Molecular physiology of protein kinases and phosphatases Molecules





































- Slides: 48

Molecular physiology of protein kinases (and phosphatases): Molecules, mechanisms, medicines… Figure 3. From: Eck & Yun. Biochem. Biophys. Acta 1804(3): 559 -566, 2010. Freely accessible at http: //www. ncbi. nlm. nih. gov/pm c/articles/PMC 2859716/ Michael Eck Dana-Farber Cancer Institute Harvard Medical School

~518 protein kinases in the human genome… Kinase signal transduction touches essentially every aspect of human physiology. Kinases of central importance in regulating cell growth and proliferation, metabolism, motility, differentiation, gene expression, intercellular communication…. Kinases have become important drug targets in diverse therapeutic areas, including: Cancer Immunology & inflammation Metabolism Infectious disease Human Kinome poster available at kinase. com/human/kinome/

Outline: Overview – the reaction, structure Mechanism of regulation by activation loop phosphorylation – Insulin receptor as an example Modular signaling domains, SH 2, SH 3, PH, PTB Epidermal growth factor receptor (EGFR) signaling – a representative example of a receptor tyrosine kinase signaling network EGFR in Cancer Non-receptor tyrosine kinases: Src, Abl SHP 2 – a tyrosine phosphatase

Kinases catalyze transfer of the g-phosphate of ATP to hydroxyl acceptor on substrate protein: Ser-OH or Thr-OH for serine/threonine kinases Tyr-OH for Tyrosine kinases

Tyrosine kinases are (almost) unique to higher eukaryotes: evolved to facilitate communication/coordination between cells/tissues. They account for ~90 kinases in human genome. Ser/thr kinases and signaling pathways tend to be conserved in all eukaryotes. Tyrosine kinase signaling pathways interconnect extensively with ser/thr kinase cascades. For example, growth factor receptors activate ras/map kinase pathway. Protein kinases interconnect with virtually every regulatory mechanism in the cell: Heterotrimeric Gproteins, small G proteins, small-molecule second messengers, Ca++, lipid kinases, phospholipases…

The effects of phosphorylation are remarkably diverse. A few examples: 1) enzyme activation or inhibition [including kinases, phosphatases and many other enzymes; activation of glycogen phosphorylase by protein kinase A (PKA) and phosphorylase kinase is a classic example] 2) Induce protein-protein interactions 3) Induce protein dimerization - for example to activate transcription factors 4) Alter subcellular localization; nuclear translocation 5) Promote or prevent protein degradation: control recognition by ubiquitin ligases

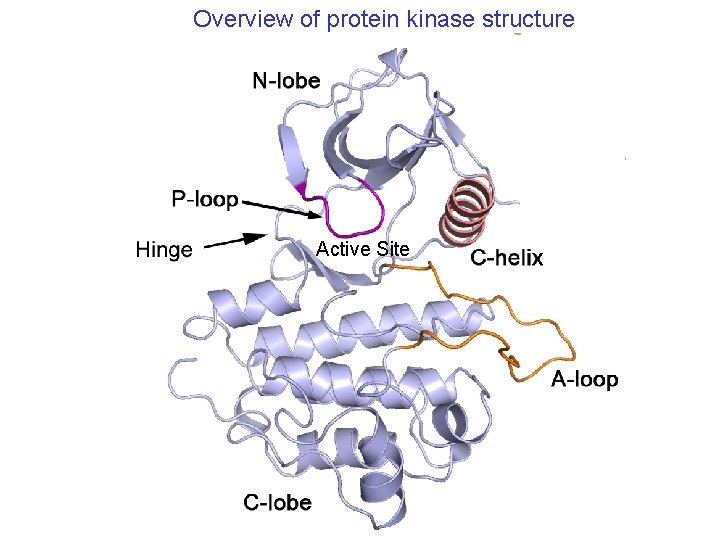
Overview of protein kinase structure Active Site

Kinases: 1. Usually regulated by phosphorylation of activation loop 2. Autoinhibition typical, often by non-phosphorylated act. loop 3. Diverse mechanisms of inactivation/regulation, both within kinase domain and by associated domains and sequences a. inactivation deconstructs or blocks active site (many variations) b. active kinases look very similar in active site 4. Recognize substrate peptides in extended form, b-sheet interactions in insulin receptor tyrosine kinase (IRTK; only Tyr kinase/substrate complex available). 5. Sequence specificity varies, determined both at level of kinase domain and by associated protein-interaction domains.

