Molecular Orbital Theory n Molecule n n Simplest

Molecular Orbital Theory n Molecule: n n Simplest: overlap of unchanged atomic orbitals (atoms stuck together) Hybridization: overlap of hybridized atomic orbitals localized between nuclei MO Theory: collection of nuclei with delocalized molecular orbitals Linear combination of atomic orbitals in the molecule to form molecular orbitals. n n n Atomic orbitals must overlap to form molecular orbitals. Wave functions can constructively and destructively interfere. Conservation of the number of orbitals – the number of molecular orbitals equals the total number of molecular orbitals. 1
![Overlap of 1 s wavefunctions n [ΨA – ΨB]2 n [ΨA + ΨB]2 n Overlap of 1 s wavefunctions n [ΨA – ΨB]2 n [ΨA + ΨB]2 n](http://slidetodoc.com/presentation_image/57c077fe6630889cb806a29a20820438/image-2.jpg)
Overlap of 1 s wavefunctions n [ΨA – ΨB]2 n [ΨA + ΨB]2 n [ΨA 2 + ΨB 2] 2
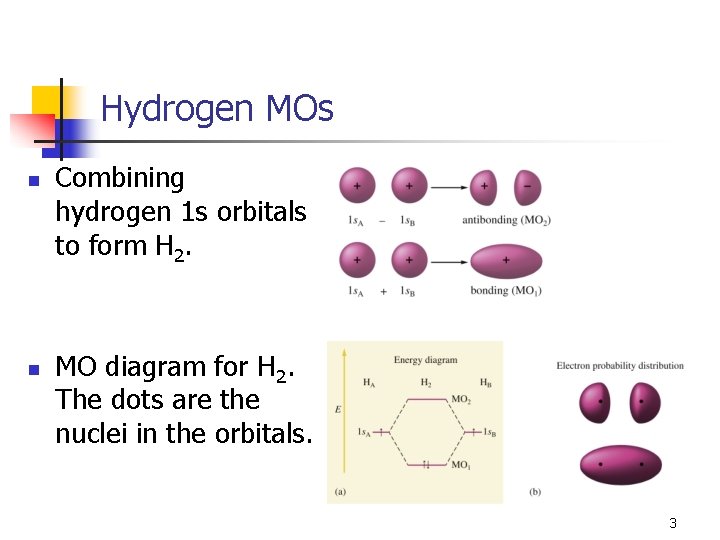
