Molecular Shapes and Hybrid Orbitals Why is molecular

Molecular Shapes and Hybrid Orbitals

Why is molecular shape important? l The shape of a molecule contributes to its properties and behavior l Changing the shape can change how the molecule functions l Extremely important in biological processes: Enzymes l Cancer-treatment drugs l

What accounts for the shape of a molecule? l Valence-Shell Electron-Pair Repulsion Theory (VSEPR Theory), pronounced “vesper”: l atoms align themselves to maximize the distance between the valence electron pairs https: //jahschem. wikispaces. com/VSEPR
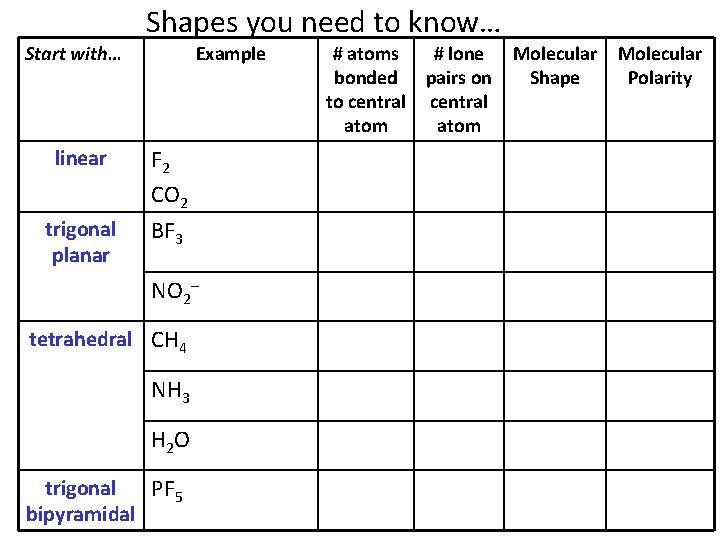
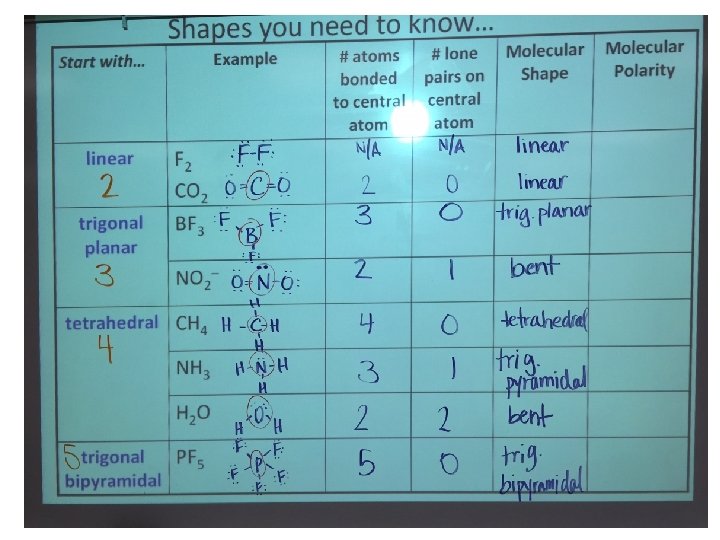
Shapes you need to know… Start with… Example linear F 2 CO 2 trigonal planar BF 3 NO 2– tetrahedral CH 4 NH 3 H 2 O trigonal PF 5 bipyramidal # atoms # lone bonded pairs on to central atom Molecular Shape Molecular Polarity
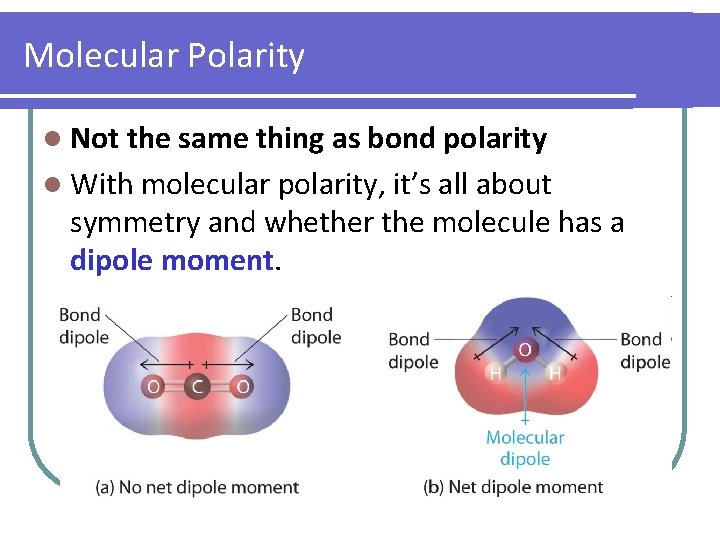
Molecular Polarity l Not the same thing as bond polarity l With molecular polarity, it’s all about symmetry and whether the molecule has a dipole moment.