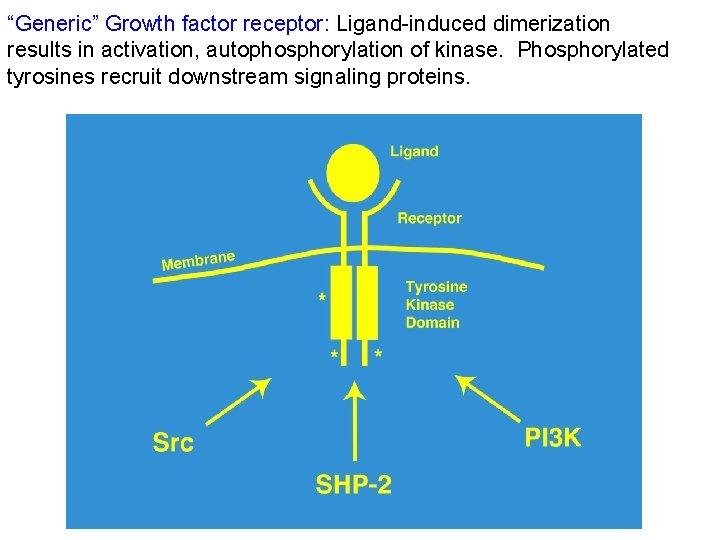
“Generic” Growth factor receptor: Ligand-induced dimerization results in activation, autophosphorylation of kinase. Phosphorylated tyrosines recruit downstream signaling proteins.

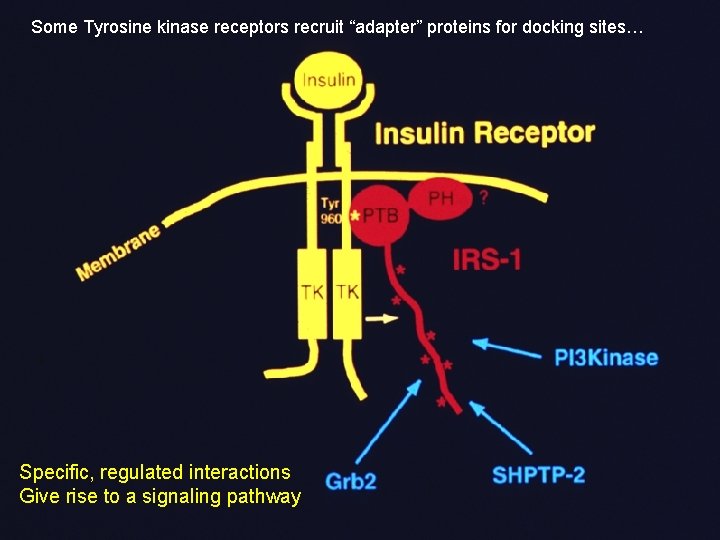
Some Tyrosine kinase receptors recruit “adapter” proteins for docking sites… Specific, regulated interactions Give rise to a signaling pathway

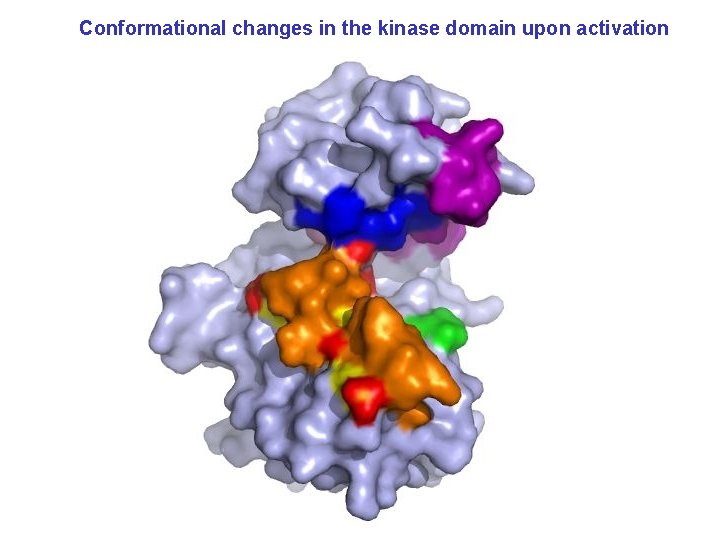
Figure 4. From: Hubbard. EMBO J. 16: 5572 – 5581, 1997 Freely accessible at www. nature. com/emboj/journal/v 16/n 18/fig_ta b/7590532 a_F 4. html#figure-title

