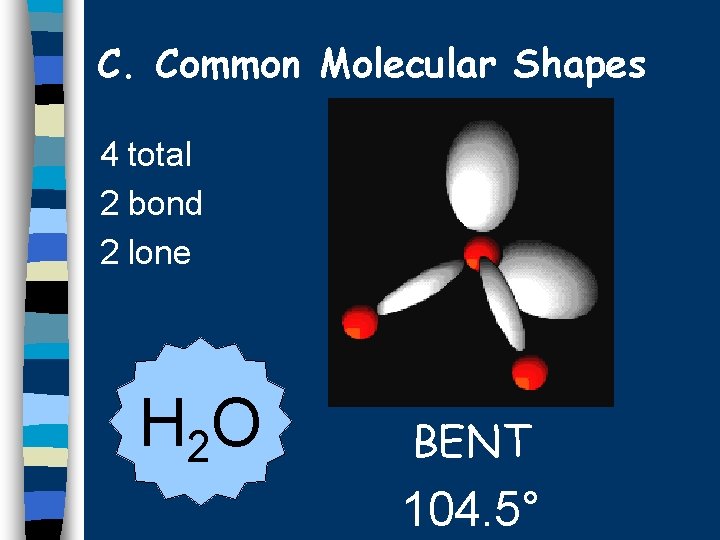
C. Common Molecular Shapes 4 total 2 bond 2 lone H 2 O BENT 104. 5°

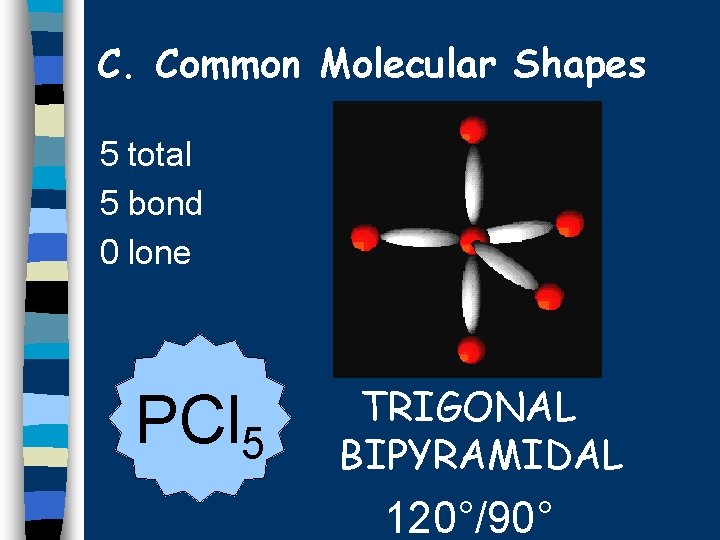
C. Common Molecular Shapes 5 total 5 bond 0 lone PCl 5 TRIGONAL BIPYRAMIDAL 120°/90°

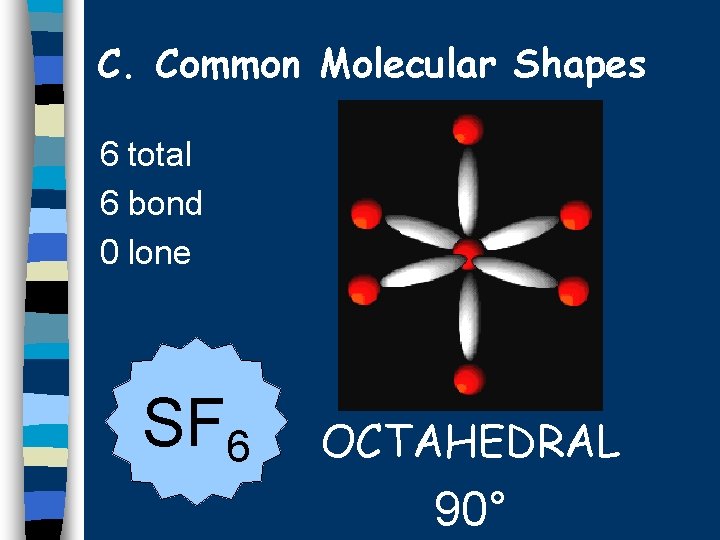
C. Common Molecular Shapes 6 total 6 bond 0 lone SF 6 OCTAHEDRAL 90°