Molecular Polarity l Two nonpolar molecule requirements: 1. Identical atoms bonded to central atom 2. No lone pairs of electrons on central atom
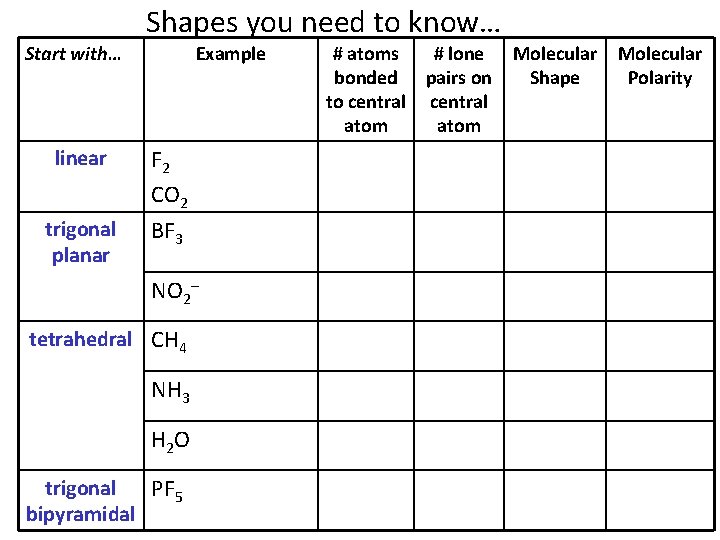
Shapes you need to know… Start with… Example linear F 2 CO 2 trigonal planar BF 3 NO 2– tetrahedral CH 4 NH 3 H 2 O trigonal PF 5 bipyramidal # atoms # lone bonded pairs on to central atom Molecular Shape Molecular Polarity
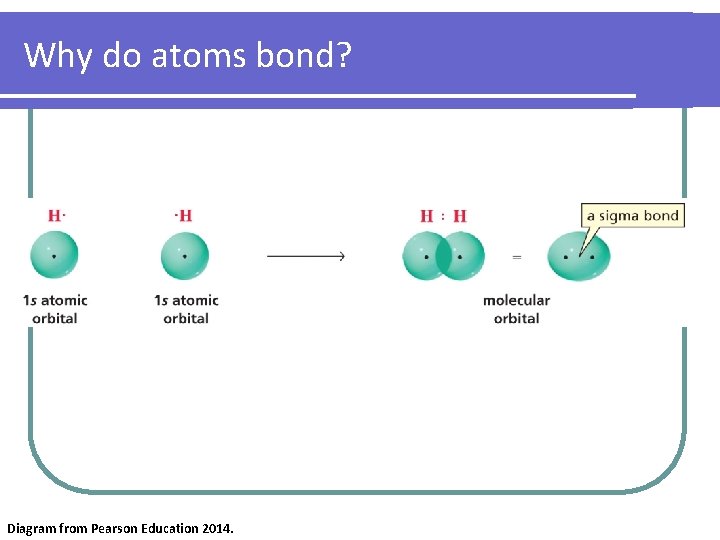
Why do atoms bond? Diagram from Pearson Education 2014.
