Molecular Geometry bond length angle determined experimentally Lewis
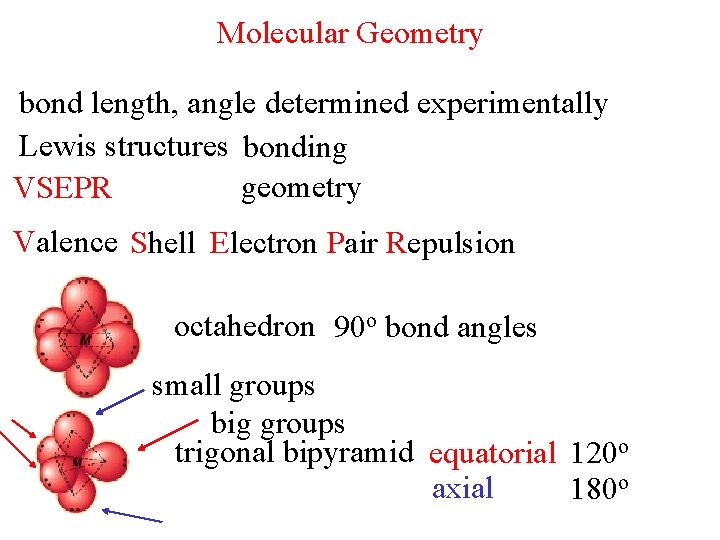
Molecular Geometry bond length, angle determined experimentally Lewis structures bonding geometry VSEPR Valence Shell Electron Pair Repulsion octahedron 90 o bond angles small groups big groups trigonal bipyramid equatorial 120 o axial 180 o

tetrahedron 109. 5 o trigonal planar 120 o linear 180 o geometry apply to Chemistry
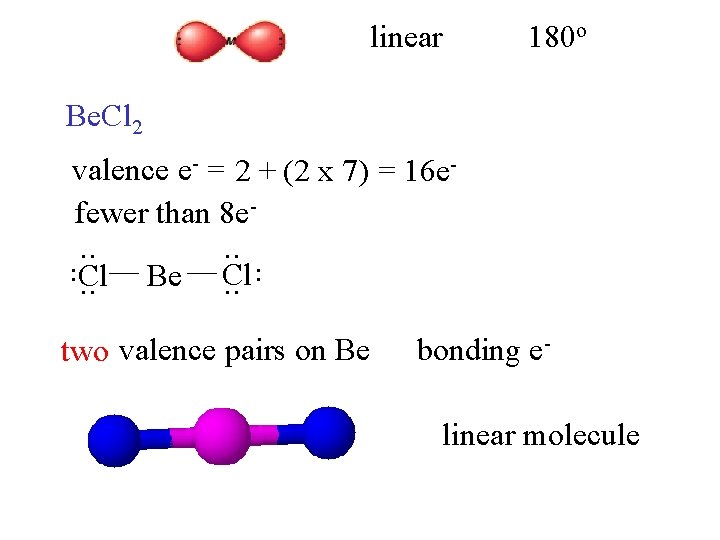
linear 180 o Be. Cl 2 valence e- = 2 + (2 x 7) = 16 efewer than 8 e. Be . . Cl. . two valence pairs on Be bonding elinear molecule
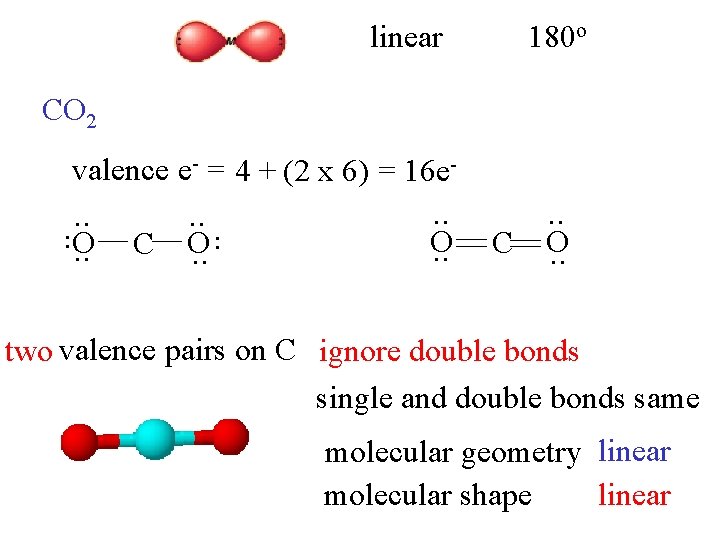
linear 180 o CO 2 valence e- = 4 + (2 x 6) = 16 e- . . C O. . two valence pairs on C ignore double bonds single and double bonds same molecular geometry linear molecular shape linear
