Molecular properties of the antiaromatic species cyclopentadienone C









- Slides: 22

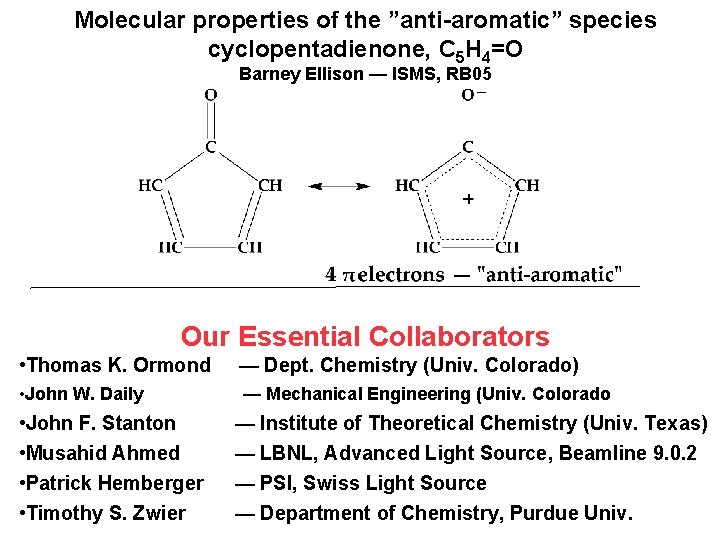
Molecular properties of the ”anti-aromatic” species cyclopentadienone, C 5 H 4=O Barney Ellison — ISMS, RB 05 Our Essential Collaborators • Thomas K. Ormond — Dept. Chemistry (Univ. Colorado) • John W. Daily — Mechanical Engineering (Univ. Colorado • John F. Stanton • Musahid Ahmed • Patrick Hemberger • Timothy S. Zwier — Institute of Theoretical Chemistry (Univ. Texas) — LBNL, Advanced Light Source, Beamline 9. 0. 2 — PSI, Swiss Light Source — Department of Chemistry, Purdue Univ.

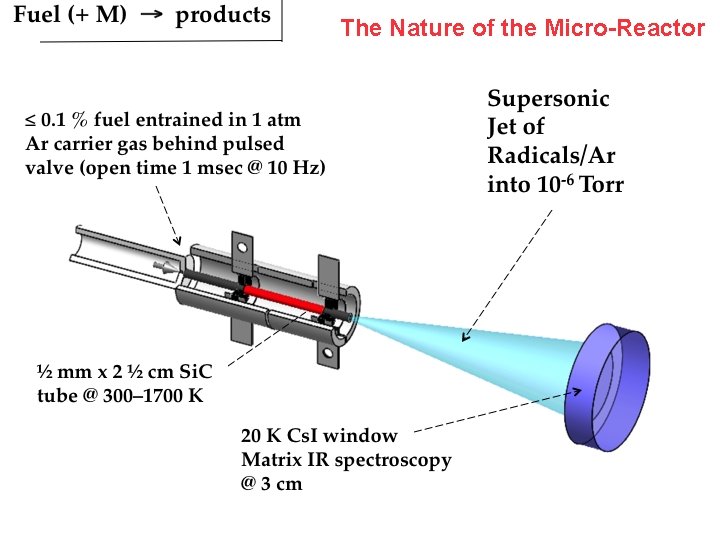
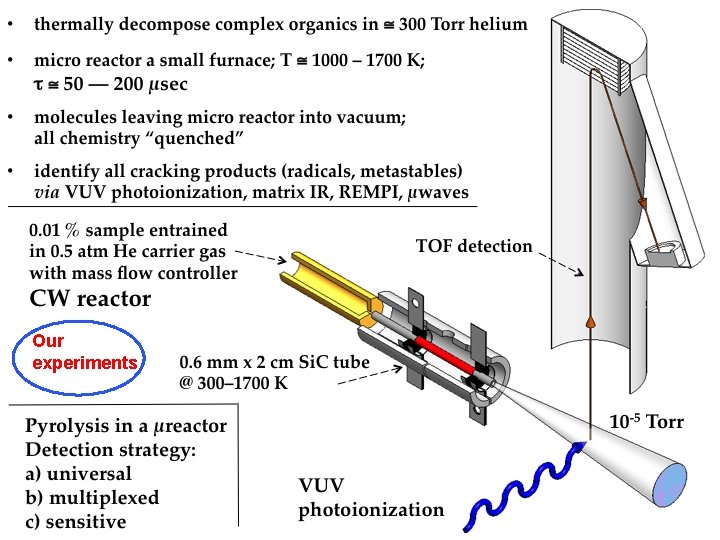
• Transportation fuels developed at engineering/chemistry interface — “Real fuels” are complicated • Engineering models are correspondingly complex — Mechanisms include 100 s of intermediates and 1000 s of reactions. Under-determined. • We use a hot micro-reactor to study thermal cracking of complex fuels. Goal identify all decomposition products (atoms, radicals, metastables) formed in first 100 µsec.