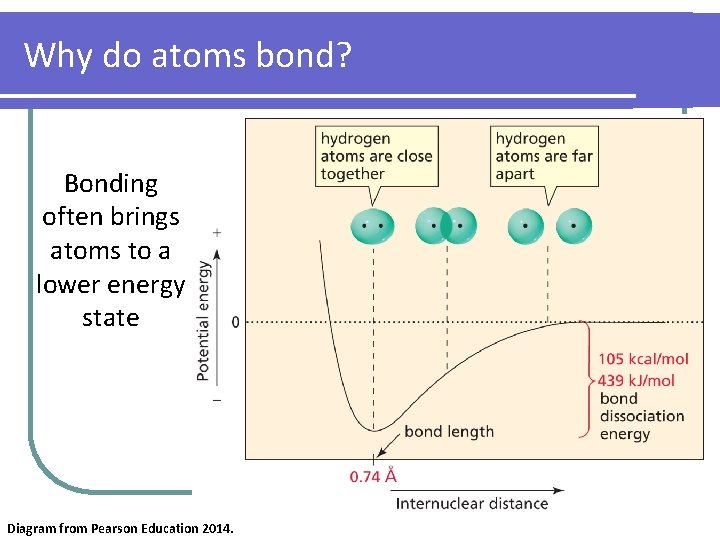
Why do atoms bond? Bonding often brings atoms to a lower energy state Diagram from Pearson Education 2014.
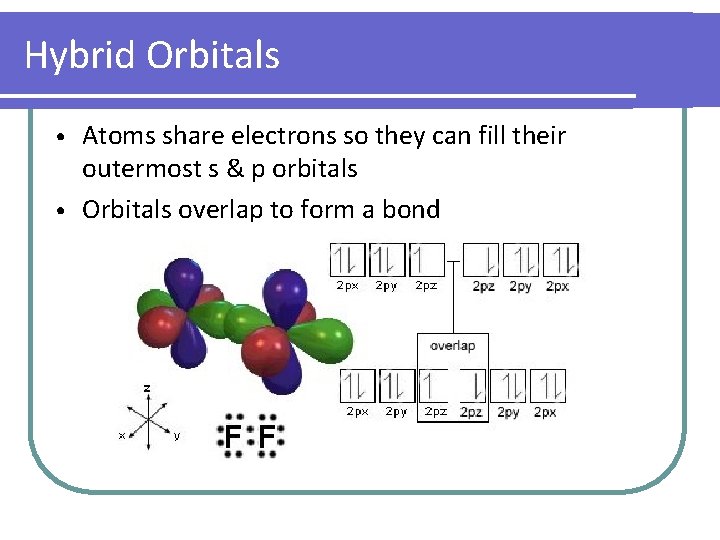
Hybrid Orbitals Atoms share electrons so they can fill their outermost s & p orbitals • Orbitals overlap to form a bond •
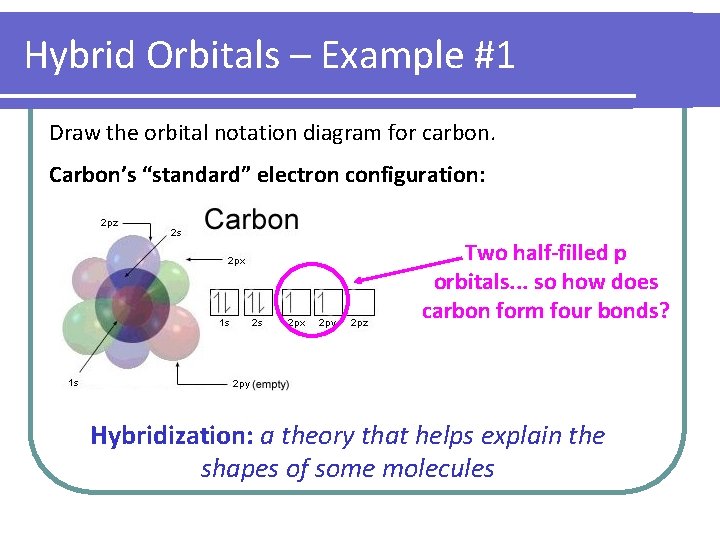
Hybrid Orbitals – Example #1 Draw the orbital notation diagram for carbon. Carbon’s “standard” electron