Molecular Orbitals from 2 p Atomic Orbitals The

Molecular Orbitals from 2 p Atomic Orbitals The 2 pz orbitals overlap in head-to-head fashion, so these bonds are. . . s bonds. -- the corresponding MOs are: s 2 p and s*2 p y x z The other 2 p orbitals (i. e. , 2 px and 2 py) overlap in sideways fashion, so the bonds are. . . p bonds. -- the corresponding MOs are: p 2 p (two of these) and p*2 p (two of these)

Rule 3 above suggests that, from low energy to high, the 2 p MOs SHOULD follow the order: LOW s 2 p < p*2 p < s*2 p ENERGY HIGH ENERGY General energy-level diagrams for MOs of second-row homonuclear diatomic molecules. . . don’t fit on this slide. (And so, they’re on the next one… …if that’s all right with you. ) (And if not, you can… …quit and go make pancakes. )
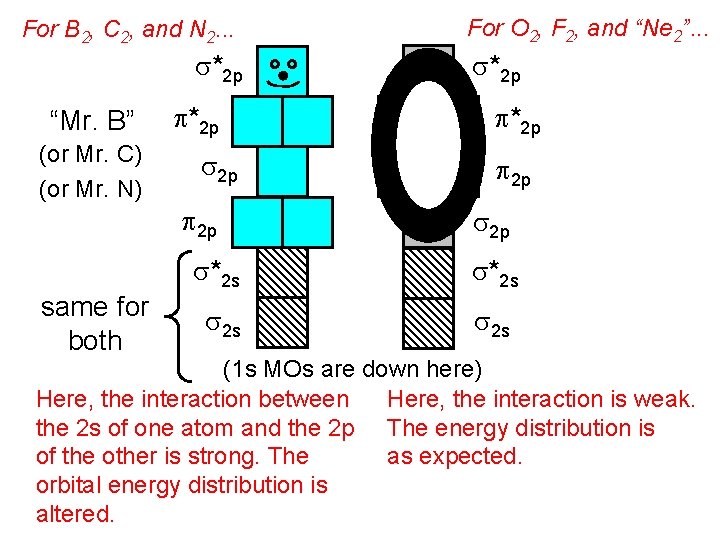
For B 2, C 2, and N 2. . . s*2 p “Mr. B” (or Mr. C) (or Mr. N) same for both p*2 p s 2 p p 2 p For O 2, F 2, and “Ne 2”. . . s*2 p p 2 p s*2 s s 2 s (1 s MOs are down here) Here, the interaction is weak. Here, the interaction between the 2 s of one atom and the 2 p The energy distribution is as expected. of the other is strong. The orbital energy distribution is altered.

paramagnetism: describes the attraction of molecules with unpaired e– to a magnetic field diamagnetism: describes substances with no unpaired e– ~ “dielectron”) (“di-” = two; diamagnetic = -- such substances are VERY weakly (almost unnoticeably) repelled by a magnetic field Use the energy diagrams above to tell if diatomic species are paramagnetic or diamagnetic. paramagnetism of liquid oxygen
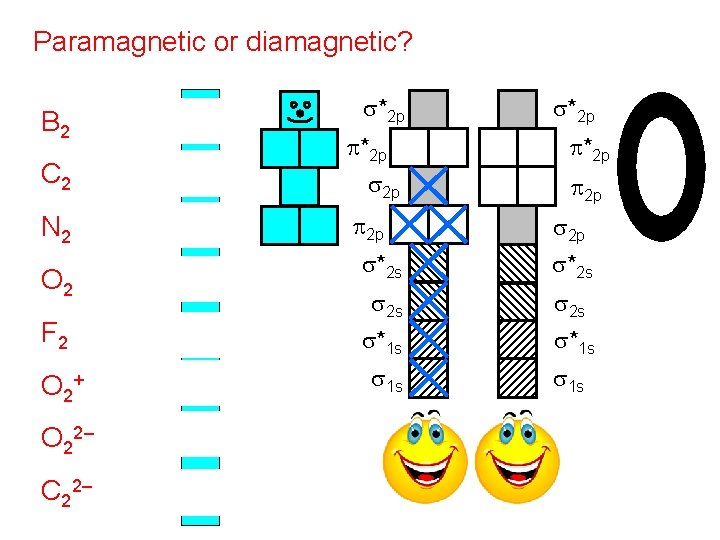
Paramagnetic or diamagnetic? B 2 (10) P C 2 (12) D N 2 (14) D s*2 p p*2 p s 2 p p 2 p s*2 p p 2 p s*2 s s 2 s O 2 (16) P F 2 (18) D s*1 s O 2+ (15) P s 1 s O 22– (18) D C 22– (14) D
- Slides: 5