Molecular Geometry and Bonding Theories The properties of
Molecular Geometry and Bonding Theories

The properties of a molecule depend on its shape and the nature of its bonds. In this unit, we will discuss three models. (1) a model for the geometry of molecules -- valence-shell electron-pair repulsion (VSEPR) theory (2) a model about WHY molecules form bonds and WHY they have the shape they do -- valence-bond theory (3) a model of chemical bonding that deals with the electronic structure of molecules -- molecular orbital (MO) theory
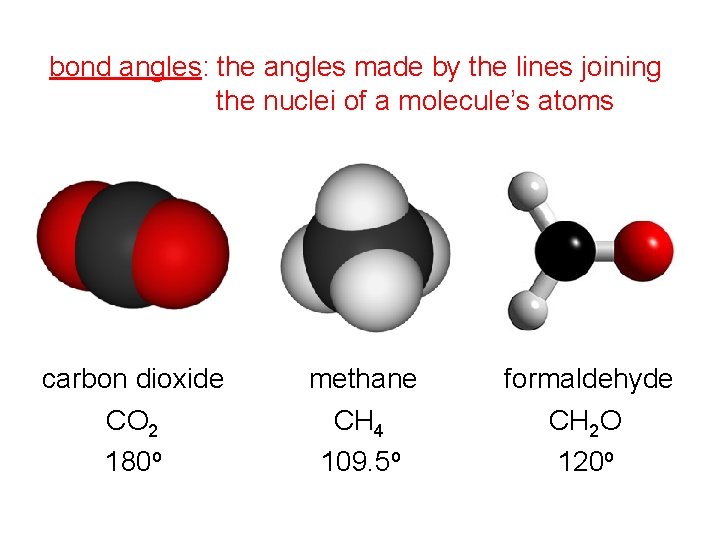
bond angles: the angles made by the lines joining the nuclei of a molecule’s atoms carbon dioxide CO 2 180 o methane CH 4 109. 5 o formaldehyde CH 2 O 120 o
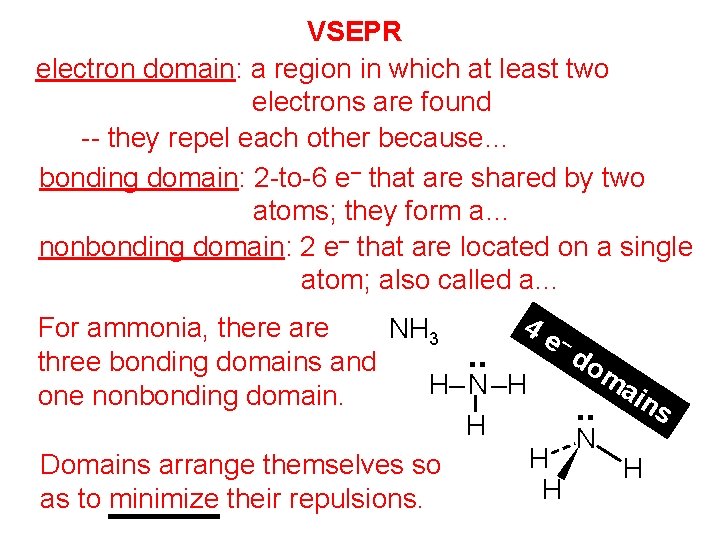
VSEPR electron domain: a region in which at least two electrons are found -- they repel each other because… they are all (–) bonding domain: 2 -to-6 e– that are shared by two atoms; they form a… covalent bond nonbonding domain: 2 e– that are located on a single atom; also called a… lone pair 4 e For ammonia, there are NH 3 – do. . three bonding domains and ma H– N –H ins one nonbonding domain. . . H N H Domains arrange themselves so H H as to minimize their repulsions.
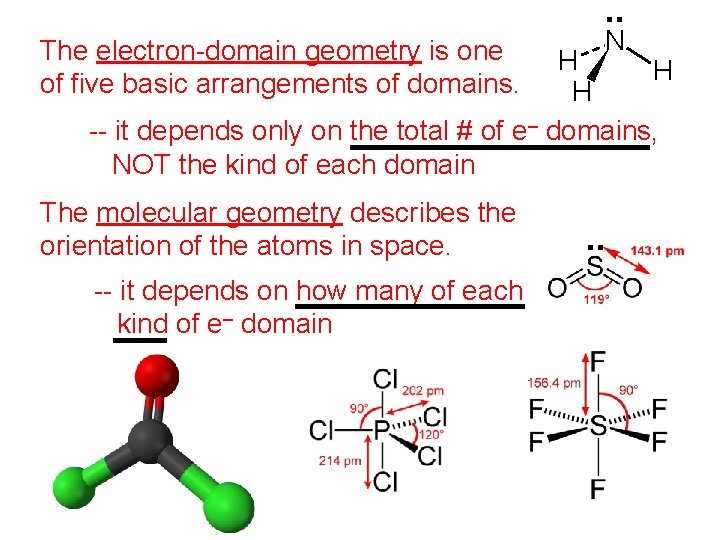
. . The electron-domain geometry is one of five basic arrangements of domains. N H H H -- it depends only on the total # of e– domains, NOT the kind of each domain The molecular geometry describes the orientation of the atoms in space. -- it depends on how many of each kind of e– domain . .