Hydrogen MOs n n Combining hydrogen 1 s orbitals to form H 2. MO diagram for H 2. The dots are the nuclei in the orbitals. 3
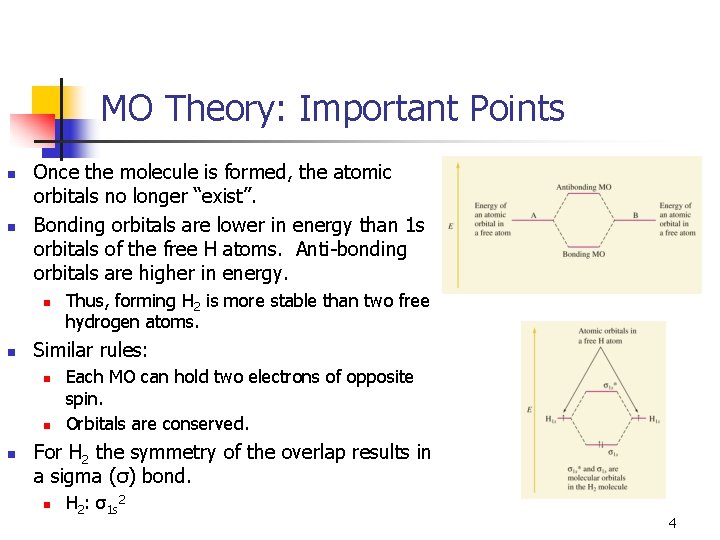
MO Theory: Important Points n n Once the molecule is formed, the atomic orbitals no longer “exist”. Bonding orbitals are lower in energy than 1 s orbitals of the free H atoms. Anti-bonding orbitals are higher in energy. n n Similar rules: n n n Thus, forming H 2 is more stable than two free hydrogen atoms. Each MO can hold two electrons of opposite spin. Orbitals are conserved. For H 2 the symmetry of the overlap results in a sigma (σ) bond. n H 2: σ1 s 2 4
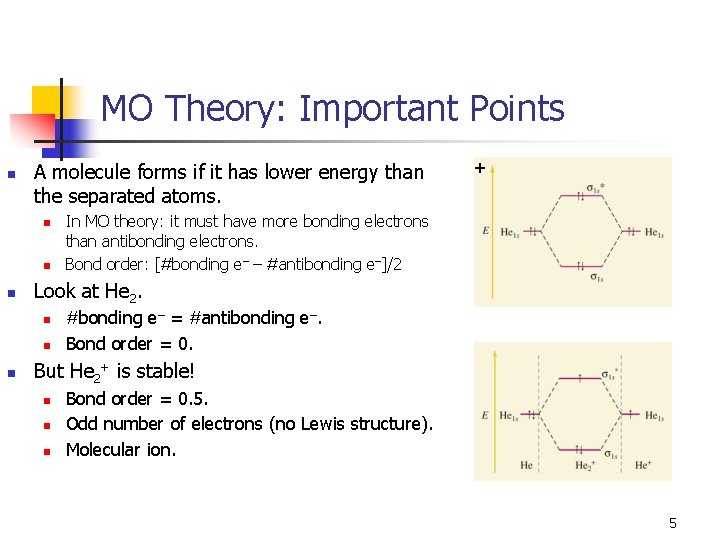
MO Theory: Important Points n A molecule forms if it has lower energy than the separated atoms. n n n In MO theory: it must have more bonding electrons than antibonding electrons. Bond order: [#bonding e– – #antibonding e–]/2 Look at He 2. n n n + #bonding e– = #antibonding e–. Bond order = 0. But He 2+ is stable! n n n Bond order = 0. 5. Odd number of electrons (no Lewis structure). Molecular ion. 5
