Molecular Selfassembled Monolayers on Au111 surface By Sung

Molecular Self-assembled Monolayers on Au{111} surface By: Sung Chou Advisors: Dr. Lloyd A. Bumm, Dr. Abhijit Biswas
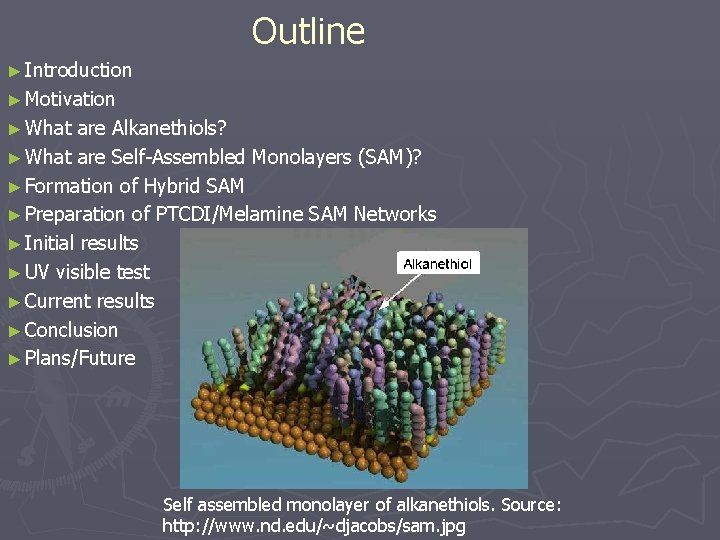
Outline ► Introduction ► Motivation ► What are Alkanethiols? ► What are Self-Assembled Monolayers (SAM)? ► Formation of Hybrid SAM ► Preparation of PTCDI/Melamine SAM Networks ► Initial results ► UV visible test ► Current results ► Conclusion ► Plans/Future Self assembled monolayer of alkanethiols. Source: http: //www. nd. edu/~djacobs/sam. jpg
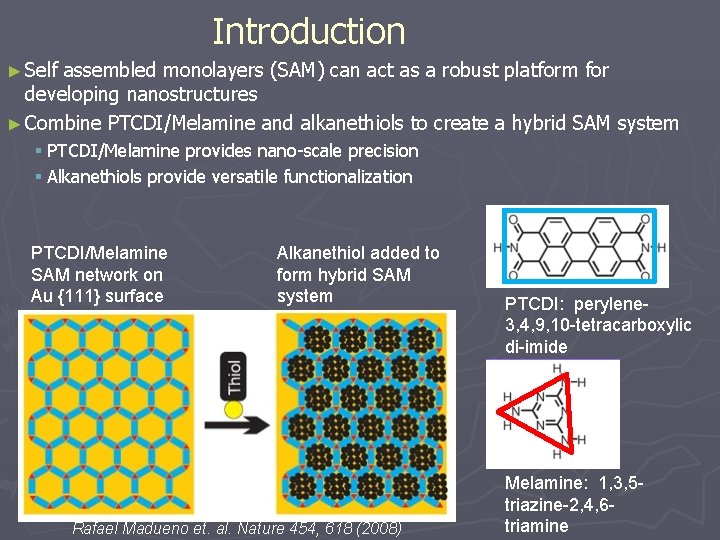
Introduction ► Self assembled monolayers (SAM) can act as a robust platform for developing nanostructures ► Combine PTCDI/Melamine and alkanethiols to create a hybrid SAM system § PTCDI/Melamine provides nano-scale precision § Alkanethiols provide versatile functionalization PTCDI/Melamine SAM network on Au {111} surface Alkanethiol added to form hybrid SAM system Rafael Madueno et. al. Nature 454, 618 (2008) PTCDI: perylene 3, 4, 9, 10 -tetracarboxylic di-imide Melamine: 1, 3, 5 triazine-2, 4, 6 triamine

