Molecular Geometry Bonding Theories l Molecules have shapes

Molecular Geometry & Bonding Theories

l Molecules have shapes and sizes that are defined by the __________ and __________ between the nuclei of atoms. l Shape and size determine the properties of that substance.

l Molecular Shapes l Lewis shape just gives __________. l The Lewis structure helps us to begin to see the __________ shape l Bond angles determine the molecular shape along with bond lengths. l 3 -D arrangement

Lewis Symbols l Lewis structures show the __________. l Maximum l of __________ electrons Deals with the valence electrons l Highest energy level, __________ sublevels

Lewis Structure l Sulfur has 6 valence electrons l Dots around the 4 sides of an atom represent the valence electrons. l 2 electrons on a side – giving a possible 8 l __________– atoms tend to gain or lose electrons, or share, to achieve 8 valence electrons (an octet)
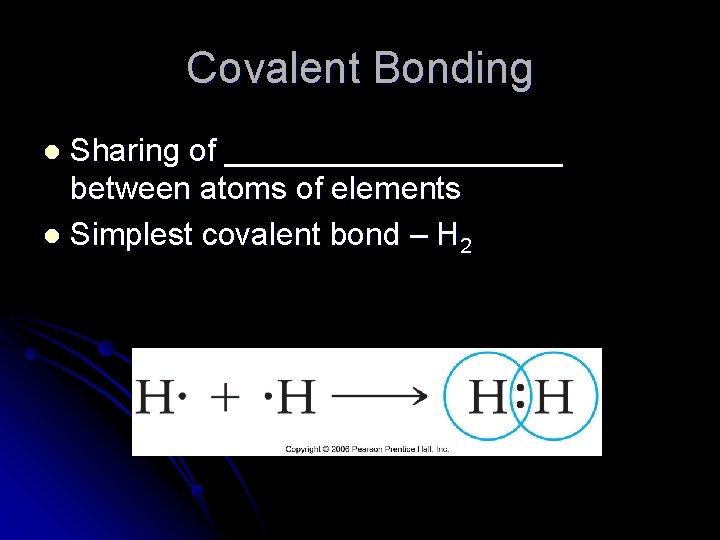
Covalent Bonding Sharing of __________ between atoms of elements l Simplest covalent bond – H 2 l

Lewis Structures l A dash, “—” represents a shared pair of __________

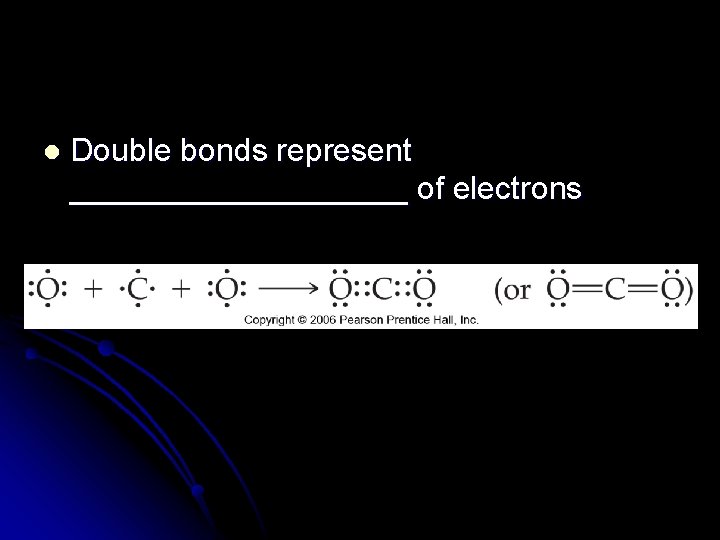
l Double bonds represent __________ of electrons

l Triple bonds represent __________ of electrons.