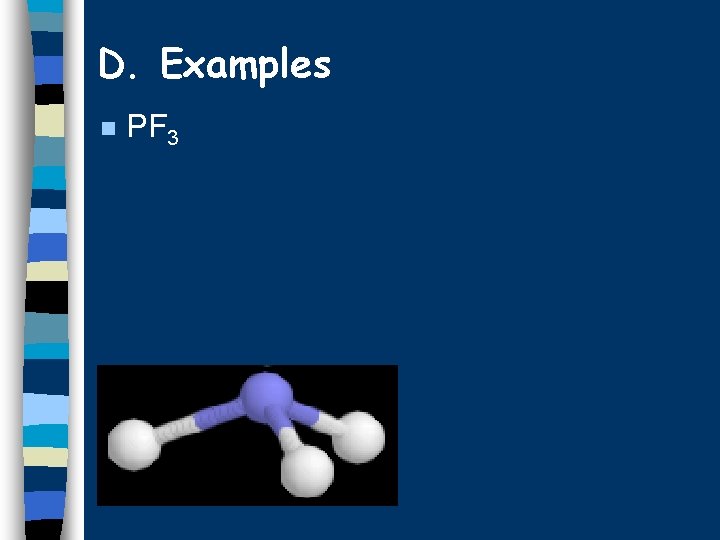
D. Examples n PF 3

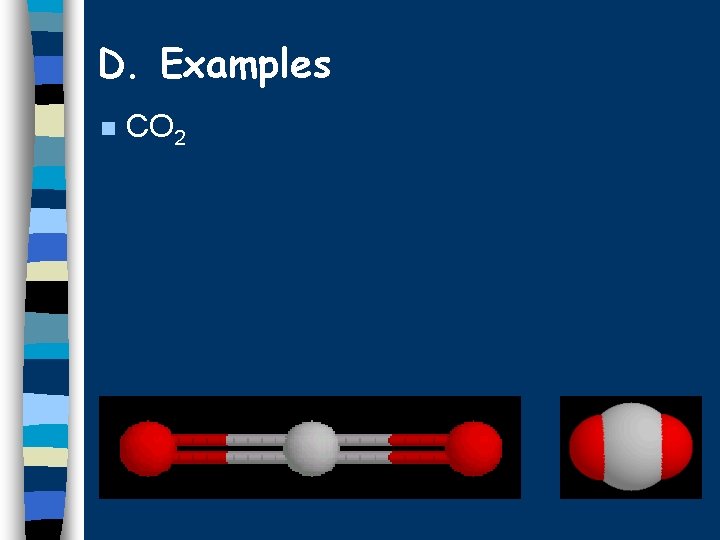
D. Examples n CO 2

Molecular Structure III. Molecular Polarity I II III

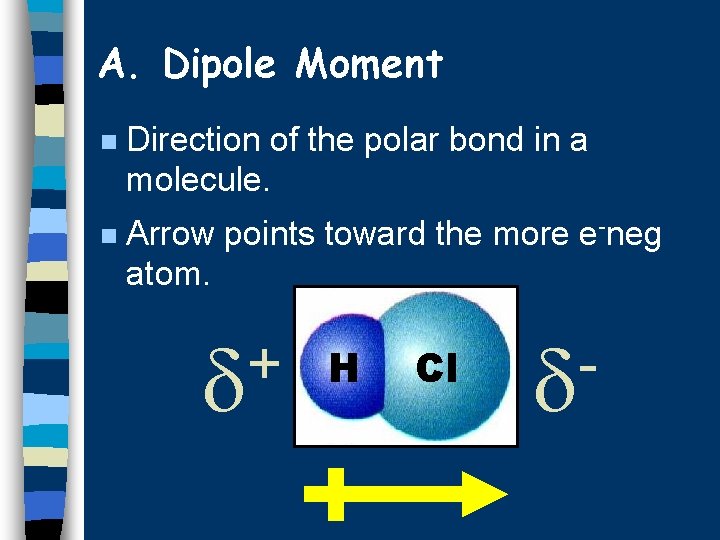
A. Dipole Moment n Direction of the polar bond in a molecule. n Arrow points toward the more e-neg atom. + H Cl