configuration: Two half-filled p orbitals. . . so how does carbon form four bonds? Hybridization: a theory that helps explain the shapes of some molecules

Hybrid Orbitals – Example #1 l Carbon hybridizes its valence s and p orbitals Move an electron from the 2 s to the 2 p orbital We call these sp 3 hybrid orbitals (1 s orbital and 3 p orbitals mix)
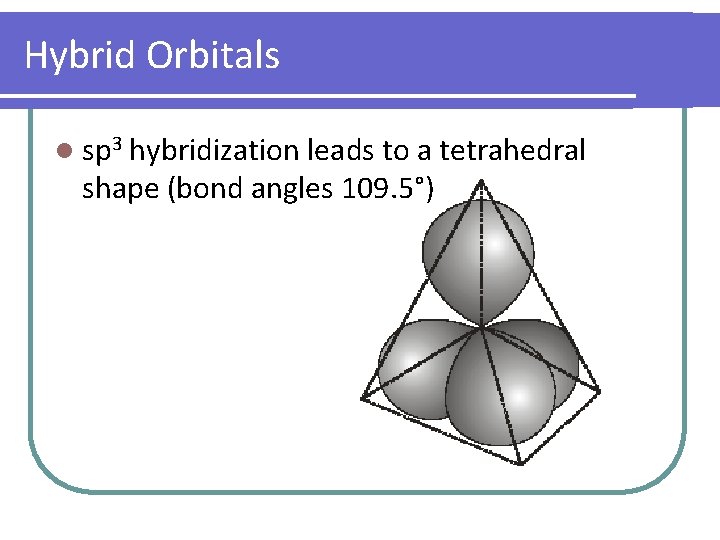
Hybrid Orbitals l sp 3 hybridization leads to a tetrahedral shape (bond angles 109. 5°)