Drawing Lewis Structures of Compounds l 1. Sum the valence electrons from all atoms PCl 3 = 5 x 1 = 5 l Cl = 7 x 3 = 21 26 valence electrons l. P

l 2. Write the symbols for the atoms to show which atoms are attached to which and attach them (using a dash) l Central atom is generally the less electronegative atom

3. Complete the octets around the atoms bonded to the central atom. l Keep track of your electrons = 24 el

l 4. Place any leftover electrons on the central atom.
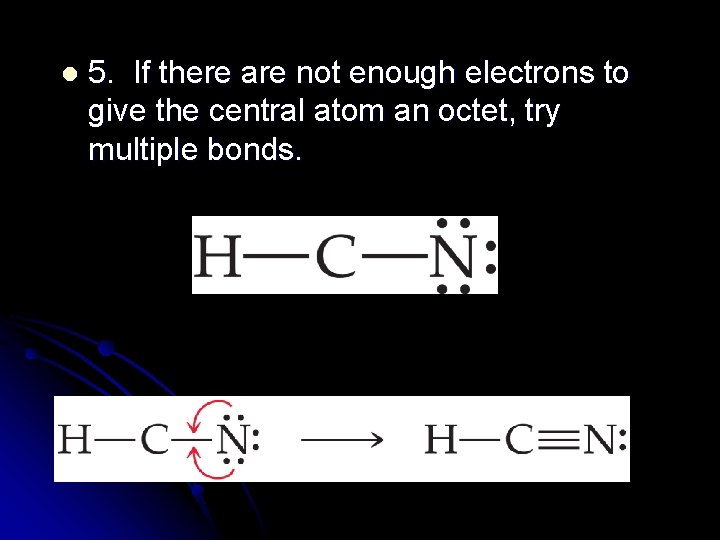
l 5. If there are not enough electrons to give the central atom an octet, try multiple bonds.
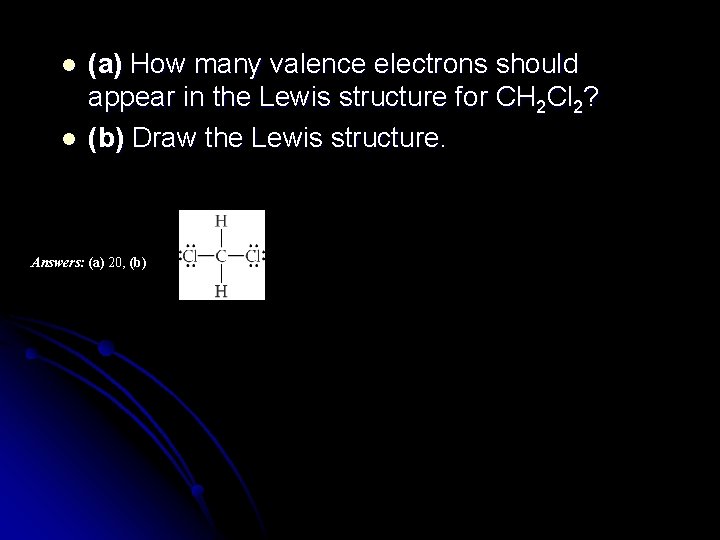
l l (a) How many valence electrons should appear in the Lewis structure for CH 2 Cl 2? (b) Draw the Lewis structure. Answers: (a) 20, (b)
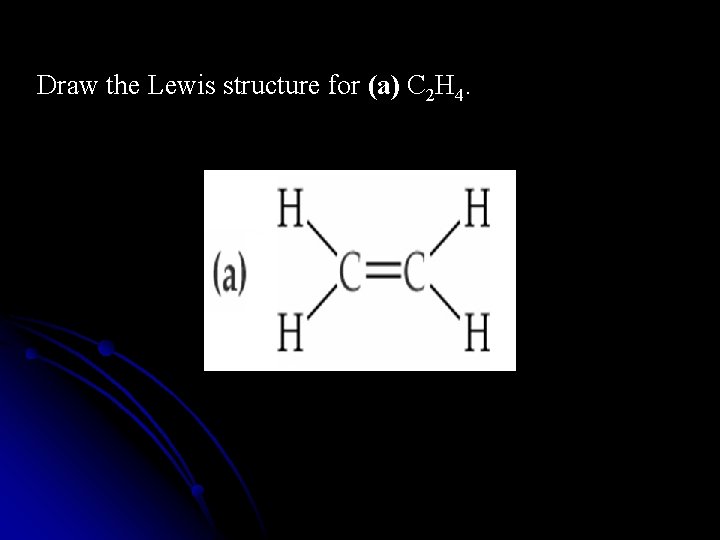
Draw the Lewis structure for (a) C 2 H 4.

VSEPR Model l Valence Shell Electron Pair Repulsion model l Is based on the number of regions of high electron density around a central atom. Can be used to predict structures of __________ or ions that contain only non-metals by minimizing the electrostatic __________ between the regions of high electron density. can also be used to predict structures of molecules or ions that contain multiple bonds or unpaired electrons.