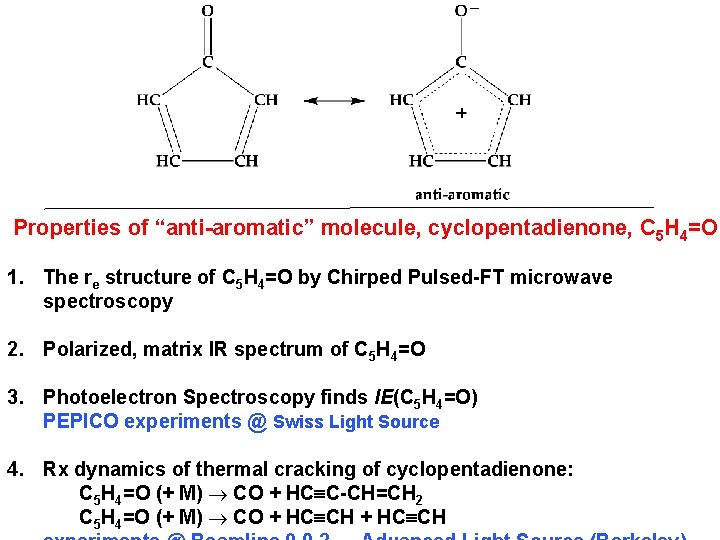
Properties of “anti-aromatic” molecule, cyclopentadienone, C 5 H 4=O 1. The re structure of C 5 H 4=O by Chirped Pulsed-FT microwave spectroscopy 2. Polarized, matrix IR spectrum of C 5 H 4=O 3. Photoelectron Spectroscopy finds IE(C 5 H 4=O) PEPICO experiments @ Swiss Light Source 4. Rx dynamics of thermal cracking of cyclopentadienone: C 5 H 4=O (+ M) CO + HCºC-CH=CH 2 C 5 H 4=O (+ M) CO + HCºCH

The Nature of the Micro-Reactor

Our experiments

Prof. John Daily (Mechanical Engineering) “not a Chen nozzle but a tubular reactor” Micro-reactor complement to shock tube 1 mm x 3 cm linguini 1 mm x 3 cm Si. C tube @ 300 K — 1700 K resistively heated by @ 10 Amps CFD modeling — numerical solutions Navier-Stokes equations Guan et al. , “The Properties of a Micro-Reactor for the Study of the Unimolecular