Conformational changes in the kinase domain upon activation

Modular domains are key elements of kinase signaling pathways. They recognize specific sequences in partner proteins, often phosphorylation-dependent. Many have evolved multiple classes of specificity… Figure 2. From: Pawson and Scott. Science 278(5346): 2075 -2080, 1997 Freely accessible at www. sciencemag. org/content/278/5346/2075. full

Assembly of cell regulatory systems through protein interaction domains Figure 1. From Pawson and Nash. Science 300(5618): 445 -52, 2003. Freely accessible at www. sciencemag. org/content/300/5618/445. full

Figure 1 A. From: Poy et al. Mol. Cell 4: 555 -561, 1999 Freely accessible at http: //www. sciencedirect. com/science/ article/pii/S 1097276500802063 Two binding pockets revealed by solvent-accessible surface of Lck SH 2 domain architecture: Central sheet flanked by helices Conserved p. Tyr binding pocket on one side of sheet. Specificity pocket on the other side binds 3 -5 residues C-terminal to p. Tyr. Figure 4 A. From: Eck et al. Nature 362: 87 -91, 1993 Available at http: //www. nature. com/nature /journal/v 362/n 6415/abs/3620 87 a 0. html

A two-pronged plug: p. YEEI-motif peptide inserts into pockets Figure 2. From: Boggon & Eck. Oncogene 23: 7918– 7927, 2004 Freely accessible at www. nature. com/onc/journal/v 23/n 48/full/1208081 a. html

Two PH/PTB mechanisms for joint phospholipid/protein recognition IRS-1: One domain for each job Figure 5 B. From: Dhe-Paganon et al. Proc. Natl. Acad. Sci. 96: 8378 -8383, 1999 Freely accessible at http: //www. pnas. org/content/96/1 5/8378. full. pdf Figure 2 A, top. From: Dhe-Paganon et al. Proc. Natl. Acad. Sci. 96: 8378 -8383, 1999 Freely accessible at http: //www. pnas. org/content/96/1 5/8378. full. pdf Dab 1: One domain does it all…

The Erb. B signaling network A simplified diagram showing the complicated Erb. B signaling network. EGFR as the most important Erb. B member, has the broadest target spectrum and so plays a very important role in a lot of cell processes, including cell growth, differentiation, adhesion, migration and apoptosis. Most of these processes are more or less connected with carcinogenesis. From: Yarden and Sliwkowski. Nat. Rev. Mol. Cell Biol. 2: 127 -137, 2001 Available at www. nature. com/nrm/journal/v 2/n 2/full/nrm 0201_127 a. html

Structure of EGFR extracellular domain in the absence of EGF reveals autoinhibition… Figure 1 A. From: Ferguson et al. Molec. Cell 11: 507 -517, 2003 Freely accessible at http: //www. sciencedirect. com/science/article/pii/S 10972 76503000479

Comparison of active and inactive conformations and a model for activation… Figure 4. From: Ferguson et al. Molec. Cell 11: 507 -517, 2003 Freely accessible at http: //www. sciencedirect. com/science/article/pii/S 10972 76503000479

How dimerization of the extracellular portion of the EGFR induces activation of the kinase domain: formation of an asymmetric dimer Figure 7 B. From: Zhang et al. Cell 125: 1137 -1149, 2006 Freely accessible at www. cell. com/fulltext/S 0092 -8674%2806%2900584 -8 .

