Molecular Geometry http www scl ameslab govMac Mol

Molecular Geometry http: //www. scl. ameslab. gov/Mac. Mol. Plt/Surface. JPG

VSEPR - Valence Shell Electron Pair Repulsion Theory ØEach group of valence electrons around a central atom is located as far away as possible from the others in order to minimize repulsions. ØThe result is five electron-group arrangements of minimum energy seen in a large majority of molecules and polyatomic ions. ØBecause valence electrons can be bonding or nonbonding, the same electron-group arrangement can give rise to different molecular shapes. A - central atom X -surrounding atom AXm. En integers Silberberg, Principles of Chemistry E -nonbonding valence electron-group
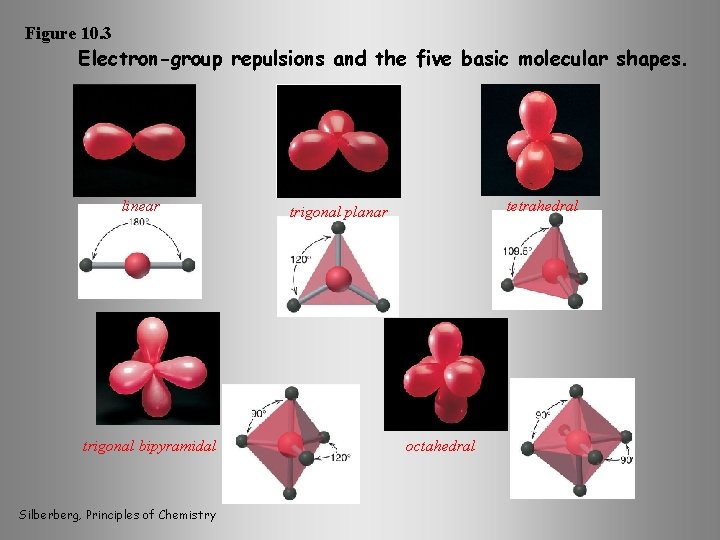
Figure 10. 3 Electron-group repulsions and the five basic molecular shapes. linear trigonal bipyramidal Silberberg, Principles of Chemistry tetrahedral trigonal planar octahedral
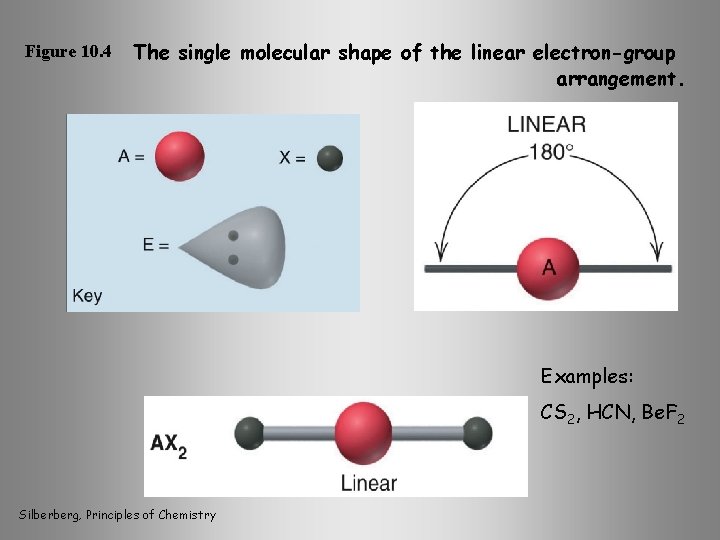
Figure 10. 4 The single molecular shape of the linear electron-group arrangement. Examples: CS 2, HCN, Be. F 2 Silberberg, Principles of Chemistry