Motivation ► SAMs provide a functional and versatile platform for nanostructures of an extended length scale § Nano and Microelectromechanical systems (NEMS and MEMS) § Modeling attachments to cell membranes ► Surface modifications § Hydrophobic coatings, e. g. for automobile windshields § Controlling electron transfer on electrodes in electrochemistry § Protecting metals from harsh substances ► Could function as a chemical sensor SAMs could potentially provide an easy to develop platform for MEMS, such as this ratcheting mechanism ~50μm across
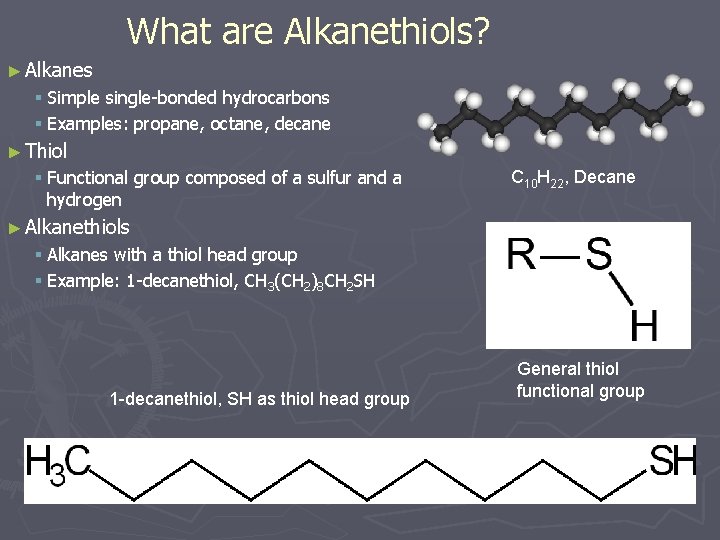
What are Alkanethiols? ► Alkanes § Simple single-bonded hydrocarbons § Examples: propane, octane, decane ► Thiol § Functional group composed of a sulfur and a hydrogen C 10 H 22, Decane ► Alkanethiols § Alkanes with a thiol head group § Example: 1 -decanethiol, CH 3(CH 2)8 CH 2 SH 1 -decanethiol, SH as thiol head group General thiol functional group
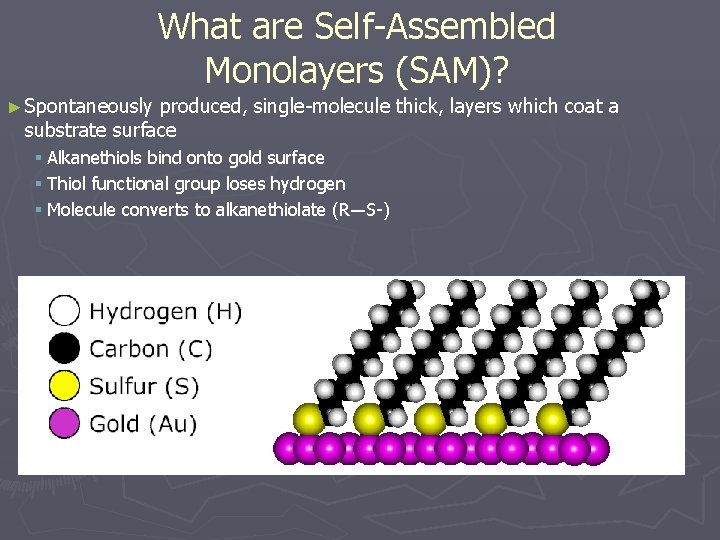
What are Self-Assembled Monolayers (SAM)? ► Spontaneously produced, single-molecule thick, layers which coat a substrate surface § Alkanethiols bind onto gold surface § Thiol functional group loses hydrogen § Molecule converts to alkanethiolate (R―S-)
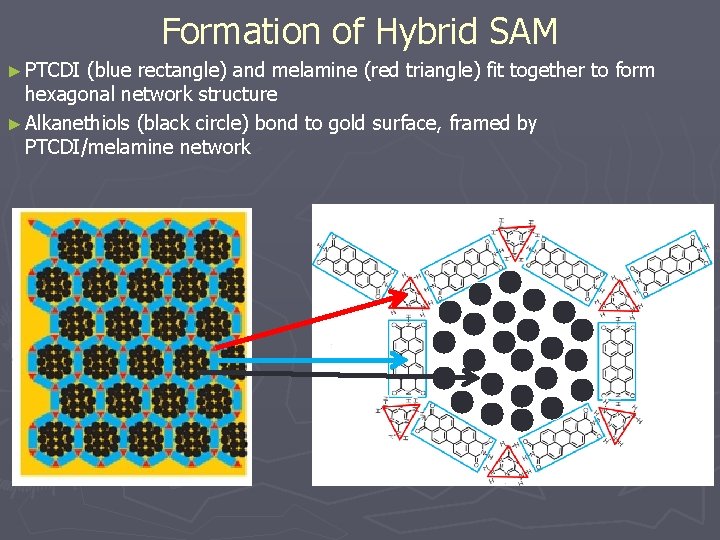
Formation of Hybrid SAM ► PTCDI (blue rectangle) and melamine (red triangle) fit together to form hexagonal network structure ► Alkanethiols (black circle) bond to gold surface, framed by PTCDI/melamine network