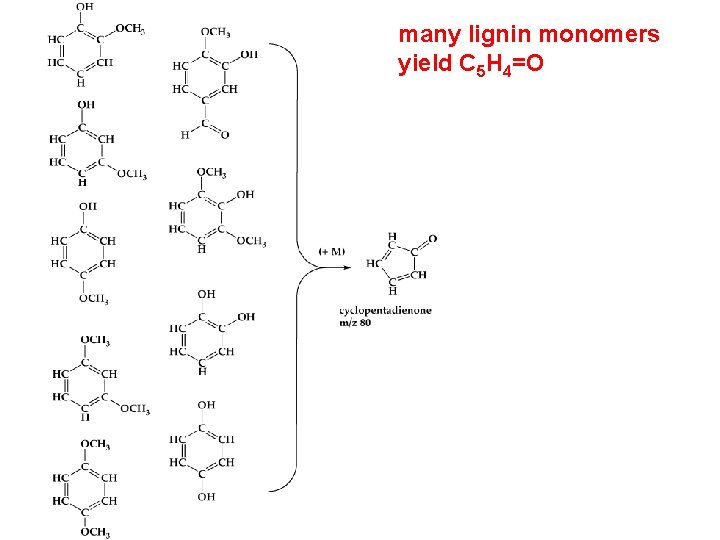
many lignin monomers yield C 5 H 4=O

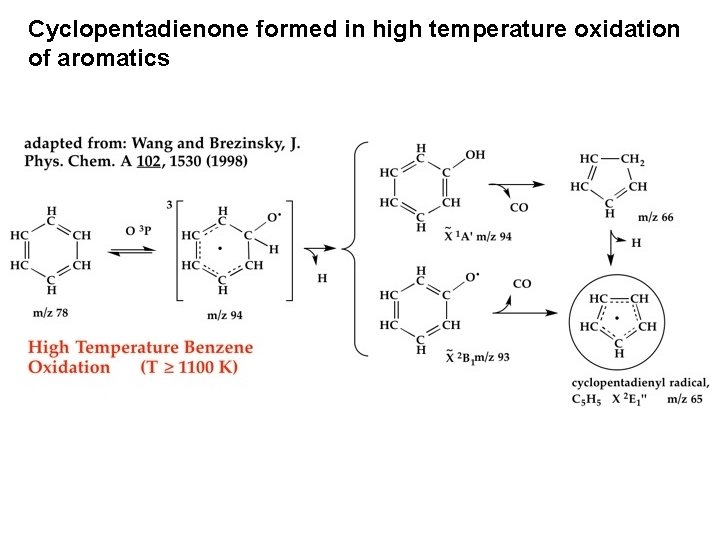
Cyclopentadienone formed in high temperature oxidation of aromatics