An allosteric mechanism for activation of the kinase domain of epidermal growth factor receptor Figure 4 A. From: Zhang et al. Cell 125: 1137 -1149, 2006 Freely accessible at www. cell. com/fulltext/S 0092 -8674%2806%2900584 -8

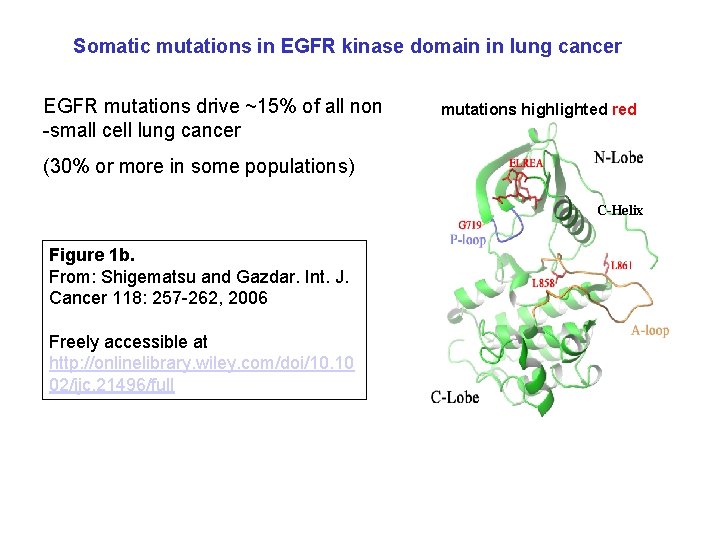
Somatic mutations in EGFR kinase domain in lung cancer EGFR mutations drive ~15% of all non -small cell lung cancer mutations highlighted red (30% or more in some populations) C-Helix Figure 1 b. From: Shigematsu and Gazdar. Int. J. Cancer 118: 257 -262, 2006 Freely accessible at http: //onlinelibrary. wiley. com/doi/10. 10 02/ijc. 21496/full

Mutations activate the kinase by disrupting the inactive conformation WT/Lapatinib L 858 R/Iressa (Wood et al. Cancer Res 64: 6652 -6659, 2004) 2007) (Yun et al. Cancer Cell 11: 217 -227, Figure 2. From: Yun et al. Cancer Cell 11: 217 -227, 2007 Freely accessible at www. sciencedirect. com/science/article/pii/S 1535610807000281

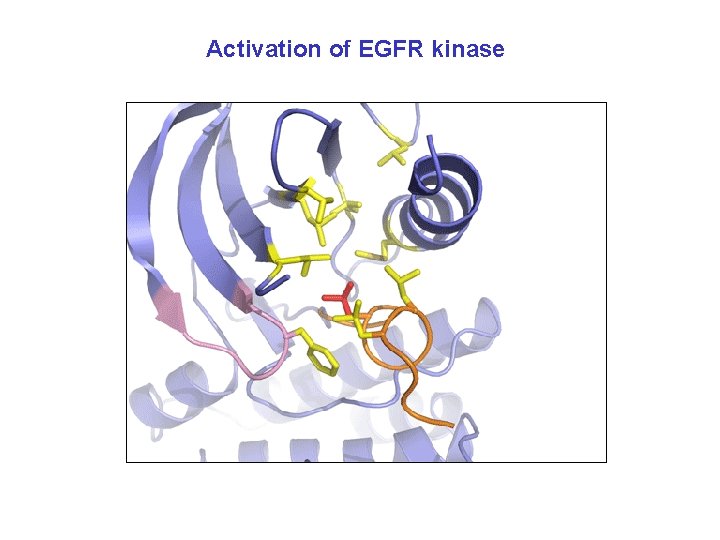
Activation of EGFR kinase

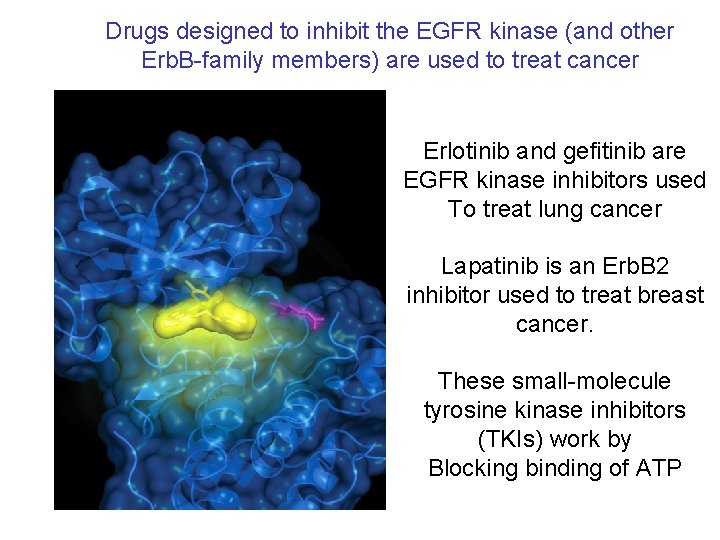
Drugs designed to inhibit the EGFR kinase (and other Erb. B-family members) are used to treat cancer Erlotinib and gefitinib are EGFR kinase inhibitors used To treat lung cancer Lapatinib is an Erb. B 2 inhibitor used to treat breast cancer. These small-molecule tyrosine kinase inhibitors (TKIs) work by Blocking binding of ATP

