Molecular Orbital Diagrams Ru 2 Multiplicity 2 S1
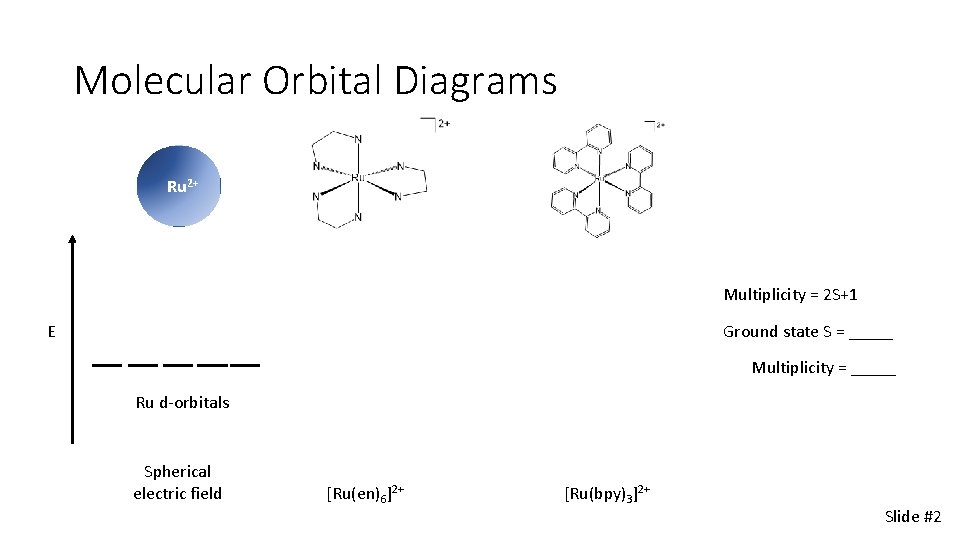
Molecular Orbital Diagrams Ru 2+ Multiplicity = 2 S+1 E Ground state S = _____ Multiplicity = _____ Ru d-orbitals Spherical electric field [Ru(en)6]2+ [Ru(bpy)3]2+ Slide #2
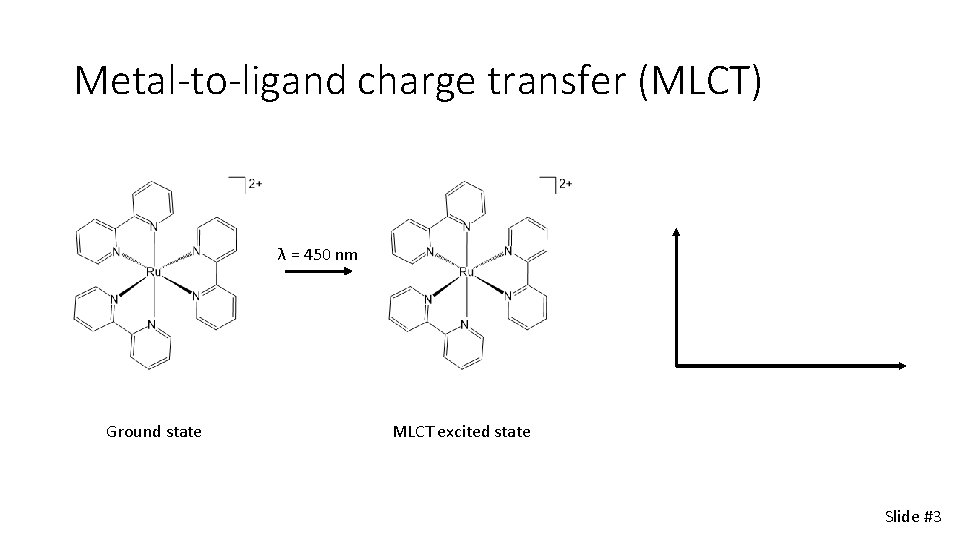
Metal-to-ligand charge transfer (MLCT) λ = 450 nm Ground state MLCT excited state Slide #3
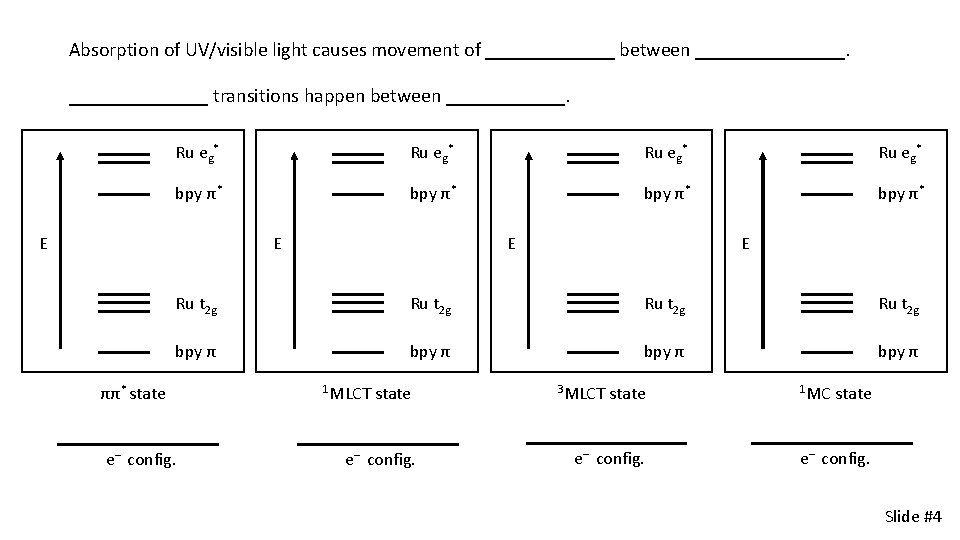
Absorption of UV/visible light causes movement of _______ between ________ transitions happen between ______. Ru eg* bpy π* E E Ru t 2 g bpy π ππ* state e– config. 