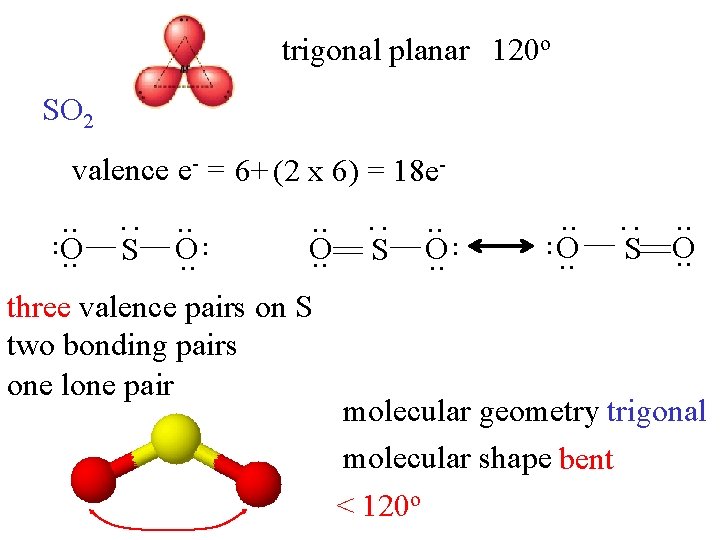
trigonal planar 120 o SO 2 three valence pairs on S two bonding pairs one lone pair . . O. . S O. . : S . . O. . : . . O. . S . . O. . : valence e- = 6+ (2 x 6) = 18 e- molecular geometry trigonal molecular shape bent < 120 o

tetrahedral 109. 5 o CH 4 valence e- = 4+(4 x 1) = 8 efour valence pairs on C 109. 5 o H H C H H molecular geometry tetrahedral molecular shape tetrahedral
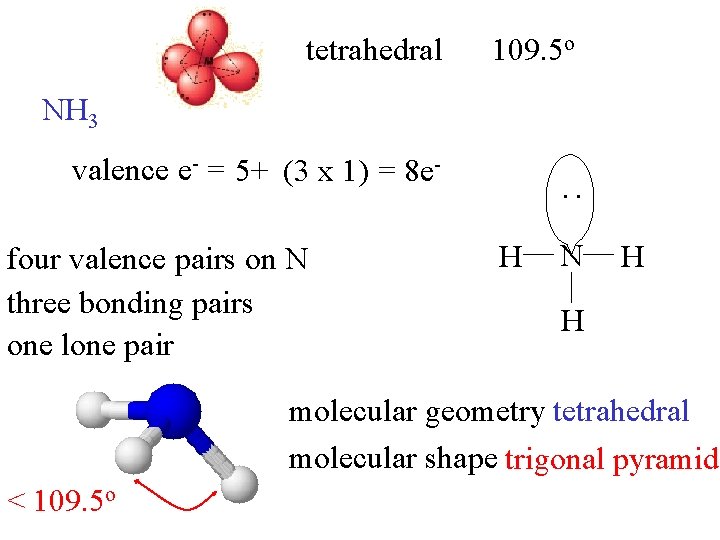
tetrahedral 109. 5 o NH 3 valence e- = 5+ (3 x 1) = 8 e- : four valence pairs on N three bonding pairs one lone pair H N H H molecular geometry tetrahedral molecular shape trigonal pyramid < 109. 5 o
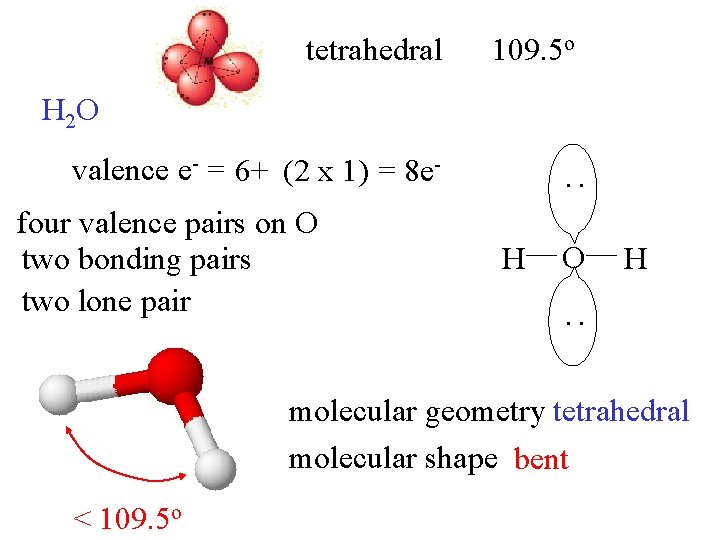