B. Determining Molecular Polarity n Depends on: · dipole moments · molecular shape

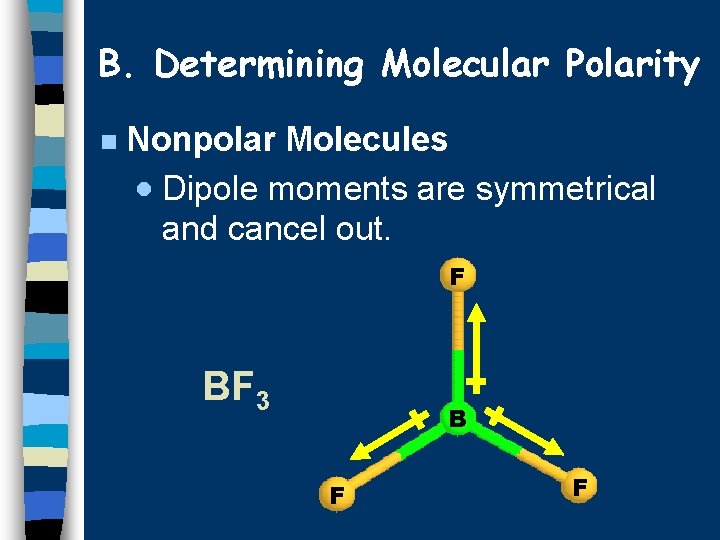
B. Determining Molecular Polarity n Nonpolar Molecules · Dipole moments are symmetrical and cancel out. F BF 3 B F F

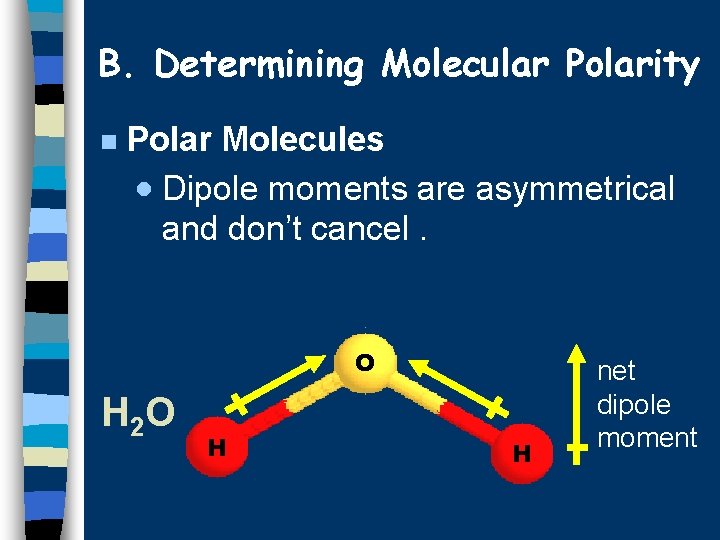
B. Determining Molecular Polarity n Polar Molecules · Dipole moments are asymmetrical and don’t cancel. O H 2 O H H net dipole moment

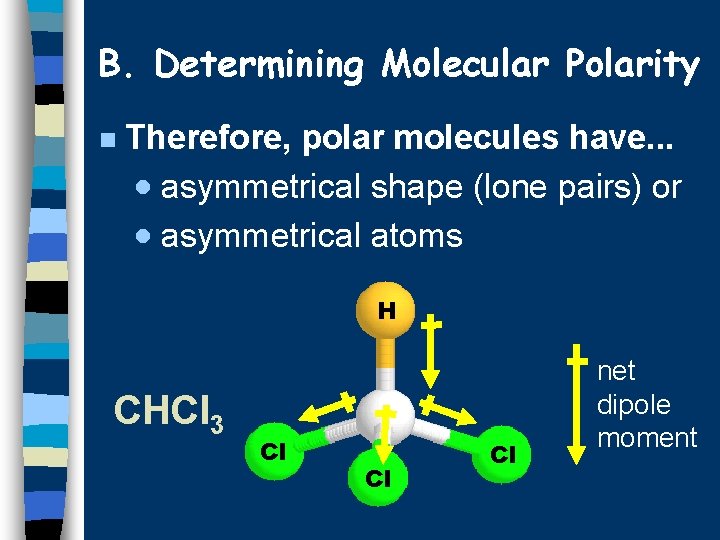
B. Determining Molecular Polarity n Therefore, polar molecules have. . . · asymmetrical shape (lone pairs) or · asymmetrical atoms H CHCl 3 Cl Cl Cl net dipole moment