1 MLCT state e– config. 3 MLCT state e– config. 1 MC state e– config. Slide #4
![[Ru(bpy)3]2+ excited state processes 1. 2. a. b. E 3. a. b. Nuclear coordinate [Ru(bpy)3]2+ excited state processes 1. 2. a. b. E 3. a. b. Nuclear coordinate](http://slidetodoc.com/presentation_image_h/b4ac03d47d201d42de32b73db7b3414c/image-4.jpg)
[Ru(bpy)3]2+ excited state processes 1. 2. a. b. E 3. a. b. Nuclear coordinate Slide #5
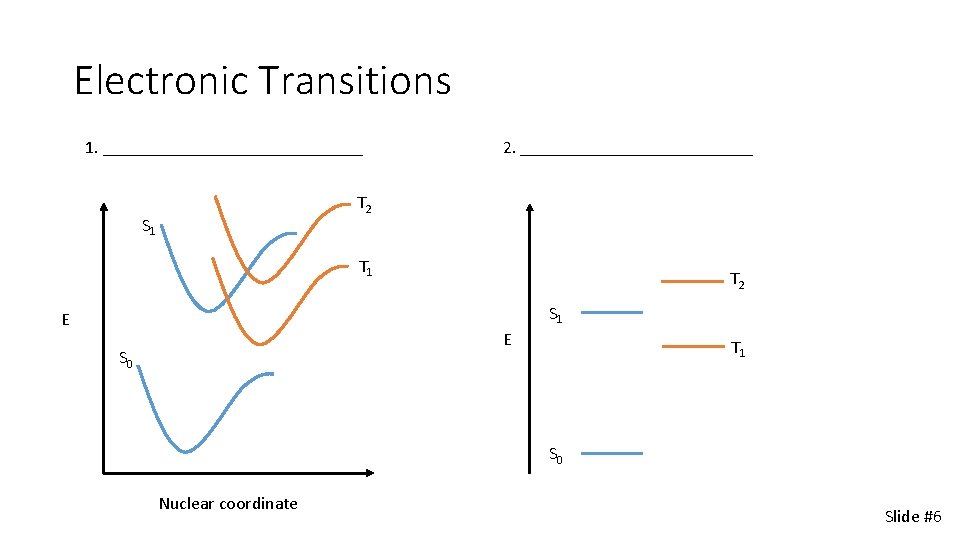
Electronic Transitions 1. _______________ 2. _____________ T 2 S 1 E E S 0 T 1 S 0 Nuclear coordinate Slide #6