tetrahedral 109. 5 o H 2 O four valence pairs on O two bonding pairs two lone pair : valence e- = 6+ (2 x 1) = 8 e. H O H : molecular geometry tetrahedral molecular shape bent < 109. 5 o
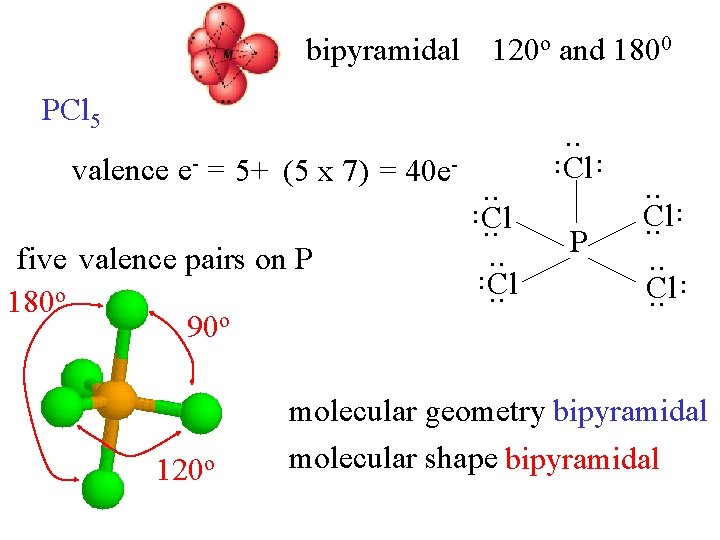
bipyramidal 120 o and 1800 PCl 5 . . 120 o . . five valence pairs on P 180 o 90 o P . . Cl. . Cl . . valence e- = 5+ (5 x 7) = 40 e- . . molecular geometry bipyramidal molecular shape bipyramidal
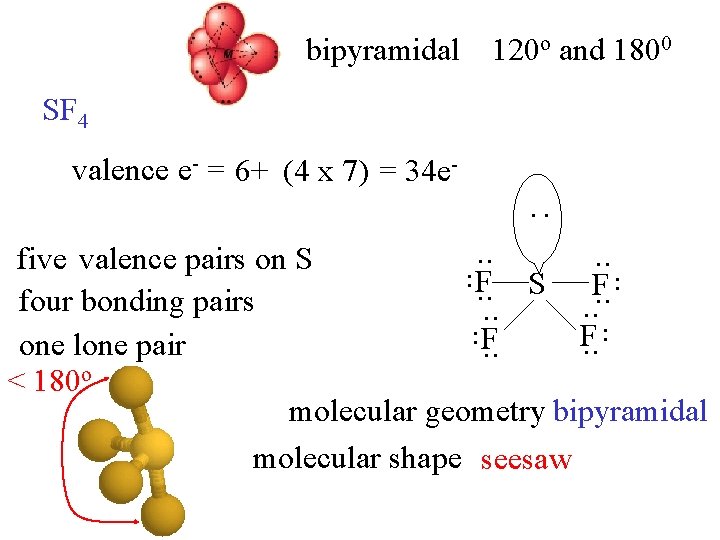
bipyramidal 120 o and 1800 SF 4 valence e- = 6+ (4 x 7) = 34 e- : . . . five valence pairs on S F. . four bonding pairs. . F F. . one lone pair. . < 180 o molecular geometry bipyramidal molecular shape seesaw
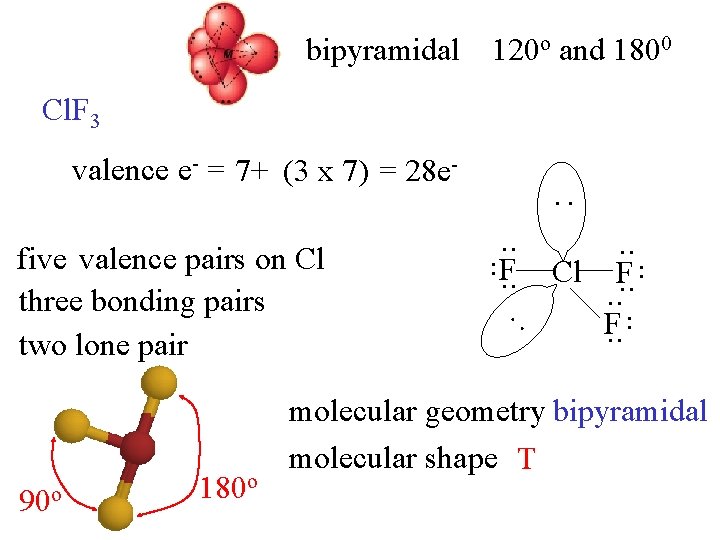
bipyramidal 120 o and 1800 Cl. F 3 valence e- = 7+ (3 x 7) = 28 e- : . . 180 o : 90 o . . five valence pairs on Cl three bonding pairs two lone pair . . F. . Cl. . F. . molecular geometry bipyramidal molecular shape T