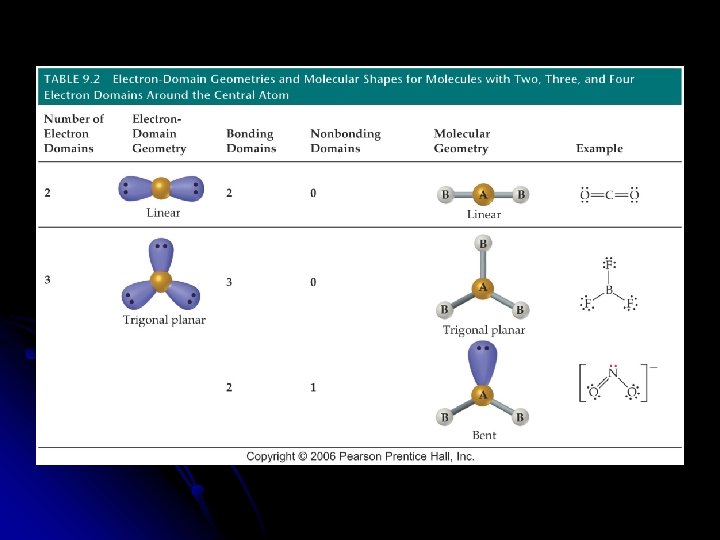
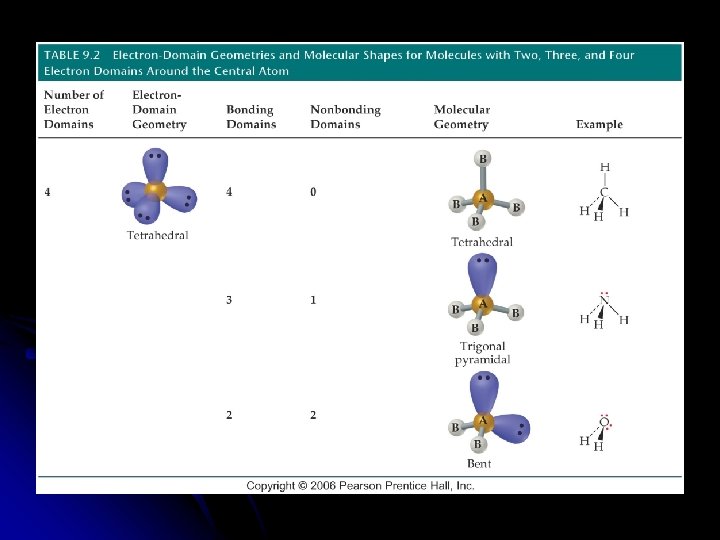
Predicting the VSEPR Model Sketch the Lewis Structure l Determine the molecular arrangement. l Calculate the electron __________ around the central atom. l l Electron domain is the __________ domain around the central atom l Double bonds & triple bonds count as 1 electron domain.

Bonding Pair l Electrons that are __________ Nonbonding Pair l Lone pairs of __________