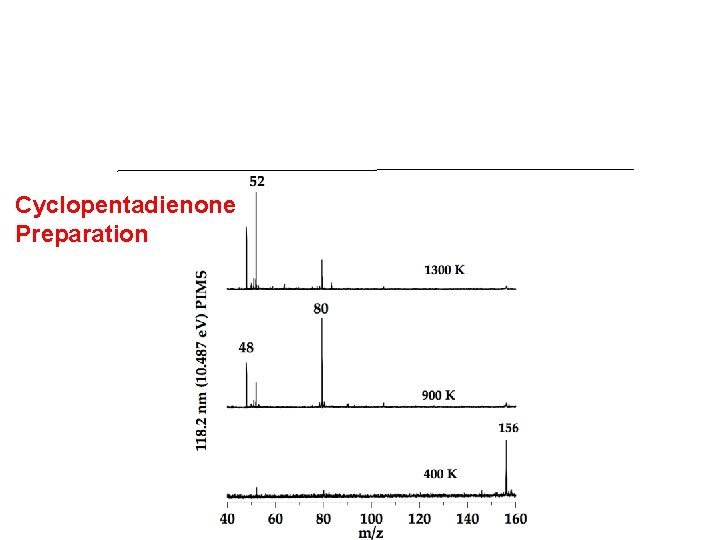
Cyclopentadienone Preparation

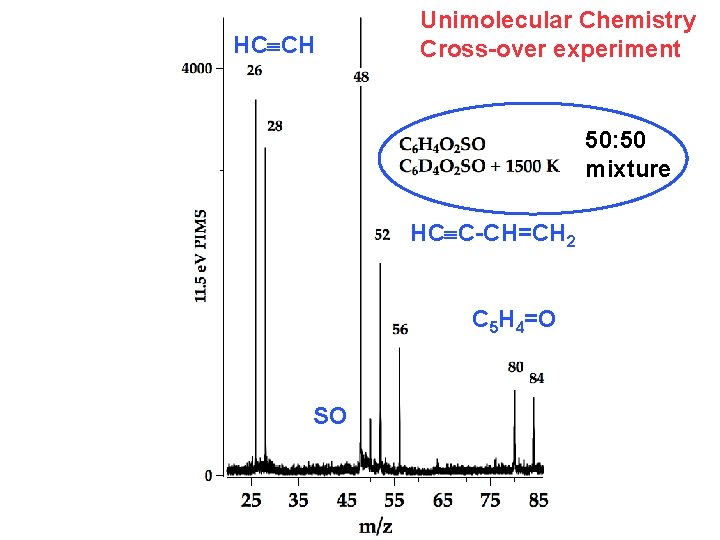
HCºCH Unimolecular Chemistry Cross-over experiment 50: 50 mixture HCºC-CH=CH 2 C 5 H 4=O SO

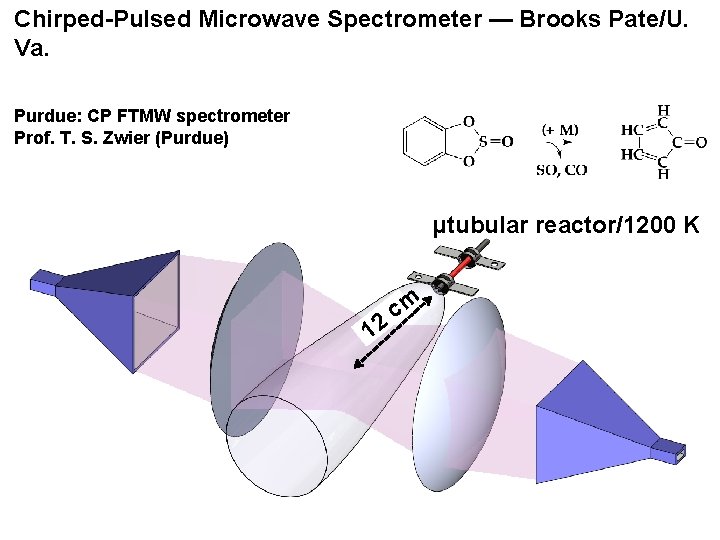
Chirped-Pulsed Microwave Spectrometer — Brooks Pate/U. Va. Purdue: CP FTMW spectrometer Prof. T. S. Zwier (Purdue) µtubular reactor/1200 K 12 cm

8 isotopically substituted cyclopentadienone species observed by CP-FTMW spectroscopy (Purdue) 12 C 5 H 4=O, 12 C 5 D 4=O C 1 13 C 5 H 4=O, C 2 13 C 5 H 4=O, C 3 13 C 5 H 4=O C 1 13 C 5 D 4=O, C 2 13 C 5 D 4=O, C 3 13 C 5 D 4=O Microwave spectra were interpreted by CCSD(T) ab initio electronic structure calculations (Univ. Texas)

Kidwell, Vaquero-Vara, Ormond, Buckingham, Zhang, Nimlos, Daily, Dian, Stanton, Ellison, & Zwier, “Chirped-Pulse Fourier Transform Microwave Spectroscopy Coupled with a Hyperthermal Reactor: Structural Determination of the “Anti-Aromatic” Molecule Cyclopentdienone, ” J. Phys. Chem. Letts. 5, 2201 -2207 (2014) molecule is exactly planar: inertial defect, ∆e = 0