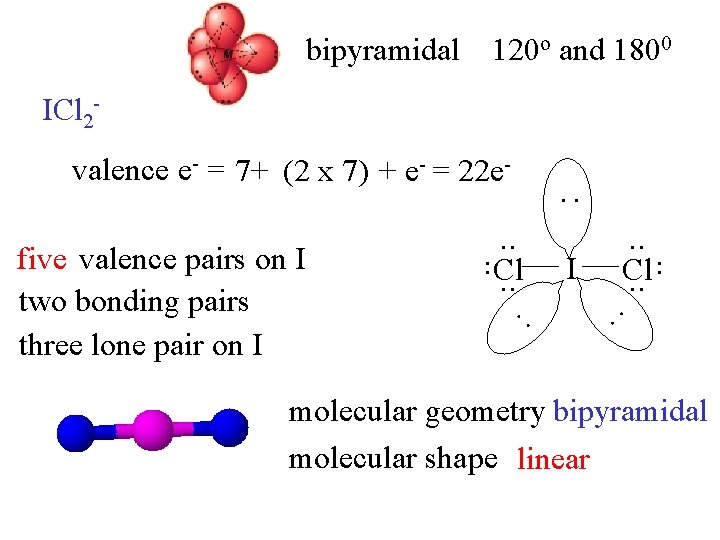
bipyramidal 120 o and 1800 ICl 2 valence e- = 7+ (2 x 7) + e- = 22 e- : : : I . . Cl. . . five valence pairs on I two bonding pairs three lone pair on I . . Cl. . molecular geometry bipyramidal molecular shape linear
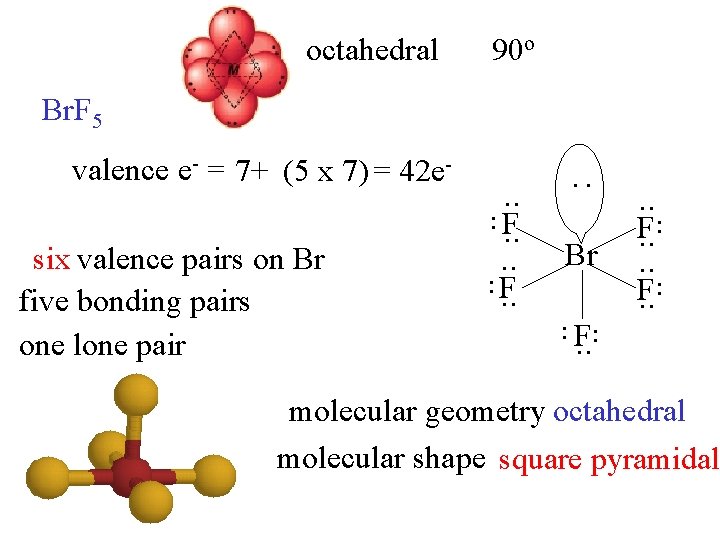
octahedral 90 o Br. F 5 Br . . . F. . six valence pairs on Br five bonding pairs one lone pair . . F. . : valence e- = 7+ (5 x 7) = 42 e- molecular geometry octahedral molecular shape square pyramidal
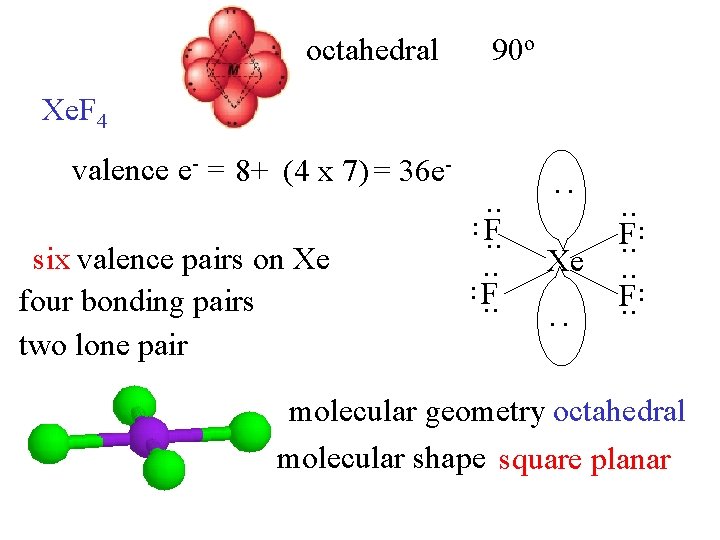
octahedral 90 o Xe. F 4 : . . six valence pairs on Xe four bonding pairs two lone pair . . F. . Xe. . F. . : valence e- = 8+ (4 x 7) = 36 e- molecular geometry octahedral molecular shape square planar
- Slides: 14