Nonbonding domain . . : O. . ─O =O. . 2 bonding domains 1 nonbonding domain

l Molecular geometry is the arrangement of __________ in space. l The best arrangement of a given number of electron domains is the one that will __________ the repulsion among them.
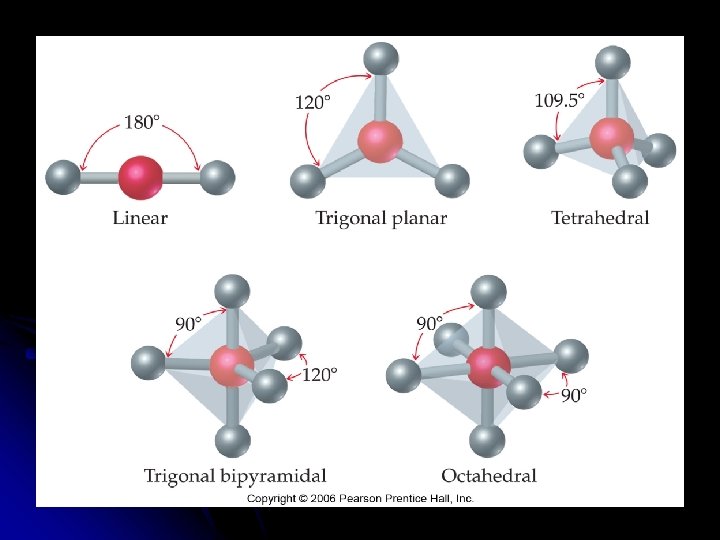
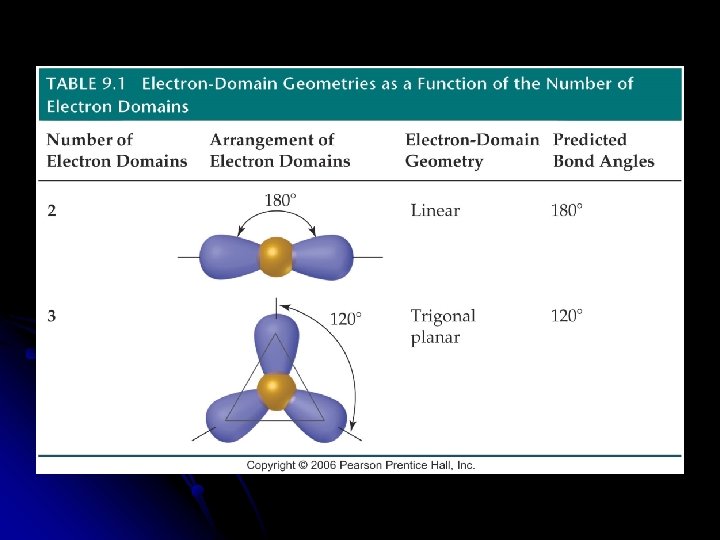

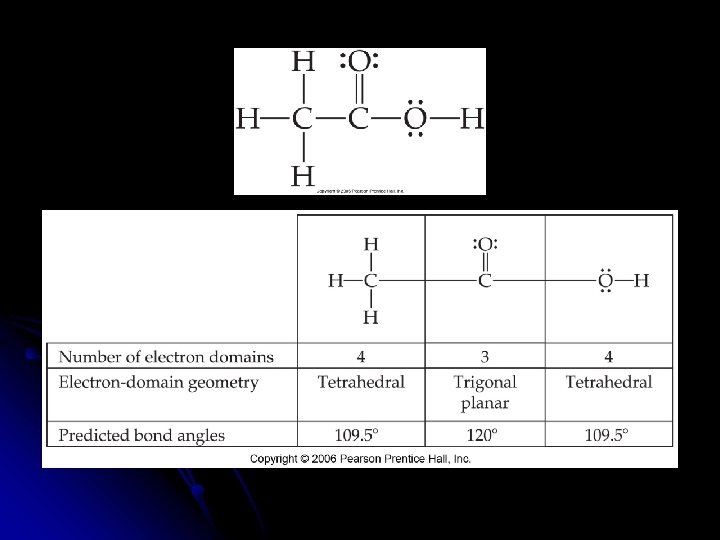
Molecular Geometry of only the atoms of the molecule or ion l Need to know the number of __________ and __________ pairs of electrons l __________ l Draw the Lewis structure to determine the bonding and nonbonding pairs of electrons
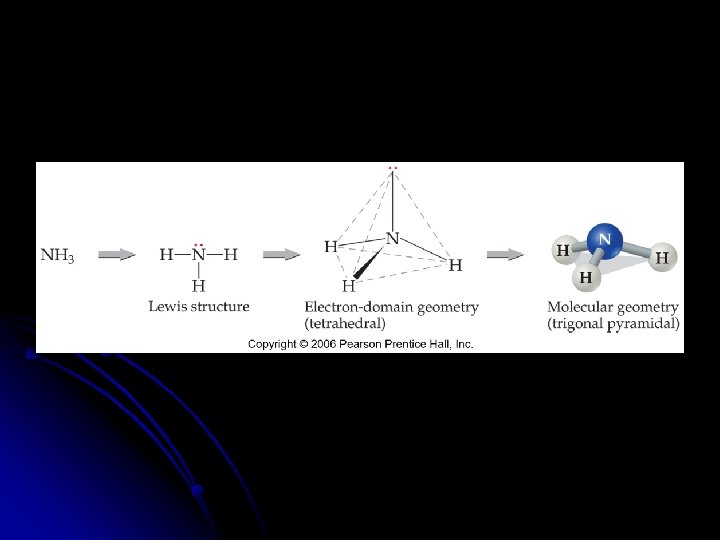