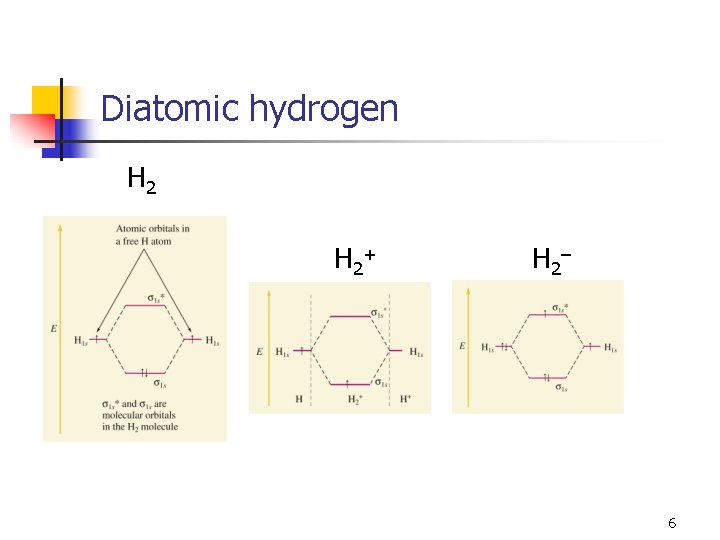
Diatomic hydrogen H 2 + H 2 – 6
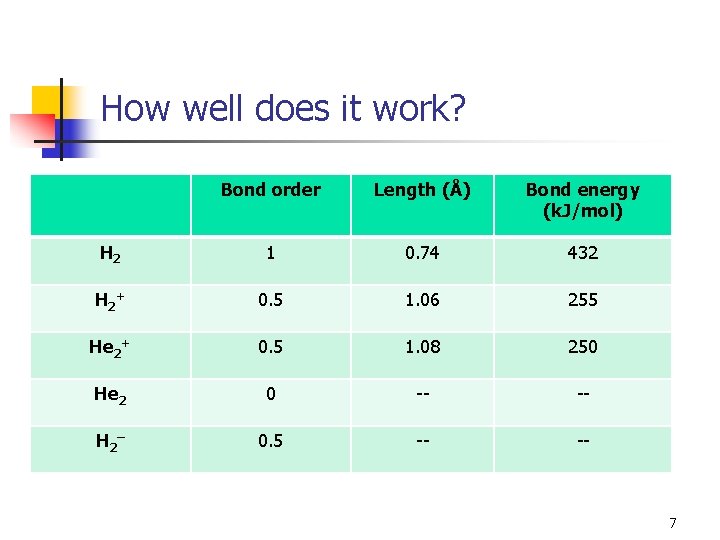
How well does it work? Bond order Length (Å) Bond energy (k. J/mol) H 2 1 0. 74 432 H 2 + 0. 5 1. 06 255 He 2+ 0. 5 1. 08 250 He 2 0 -- -- H 2 – 0. 5 -- -- 7

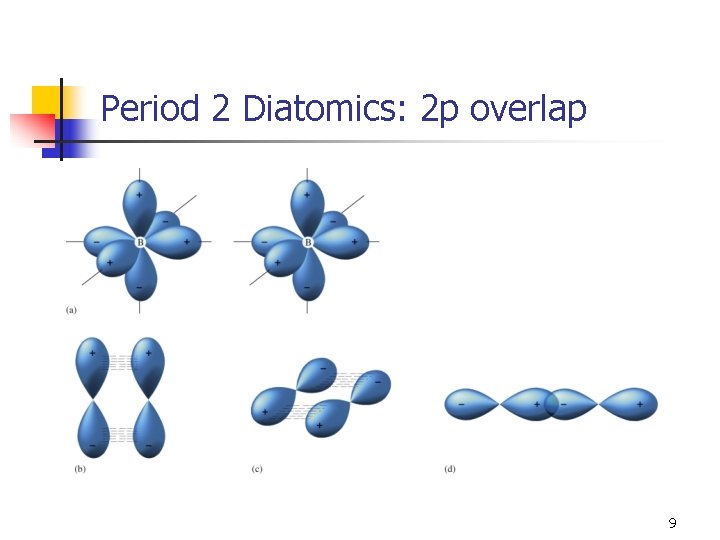
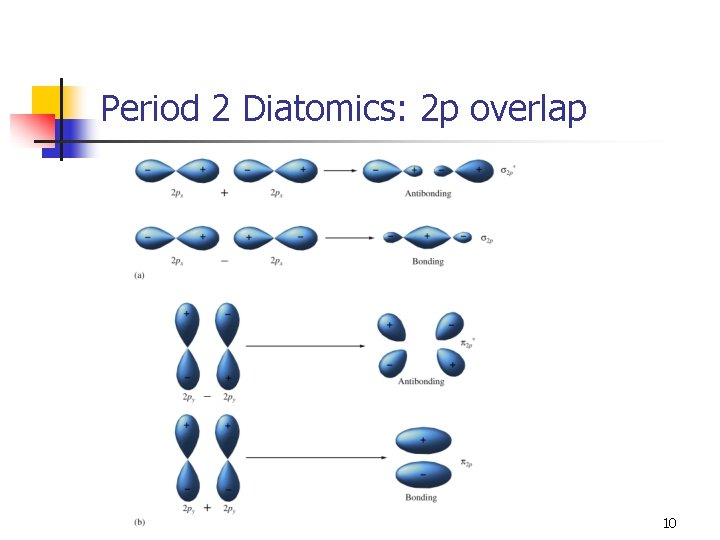
Period 2 Diatomics n n The 1 s orbitals are not involved in forming MOs (there is no overlap). The MO diagram for Li 2 predicts a stable molecule. n n Bond order = 1 Bond energy = 110 k. J/mol Bond length = 2. 67 Å Not the most stable form of lithium. 8
Period 2 Diatomics: 2 p overlap 9
Period 2 Diatomics: 2 p overlap 10

“Simple” MOs for 2 p overlap n n Because σp overlap has increased electron density between nuclei, it would be expected to be lower in energy than pi overlap. There is some p-s mixing that occurs which complicates matters. n The s orbital can mix with the p orbital with σ symmetry. 11
- Slides: 11