Preparation of PTCDI/Melamine SAM Networks

Preparation of PTCDI/Melamine SAM Networks ► Step 1: § The PTCDI and Melamine are both mixed with DMF (Dimethylformamide) in separate test tubes § Both solutions shaken over night by the shaker machine § Centrifuge both solutions about 30 minutes § Extract the clear liquid, leaving behind undissolved solids § Repeat the process until little or no solid substance can be seen in either solution Undissolved PTCDI or Melamine Uniformly dissolved solution
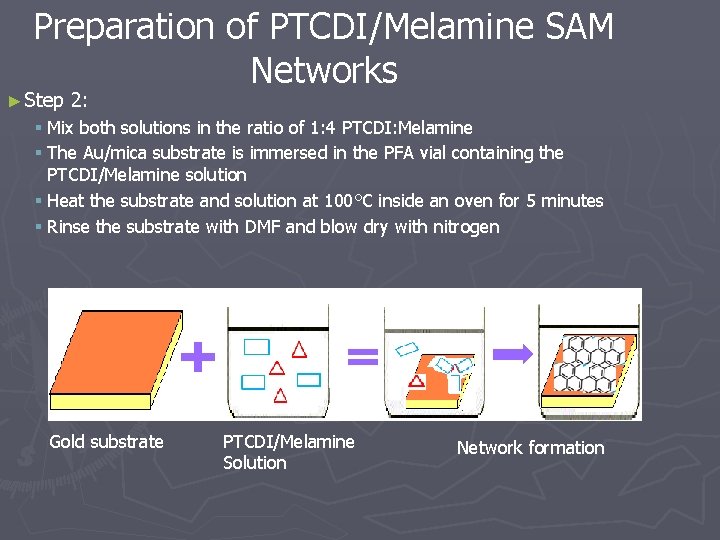
Preparation of PTCDI/Melamine SAM Networks ► Step 2: § Mix both solutions in the ratio of 1: 4 PTCDI: Melamine § The Au/mica substrate is immersed in the PFA vial containing the PTCDI/Melamine solution § Heat the substrate and solution at 100°C inside an oven for 5 minutes § Rinse the substrate with DMF and blow dry with nitrogen Gold substrate PTCDI/Melamine Solution Network formation