Protein kinase inhibitors approved for clinical use (in US) Table 1. From: Johnson LN. Q. Rev. Biophys. 42: 1 -40, 2009 Available at http: //www. esaimcocv. org/action/display. Issue? decade=2000&jid= QRB&volume. Id=42&issue. Id=01&iid=5827576

Monoclonal antibodies directed against the extracellular domain of the EGFR are also used to treat cancer, especially cancers that involve over expression of the receptor. A quick look at three different modes of antibody inhibition: compare cetuximab/EGFR, Traztuzumab (herceptin)/Erb. B 2, and pertuzumab/Erb. B 2 Figures 3 A & 4 A From: Li et al. Cancer Cell 7: 301 -311, 2005 Freely accessible at http: //www. sciencedirect. com/science/article/pii/S 1535610805 000905 Blocks EGF binding site

Three different modes of antibody inhibition: compare cetuximab/EGFR, Traztuzumab (herceptin)/Erb. B 2, and pertuzumab/Erbb 2 Figure 1. From: Cho et al. Nature 421: 756 -760, 2003 Freely accessible at http: //www. nature. com/nature/journal/v 421/n 6924/full/ nature 01392. html Blocks dimerization (indirectly)

Three different modes of antibody inhibition: compare cetuximab/EGFR, Traztuzumab (herceptin)/Erb. B 2, and pertuzumab/Erb. B 2 Figure 6. From: Franklin et al. Cancer Cell 5: 317 -328, 2004 Freely accessible at http: //www. sciencedirect. com/science/article/pii/S 1535610804000832 Pertuzumab binds Domain II and directly blocks heterodimerization

EGFR is just one type of Receptor Tyrosine kinase… Figure 1. From: Lemmon and Schlessinger Cell: 141: 1117 -1134, 2010 Freely accessible at http: //www. sciencedirect. com/science/article/pii/S 00928674 10006653

Other tyrosine kinases are entirely cytoplasmic; these are called non-receptor tyrosine kinases… Small protein domains/modules important for protein-protein interactions in signal transduction. They form adaptors to create a signaling network. They can also regulate catalytic activity when they are coupled to kinases And other signaling enzymes (phosphatases, lipid kinases, phospholipases…) Figure 13. 25 From: Branden and Tooze. Introduction to Protein Structure (2 nd ed. ). 1999. Garland Science. ISBN: 9780815323051

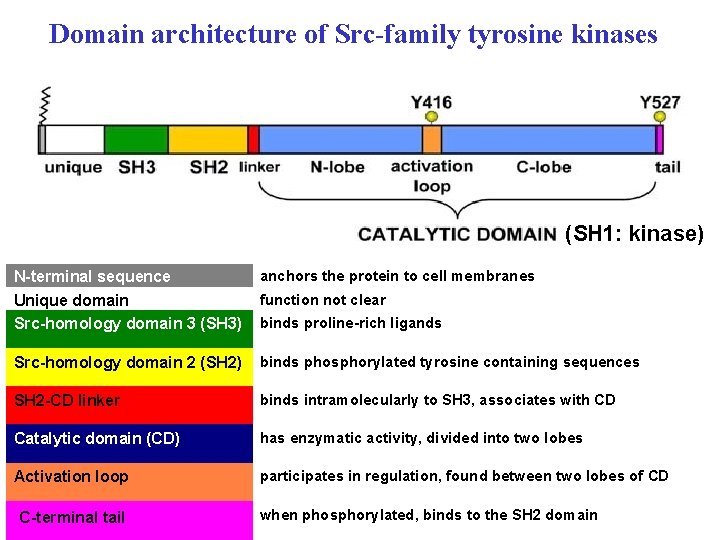
Domain architecture of Src-family tyrosine kinases REGION FUNCTION (SH 1: kinase) N-terminal sequence Unique domain Src-homology domain 3 (SH 3) anchors the protein to cell membranes Src-homology domain 2 (SH 2) binds phosphorylated tyrosine containing sequences SH 2 -CD linker binds intramolecularly to SH 3, associates with CD Catalytic domain (CD) has enzymatic activity, divided into two lobes Activation loop participates in regulation, found between two lobes of CD C-terminal tail function not clear binds proline-rich ligands when phosphorylated, binds to the SH 2 domain