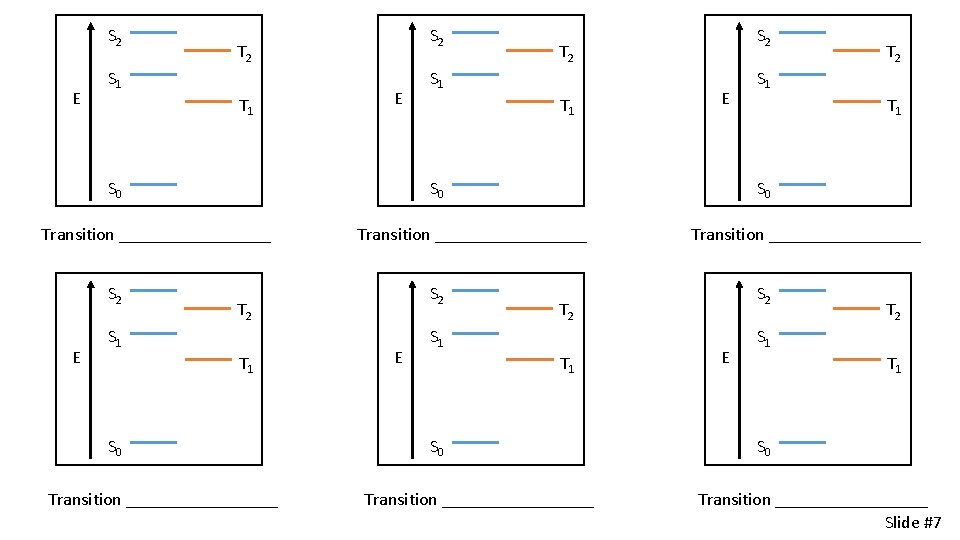
S 2 E S 2 T 2 S 1 T 1 E Transition _________ E T 2 S 1 T 1 E Transition _________ S 2 T 2 S 1 T 1 S 0 Transition _________ E T 2 S 1 T 1 S 0 S 0 S 2 Transition _________ S 2 T 2 S 1 T 1 S 0 Transition _________ E T 2 S 1 T 1 S 0 Transition _________ Slide #7
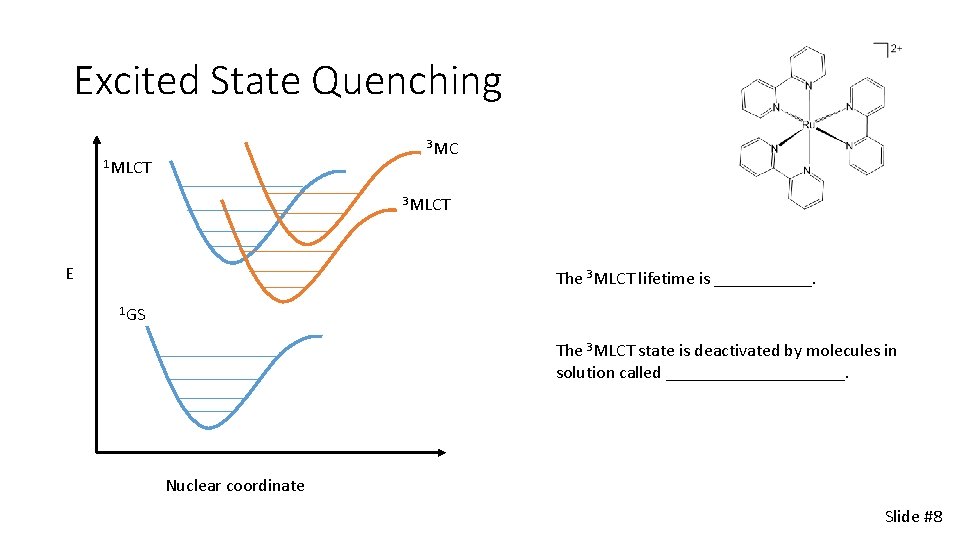
Excited State Quenching 3 MC 1 MLCT 3 MLCT E The 3 MLCT lifetime is ______. 1 GS The 3 MLCT state is deactivated by molecules in solution called __________. Nuclear coordinate Slide #8
- Slides: 7