Effect of Nonbonding Electrons on Bond Angles l As the number of __________ electrons increases, the bond angle decreases. l Nonbonding pairs of electrons experience less nuclear attraction because the domain spreads out which compresses the angles l __________ represent larger domains l Because of the higher electronic charge, it acts almost like a nonbonding pair of electrons
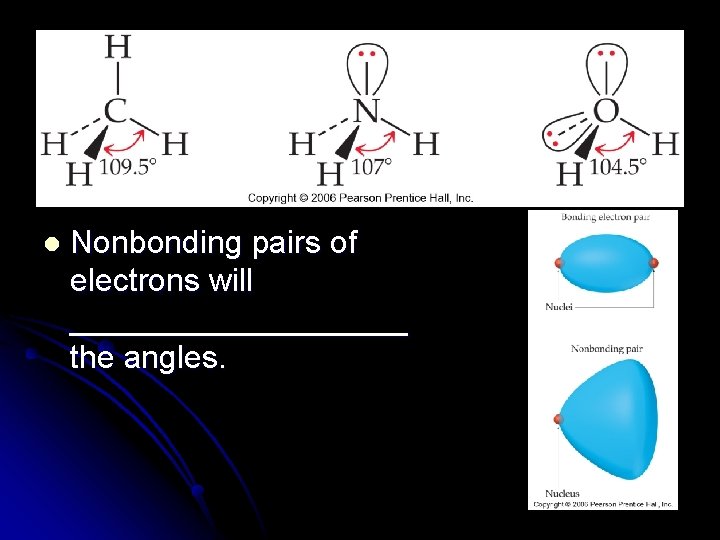
l Nonbonding pairs of electrons will __________ the angles.
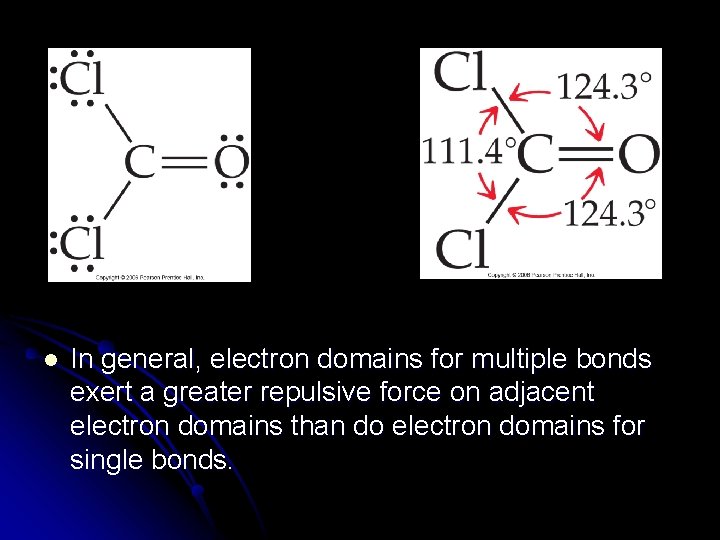
l In general, electron domains for multiple bonds exert a greater repulsive force on adjacent electron domains than do electron domains for single bonds.