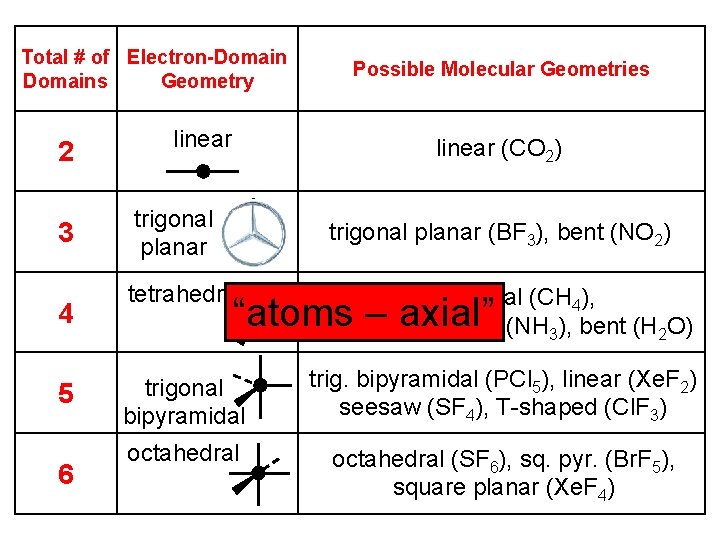
Total # of Electron-Domains Geometry 2 3 4 5 6 linear Possible Molecular Geometries linear (CO 2) trigonal planar (BF 3), bent (NO 2) tetrahedral (CH 4), trigonal pyramidal (NH 3), bent (H 2 O) trigonal bipyramidal trig. bipyramidal (PCl 5), linear (Xe. F 2) seesaw (SF 4), T-shaped (Cl. F 3) octahedral (SF 6), sq. pyr. (Br. F 5), square planar (Xe. F 4) “atoms – axial”
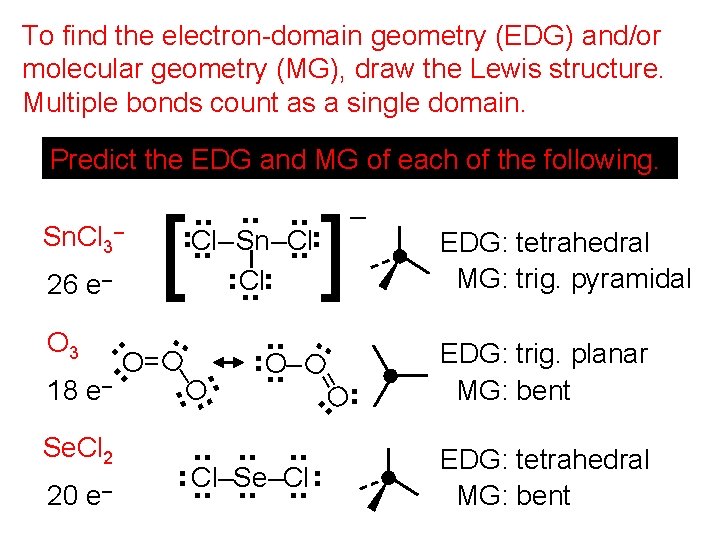
To find the electron-domain geometry (EDG) and/or molecular geometry (MG), draw the Lewis structure. Multiple bonds count as a single domain. Predict the EDG and MG of each of the following. . . . – Sn. Cl 3 Cl–. . Sn–Cl. . Cl 26 e–. . . . O 3 O O=O O–. . – 18 e. . Se. Cl 2. . . Cl–Se–Cl –. . . 20 e = O. . . ] . . . –O. . . . [ – EDG: tetrahedral MG: trig. pyramidal EDG: trig. planar MG: bent EDG: tetrahedral MG: bent
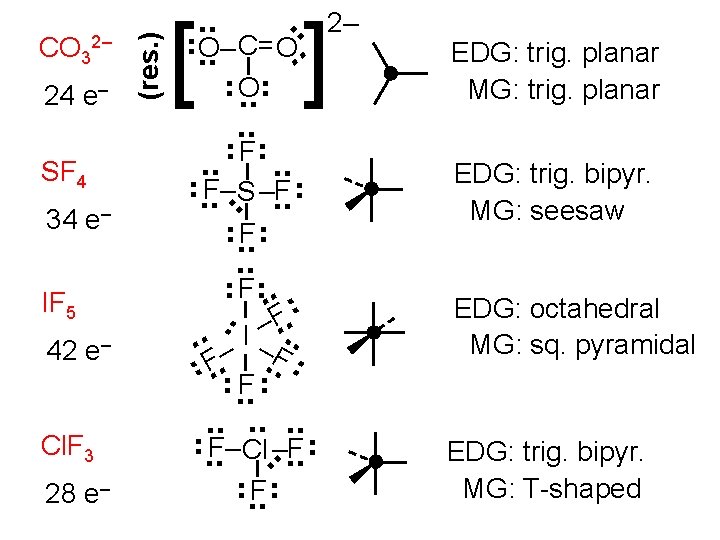
28 e– . . . EDG: trig. bipyr. MG: T-shaped . . EDG: octahedral MG: sq. pyramidal . . Cl. F 3 EDG: trig. bipyr. MG: seesaw . . 42 e– . . . IF 5 ] . . . . 34 e– [ EDG: trig. planar MG: trig. planar . . SF 4 (res. ) 24 e– 2– . . CO 32– . . O– C= O. . . F– S –F. . F. . – I –F. . . . F– Cl –F. . F
- Slides: 8