Hybrid Orbitals – Example #2 Draw the orbital notation diagram for boron.
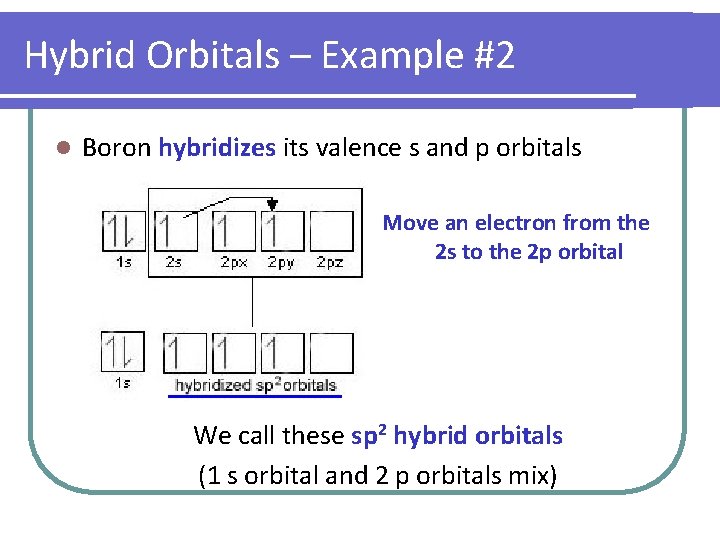
Hybrid Orbitals – Example #2 l Boron hybridizes its valence s and p orbitals Move an electron from the 2 s to the 2 p orbital We call these sp 2 hybrid orbitals (1 s orbital and 2 p orbitals mix)
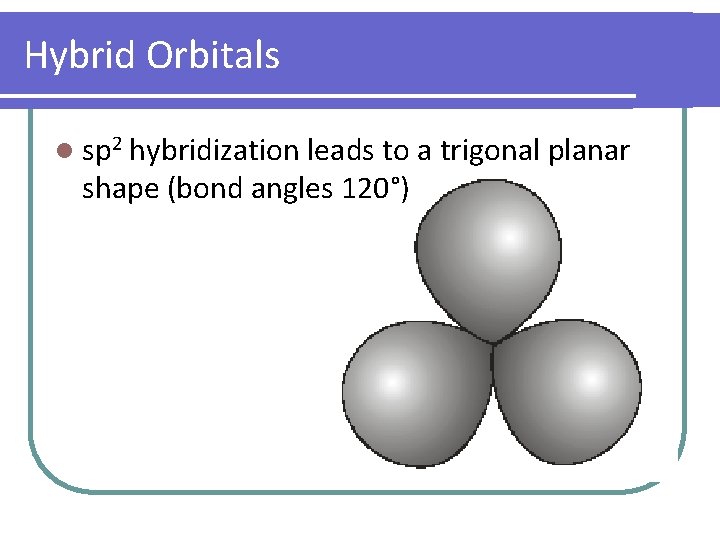
Hybrid Orbitals l sp 2 hybridization leads to a trigonal planar shape (bond angles 120°)