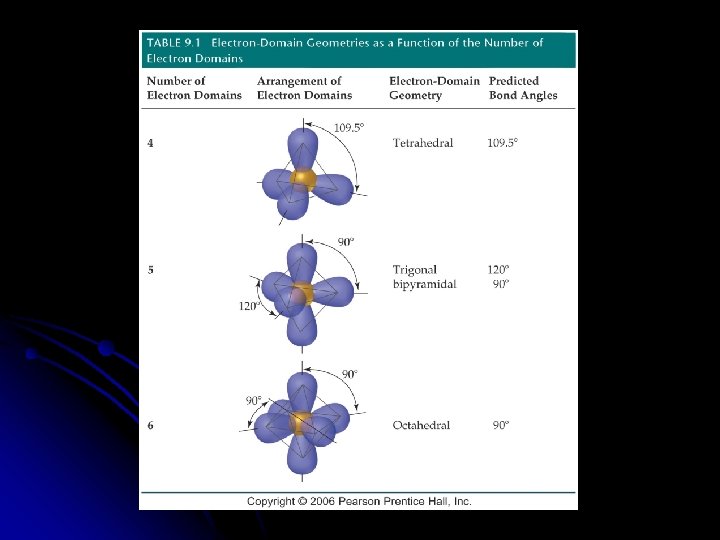
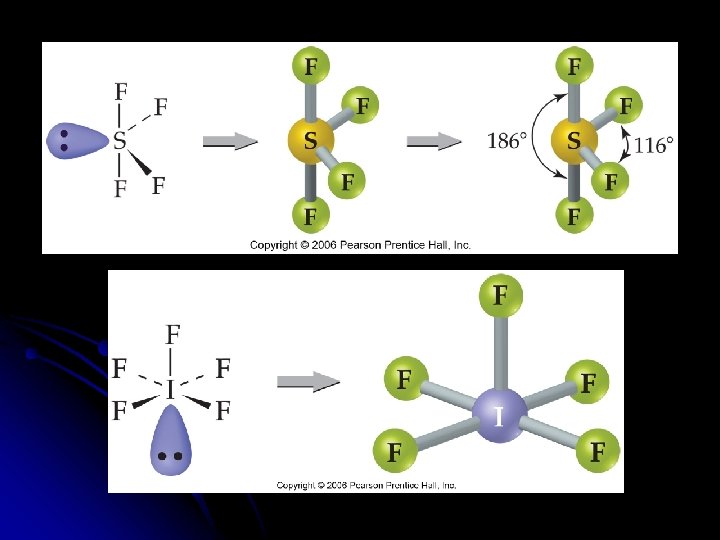
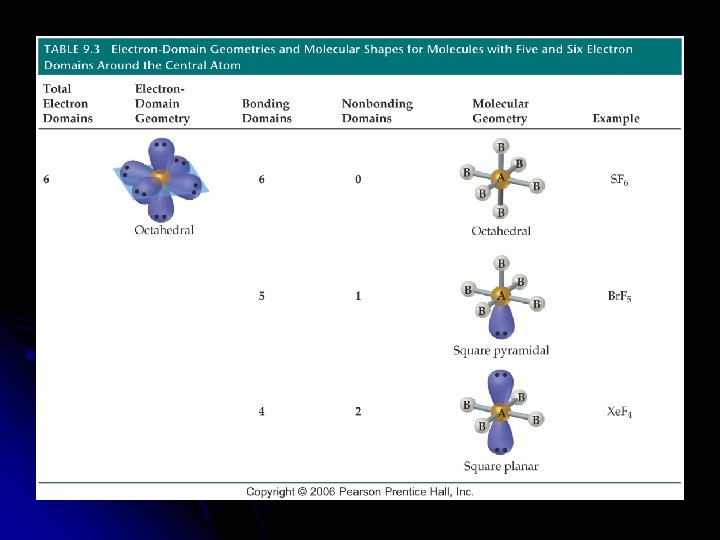
Molecules with Expanded Valence Shells l Molecules with five or six electron domains around the central atom l Trigonal bipyramidal l Octahedral

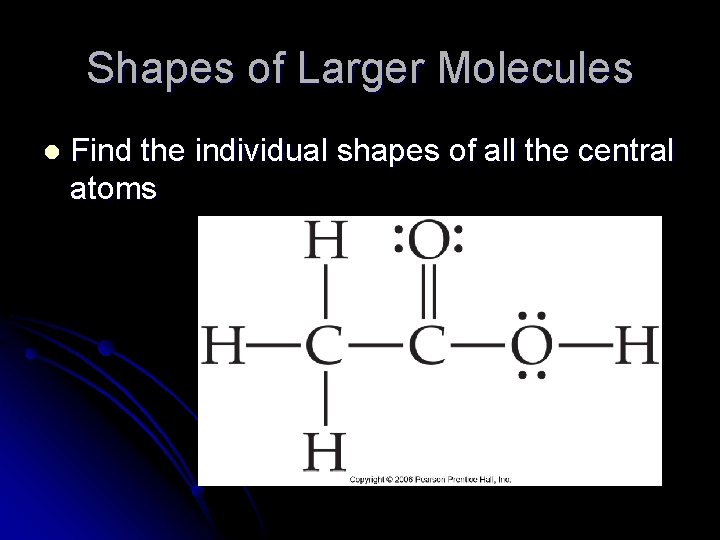
Shapes of Larger Molecules l Find the individual shapes of all the central atoms
- Slides: 38