Initial results
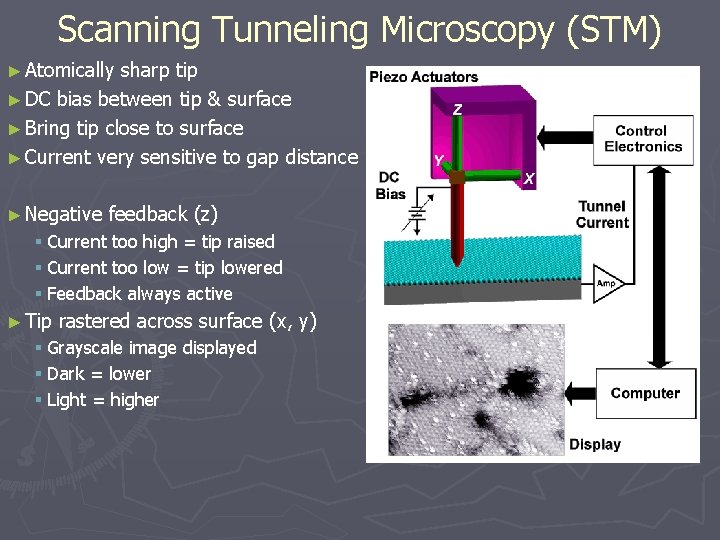
Scanning Tunneling Microscopy (STM) ► Atomically sharp tip ► DC bias between tip & surface ► Bring tip close to surface ► Current very sensitive to gap distance ► Negative feedback (z) § Current too high = tip raised § Current too low = tip lowered § Feedback always active ► Tip rastered across surface (x, y) § Grayscale image displayed § Dark = lower § Light = higher
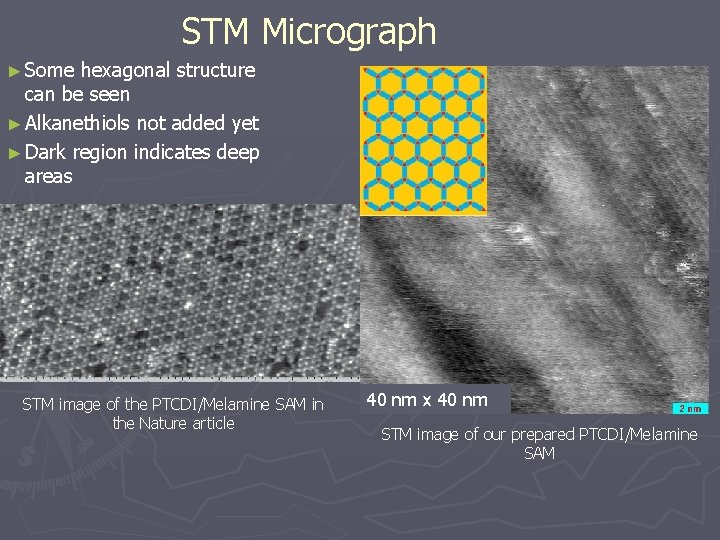
STM Micrograph ► Some hexagonal structure can be seen ► Alkanethiols not added yet ► Dark region indicates deep areas STM image of the PTCDI/Melamine SAM in the Nature article 40 nm x 40 nm STM image of our prepared PTCDI/Melamine SAM
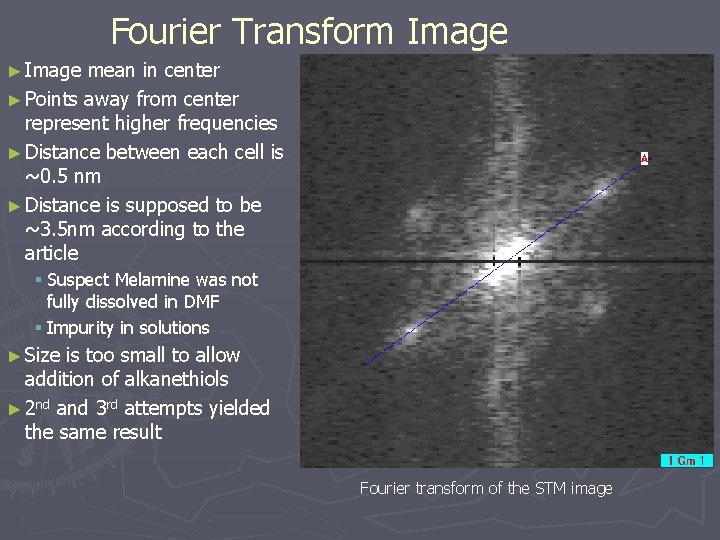
Fourier Transform Image ► Image mean in center ► Points away from center represent higher frequencies ► Distance between each cell is ~0. 5 nm ► Distance is supposed to be ~3. 5 nm according to the article § Suspect Melamine was not fully dissolved in DMF § Impurity in solutions ► Size is too small to allow addition of alkanethiols ► 2 nd and 3 rd attempts yielded the same result Fourier transform of the STM image

The Ultra violet visible test

UV Visible Test ► UV-Vis test performed to check concentration of dissolved solids ► UV light shined on sample Sample Light source Separate detector Diffraction grating Detector mirrors
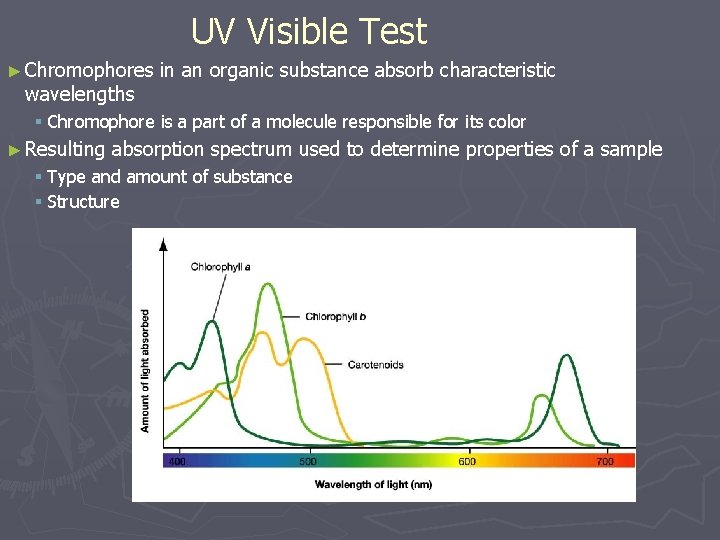
UV Visible Test ► Chromophores wavelengths in an organic substance absorb characteristic § Chromophore is a part of a molecule responsible for its color ► Resulting absorption spectrum used to determine properties of a sample § Type and amount of substance § Structure
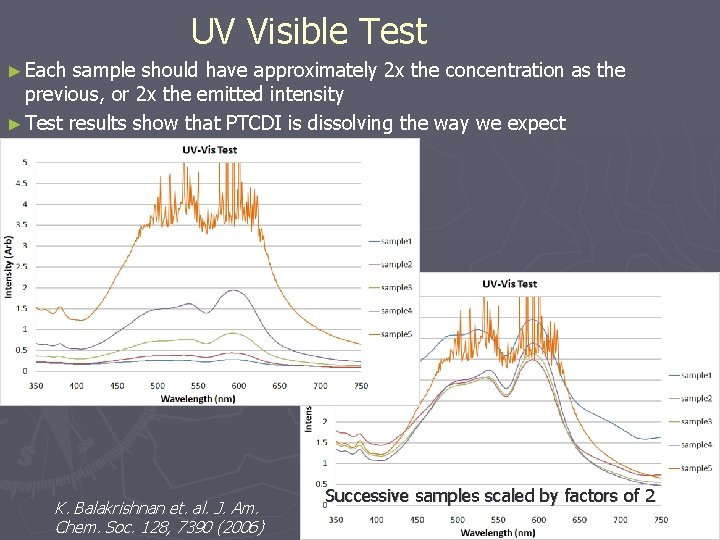
UV Visible Test ► Each sample should have approximately 2 x the concentration as the previous, or 2 x the emitted intensity ► Test results show that PTCDI is dissolving the way we expect K. Balakrishnan et. al. J. Am. Chem. Soc. 128, 7390 (2006) Successive samples scaled by factors of 2