Hybrid Orbitals – Example #3 Draw the orbital notation diagram for beryllium.
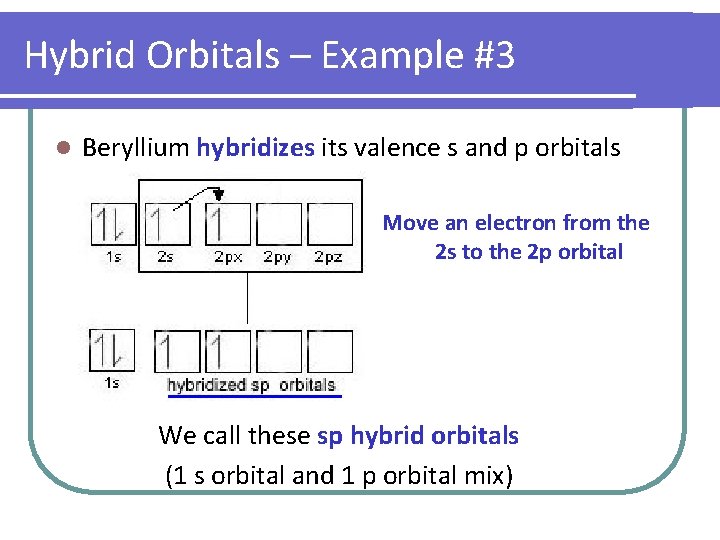
Hybrid Orbitals – Example #3 l Beryllium hybridizes its valence s and p orbitals Move an electron from the 2 s to the 2 p orbital We call these sp hybrid orbitals (1 s orbital and 1 p orbital mix)
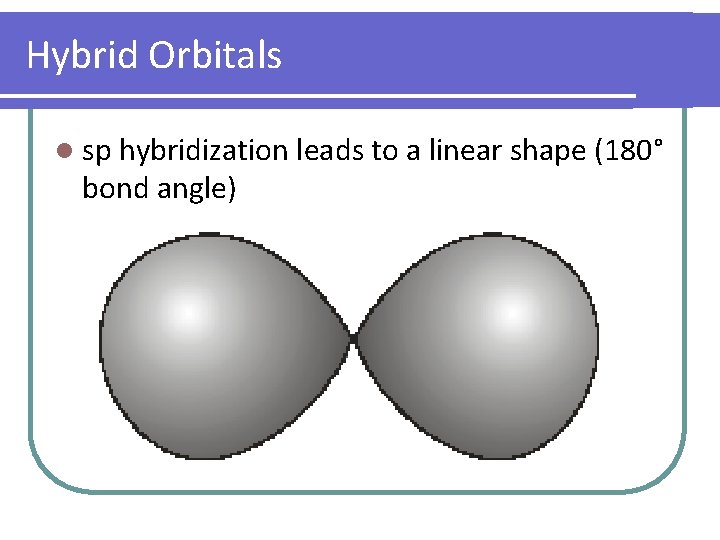
Hybrid Orbitals l sp hybridization leads to a linear shape (180° bond angle)
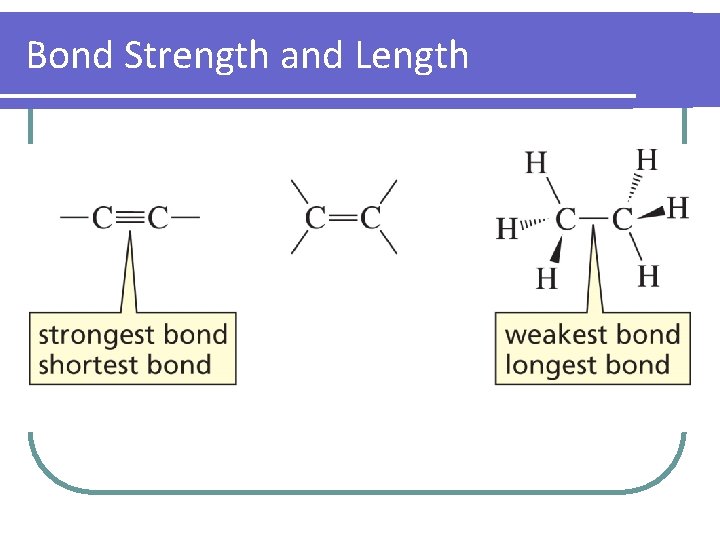