Autoinhibited Src kinase Figure 3. From: Xu et al. Mol. Cell 3: 629 -638, 1999 Freely accessible at http: //www. sciencedirect. com/science/article/pii/S 1097 276500803561

A model for Src activation Figure 7. From: Xu et al. Molec. Cell 3: 629 -638, 1999 Freely accessible at http: //www. sciencedirect. com/science/article/pii/ S 1097276500803561

Common themes, variations. . . A multidomain regulatory architecture reused. Figure 1. From: Harrison. Cell 112: 737 -740, 2003 Freely accessible at http: //crystal. harvard. edu/lib-sch/Harrison-2003 -CELL 112 -737. pdf

Figure 2 A (right) From Nagar et al. Cell 112: 859 -871, 2003 Freely accessible at http: //www. sciencedirect. com/science/article/pii/S 0 092867403001946 Chronic myelogenous leukemia (CML) is caused A 9: 22 chromosomal translocation – the Philadelphia Chromosome. This results in expression of a fusion Between two proteins, BCR and the Abl tyrosine kinase In BCR-Abl, the “cap” region and N-terminal myristoylation are absent…yielding a de-repressed kinase… Figure 1 A. From Nagar et al. Cell 112: 859 -871, 2003 Freely accessible at http: //www. sciencedirect. com/science/article/pii/S 0092867403001946

Pak Domain Structure Couples Cdc 42 Binding motif with a kinase domain Figure 1. From: Lei et al. Cell 102: 387 -397, 2000 Freely accessible at https: //crystal. harvard. edu/libsch/Lei. M-00 -Cell-102 -387. pdf

Direct activation of a kinase by a guanine nucleotide binding protein… Pak autoinhibition and release of inhibition by Cdc 42 Figure 6. From: Lei et al. Cell 102: 387 -397, 2000 Freely accessible at https: //crystal. harvard. edu/libsch/Lei. M-00 -Cell-102 -387. pdf A mechanism for intercommunication in signaling pathways…

Protein phosphatases reverse the effects of kinases. They catalyze the removal of the phosphate group: -Ser-OH or Thr-OH for serine/threonine kinases -Tyr-OH for Tyrosine kinases Figure 1. From: Tonks and Neel. Curr. Opin. Cell Biol. 13: 182 -195, 2001 Available at http: //www. sciencedirect. com/science/article/ pii/S 0955067400001964


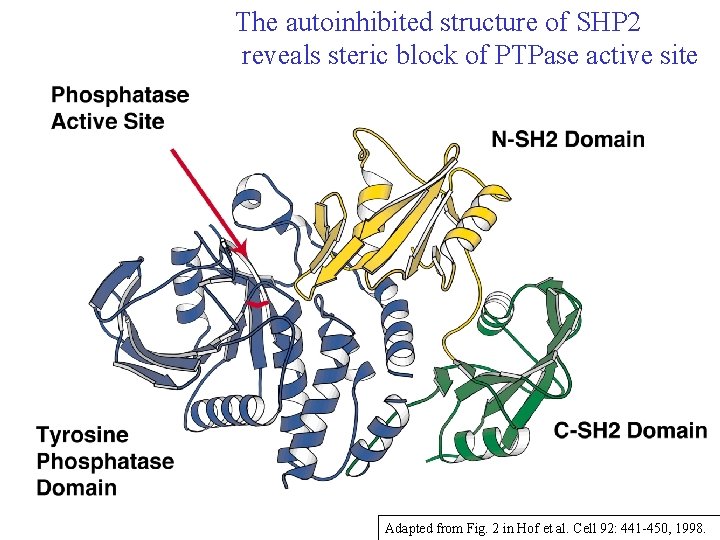
The autoinhibited structure of SHP 2 reveals steric block of PTPase active site Adapted from Fig. 2 in Hof et al. Cell 92: 441 -450, 1998.