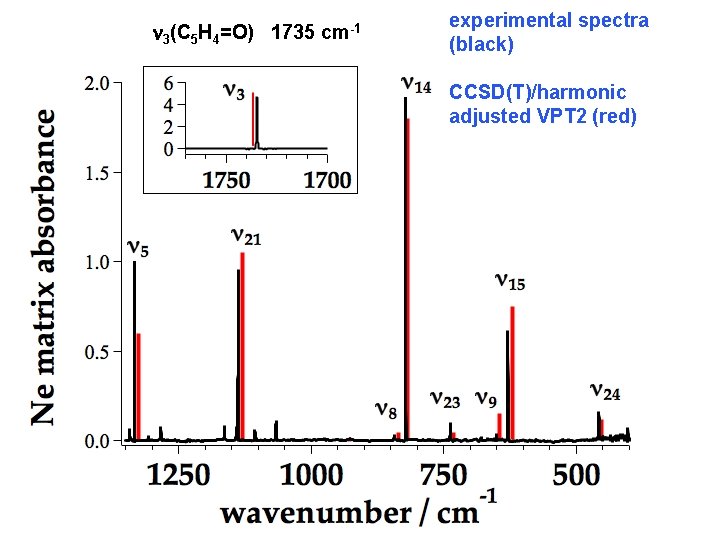
C 5 H 4=O Polarized Infrared Spectrum Matrix (Ne) isolation of C 5 H 4=O and C 5 D 4=O achieved @ 4 K Gvib = 9 a 1 3 a 2 4 b 1 8 b 2 20/24 fundamentals assigned for C 5 H 4=O 17/24 fundamentals assigned for C 5 D 4=O ~1 X A 1 C 5 H 4=O • a 1 modes: n 1 = 3107, n 2 = (3100, 3099), n 3 = 1735, n 5 = 1333, n 7 = 952, n 8 = 843 and n 9 = 651. • inferred a 2 modes are: n 10 = 933, and n 11 = 722. • b 1 modes are: n 13 = 932, n 14 = 822, and n 15 = 629. • b 2 fundamentals are: n 17 = 3143, n 18 = (3078, 3076) n 19 = (1601 or 1595), n 20 = 1283, n 21 = 1138, n 22 = 1066, n 23 = 738, and n 24 = 458. Ormond, Scheer, Nimlos, Daily, Ellison, and Stanton, “Polarized Infrared Spectroscopy of Cyclopentadienone, an Important Biomass Decomposition Product, ” J. Phys. Chem. A, 118 708 – 718 (2014).

n 3(C 5 H 4=O) 1735 cm-1 experimental spectra (black) CCSD(T)/harmonic adjusted VPT 2 (red)

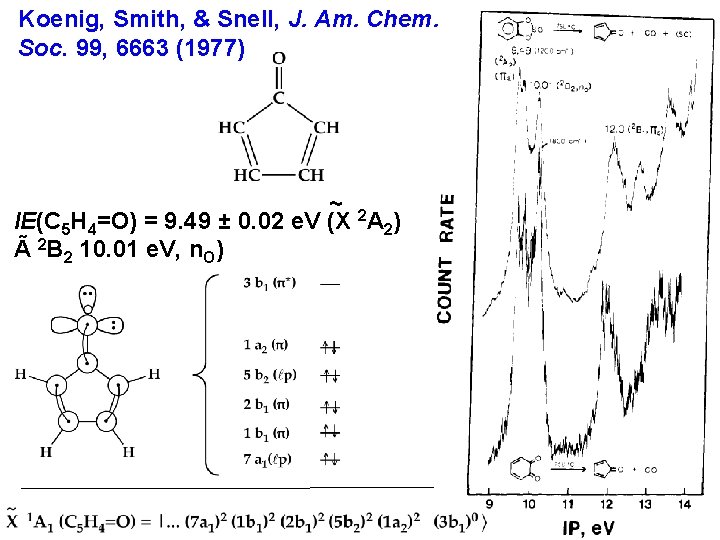
Koenig, Smith, & Snell, J. Am. Chem. Soc. 99, 6663 (1977) ~ 2 IE(C 5 H 4=O) = 9. 49 ± 0. 02 e. V (X A 2) Ã 2 B 2 10. 01 e. V, n. O)