Bond Strength and Length
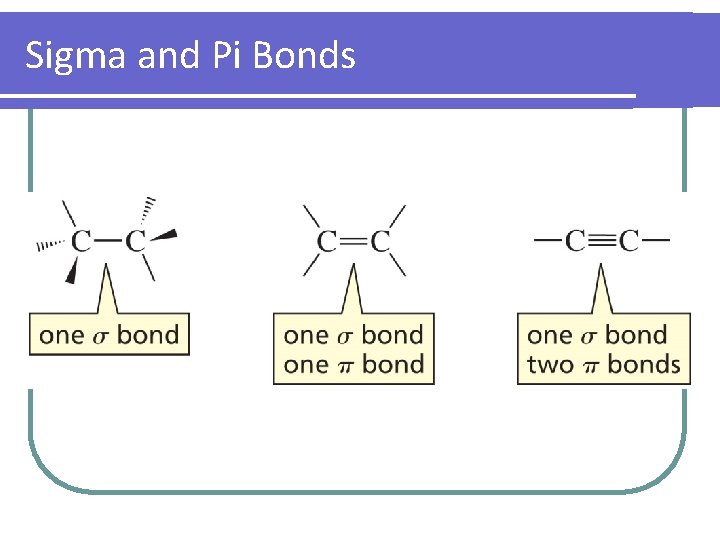
Sigma and Pi Bonds

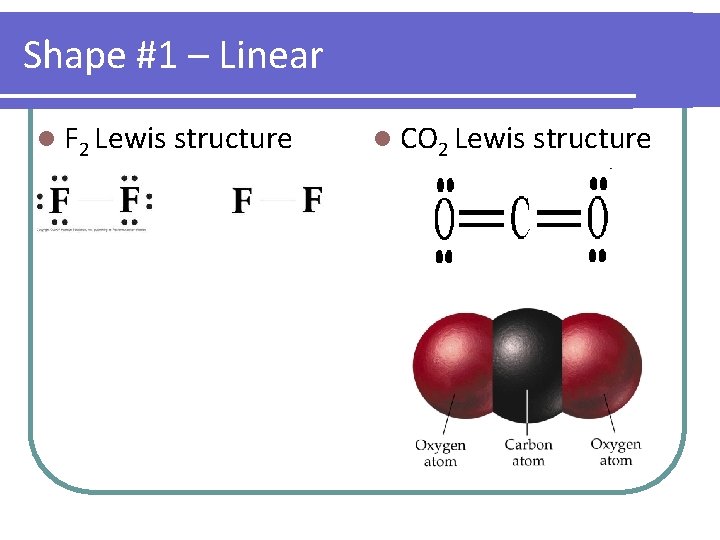
Shape #1 – Linear l F 2 Lewis structure l CO 2 Lewis structure
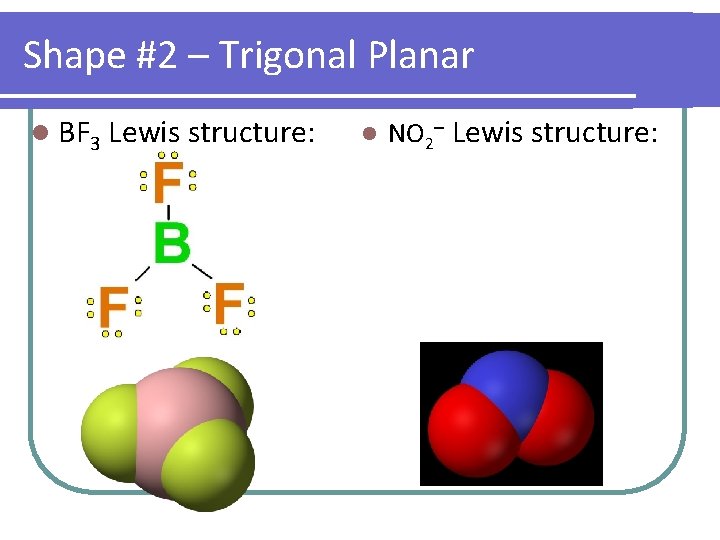
Shape #2 – Trigonal Planar l BF 3 Lewis structure: l NO 2– Lewis structure:
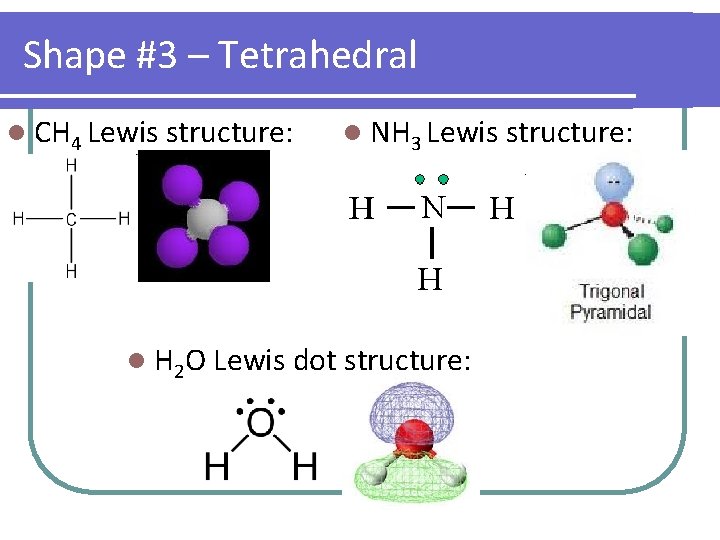
Shape #3 – Tetrahedral l CH 4 Lewis structure: l NH 3 Lewis structure: l H 2 O Lewis dot structure:
- Slides: 26