Figure 4. From: Hof et al. Cell 92: 441 -450, 1998 Freely accessible at http: //www. sciencedirect. com/science/article/pii/S 009286740 0809381

Figure 6 A. From: Hof et al. Cell 92: 441 -450, 1998 Freely accessible at http: //www. sciencedirect. com/science/article/pii/S 009286740 0809381

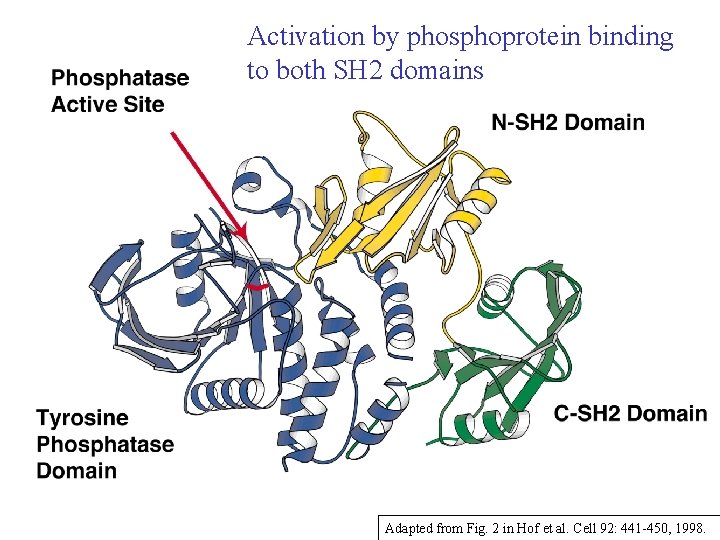
Activation by phosphoprotein binding to both SH 2 domains Adapted from Fig. 2 in Hof et al. Cell 92: 441 -450, 1998.

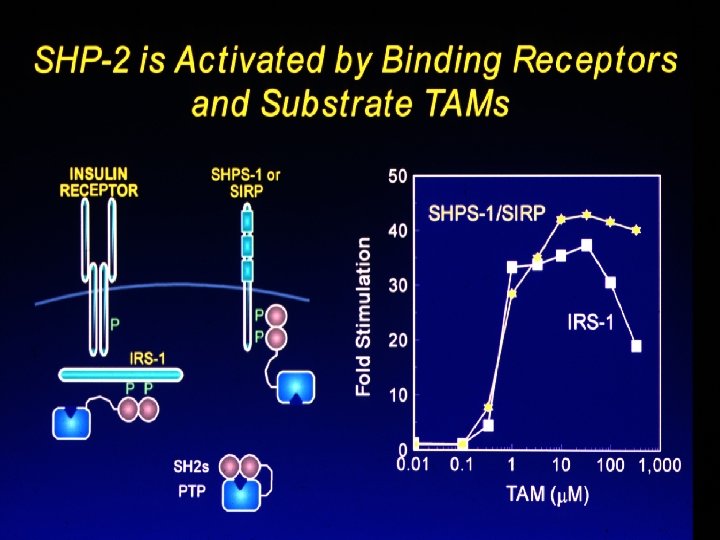
SHP-2 Tyrosine Phosphatase: N-terminal SH 2 domain blocks active site ==> catalytically inactive Phosphopeptide binding changes the shape of the N-SH 2 domain so that it no longer “fits” the PTPase ==> inhibition is released N + C binding contributes entropically

Common themes. . . • Separate domains for recognition and catalysis • In Src, SHP 2 see how targeting domains also serve as inhibitors of enzyme activity • Proper intermolecular contacts release inhibitory intramolecular interactions • Result: intrinsic coupling of activity with targeting (the essence of signal transduction).

The Erb. B signaling network Figure 1. From: Yarden and Sliwkowski, Nat. Rev. Mol. Cell Biol. 2: 127 -137, 2001 Available at http: //www. nature. com/nrm/journal/v 2/n 2/fu ll/nrm 0201_127 a. html