Current results
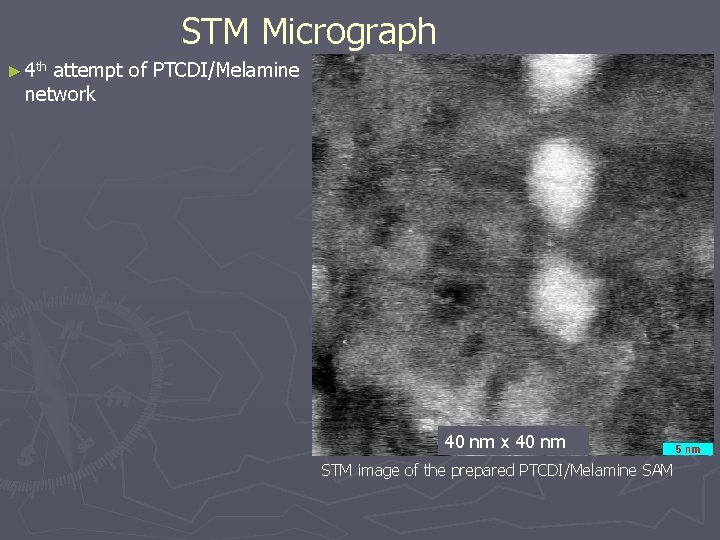
STM Micrograph ► 4 th attempt of PTCDI/Melamine network 40 nm x 40 nm STM image of the prepared PTCDI/Melamine SAM
![Fourier Transform Image measured distance is 0. 488 nm FFT image[pm] ► The Distance[Gm-1] Fourier Transform Image measured distance is 0. 488 nm FFT image[pm] ► The Distance[Gm-1]](http://slidetodoc.com/presentation_image_h2/05fbc10435d1f3053d9f25b66e27b8e6/image-21.jpg)
Fourier Transform Image measured distance is 0. 488 nm FFT image[pm] ► The Distance[Gm-1]

Conclusion ► We were able to produce saturated solutions of PTCDI-Melamine along with their optical characterization ► The experiment was performed 4 times ► Our results are fairly consistent despite our effort to obtain the original result ► Some vital information might have been left out in the paper ► Efforts to contact the research group failed ? Trial Spacing (nm) 1 0. 500 2 0. 503 3 0. 497 4 0. 488

Plans/Future ► UV-Vis characterization of saturated solutions of PTCDI and Melamine in order to examine the absorption properties ► Optimization of the SAM preparation conditions: § ratio of mixing § growth temperature § time, etc. ► Insertion ü of alkanethiol and other less-studied thiols and functional molecules (e. g. octylthiocyanates, Azides) in the PTCDI/Melamine network ► Putting PTCDI/Melamine SAM network on Flat Gold Nano Particles (FGNPs) and study optical properties (absorption/fluorescence). ü

Acknowledgements ► Dr. Lloyd A Bumm, Dr. Abhijit Biswas, Daminda Dahanayaka, Chris Schroeder, Matt Whiteway THANK YOU
- Slides: 24