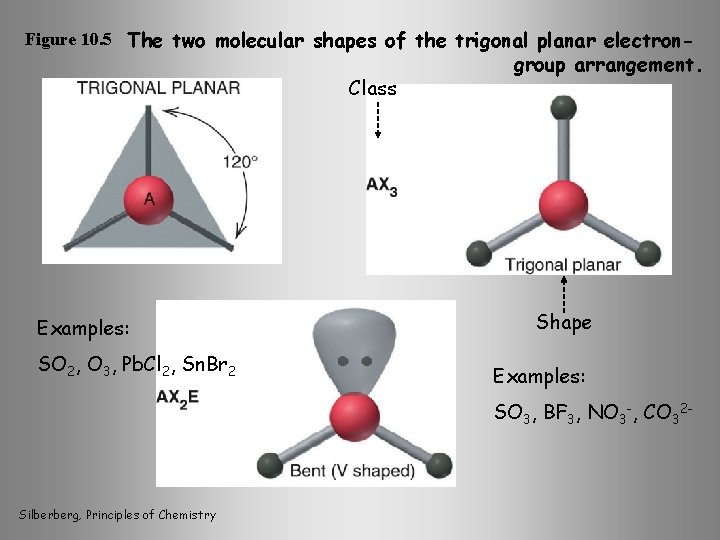
Figure 10. 5 The two molecular shapes of the trigonal planar electron- Class Examples: SO 2, O 3, Pb. Cl 2, Sn. Br 2 group arrangement. Shape Examples: SO 3, BF 3, NO 3 -, CO 32 - Silberberg, Principles of Chemistry
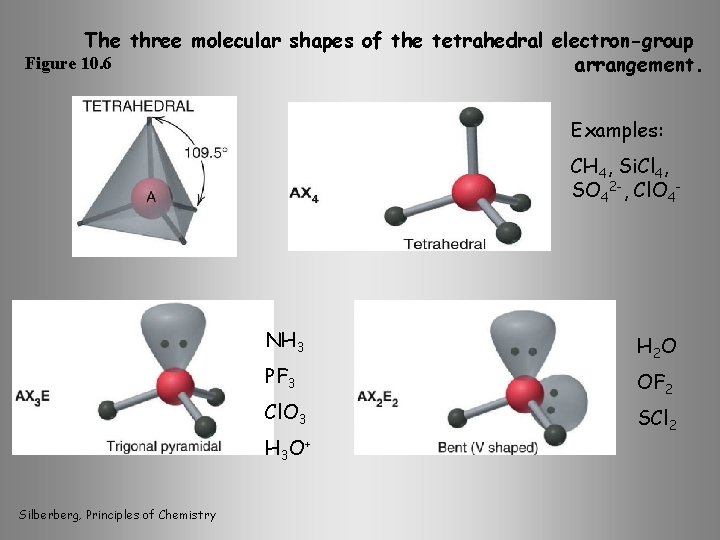
The three molecular shapes of the tetrahedral electron-group Figure 10. 6 arrangement. Examples: CH 4, Si. Cl 4, SO 42 -, Cl. O 4 - NH 3 PF 3 Cl. O 3 H 3 O+ Silberberg, Principles of Chemistry H 2 O OF 2 SCl 2
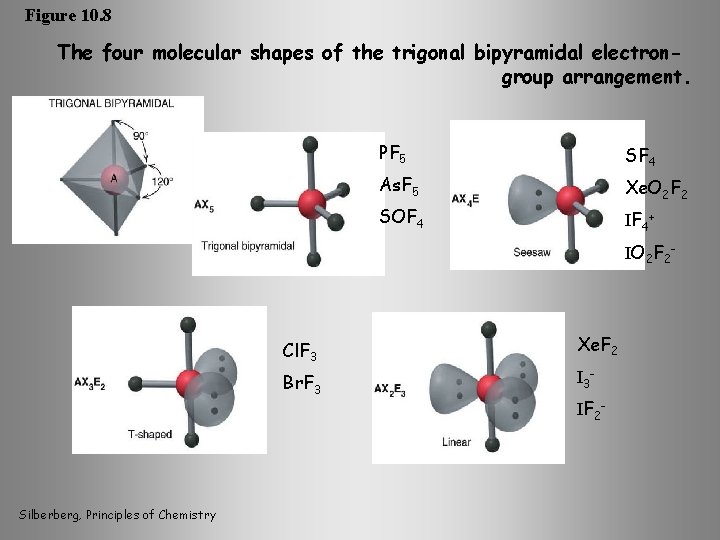
Figure 10. 8 The four molecular shapes of the trigonal bipyramidal electrongroup arrangement. PF 5 SF 4 As. F 5 Xe. O 2 F 2 SOF 4 IF 4+ IO 2 F 2 - Silberberg, Principles of Chemistry Cl. F 3 Xe. F 2 Br. F 3 IF 2 -
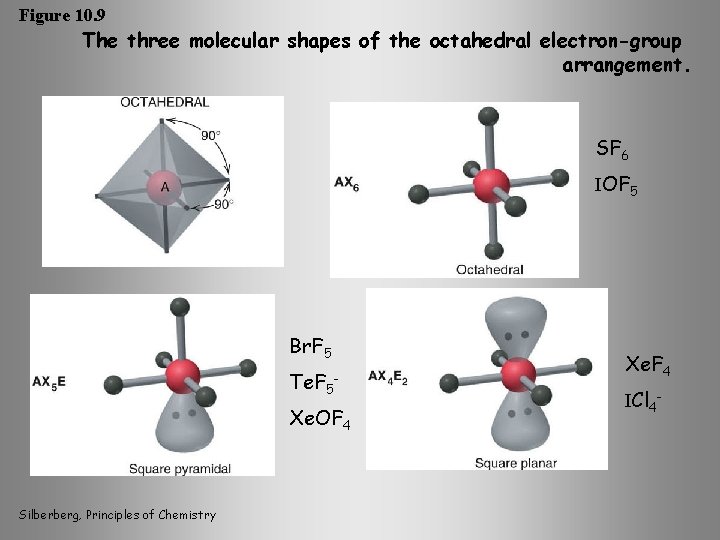
Figure 10. 9 The three molecular shapes of the octahedral electron-group arrangement. SF 6 IOF 5 Br. F 5 Te. F 5 Xe. OF 4 Silberberg, Principles of Chemistry Xe. F 4 ICl 4 -
- Slides: 8