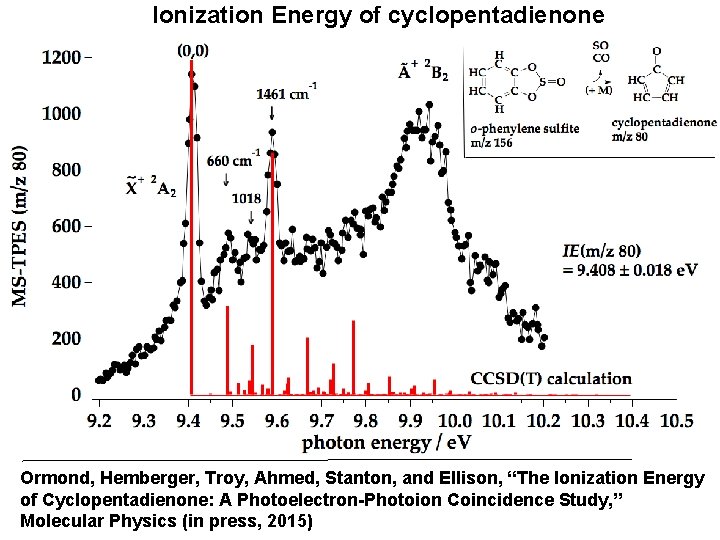
Ionization Energy of cyclopentadienone Ormond, Hemberger, Troy, Ahmed, Stanton, and Ellison, “The Ionization Energy of Cyclopentadienone: A Photoelectron-Photoion Coincidence Study, ” Molecular Physics (in press, 2015)


Chemistry How does C 5 H 4=O thermally crack apart? To measure channels? Beer’s Law M + hw M+ + e I = Ioe-ns(n)z Ion current = j+ = (Io – I) = Io(1 - e-ns(n)z) @ ns(n)z Io or S 26+ = n. HCCH s. HCCH(E) C F(E) S 52+ = n. HCC-CH=CH 2 F(E’) s. HCC-CH=CH 2(E’) C F(E’) Possible to measure n(HCºCH), n(HCºCH-CH=CH 2) with tunable VUV radiation (synchrotron)

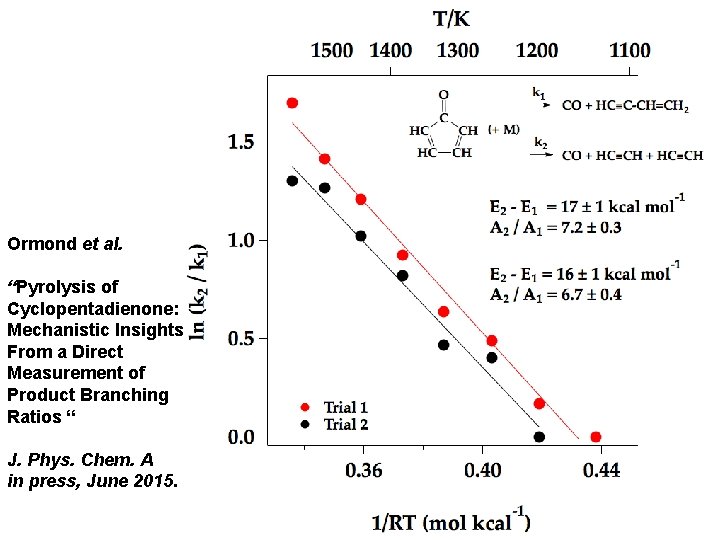
Ormond et al. “Pyrolysis of Cyclopentadienone: Mechanistic Insights From a Direct Measurement of Product Branching Ratios “ J. Phys. Chem. A in press, June 2015.

Molecular properties of C 5 H 4=O now well known: • Chirped Pulsed-FT microwave re structure • Polarized Infrared Spectra (Ne matrix) Gvib = 9 a 1 3 a 2 4 b 1 8 b 2 20/24 fundamentals • IE(C 5 H 4=O) = 9. 408 ± 0. 018 e. V assigned for C 5 H 4=O • EA(C 5 H 4=O) = 1. 06 ± 0. 01 e. V Sanov et al. J. Phys. Chem. A (2014) • C H =O CO + 2 HCºCH or HCºC-CH=CH measured