Molecular Geometry and Bonding Theories AP Chemistry Ch

Molecular Geometry and Bonding Theories AP Chemistry – Ch 9 Mr. Christopherson

Bonding Theories & Geometry • • Molecular Geometry (shapes) VSEPR Theory Lewis Structures Molecular Polarity (dipoles) Covalent Bonds Hybridization Ionic Bonds
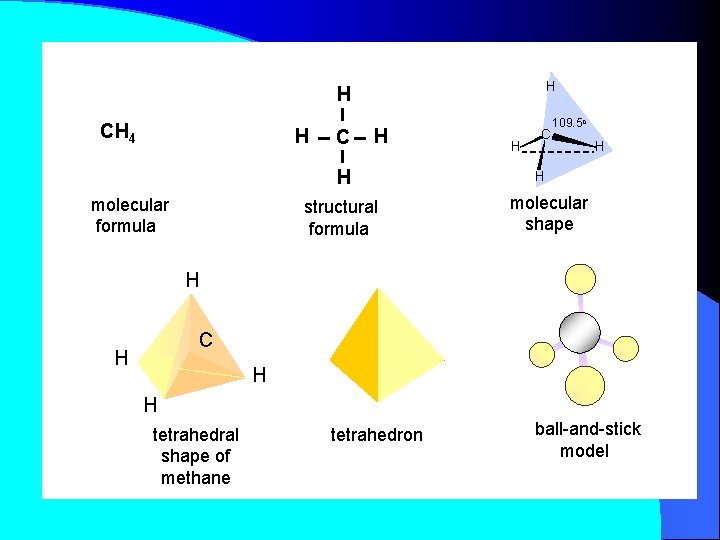
H H CH 4 H C H H molecular formula structural formula H C 109. 5 o H H molecular shape H C H H H tetrahedral shape of methane tetrahedron ball-and-stick model
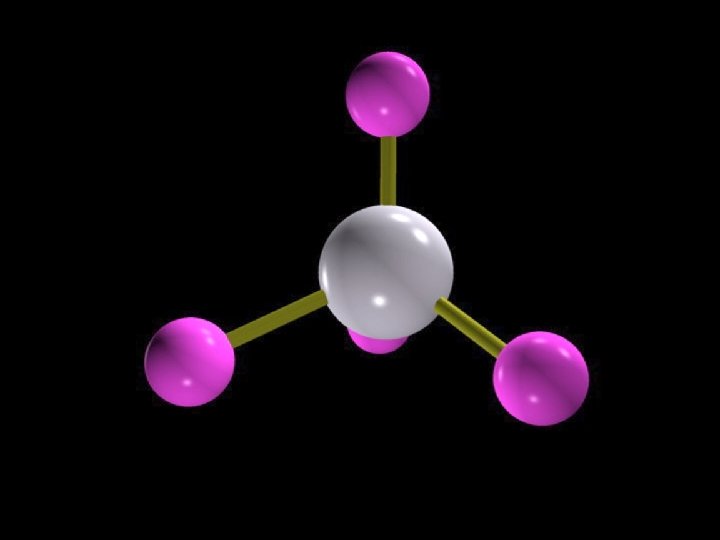
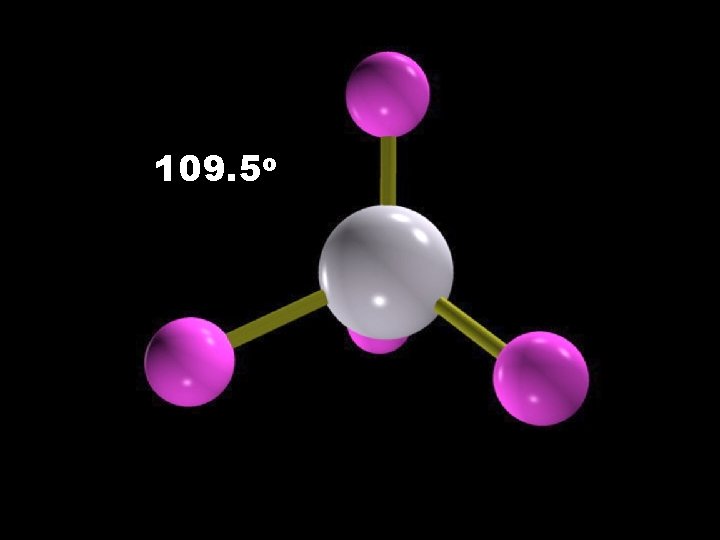
109. 5 o
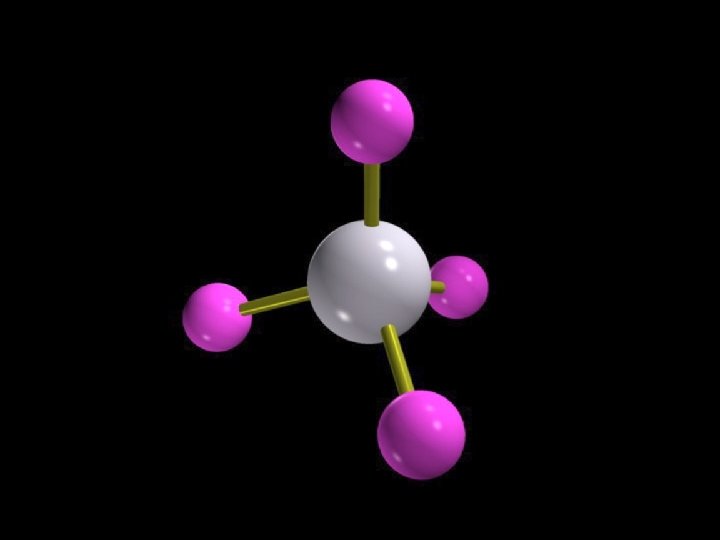
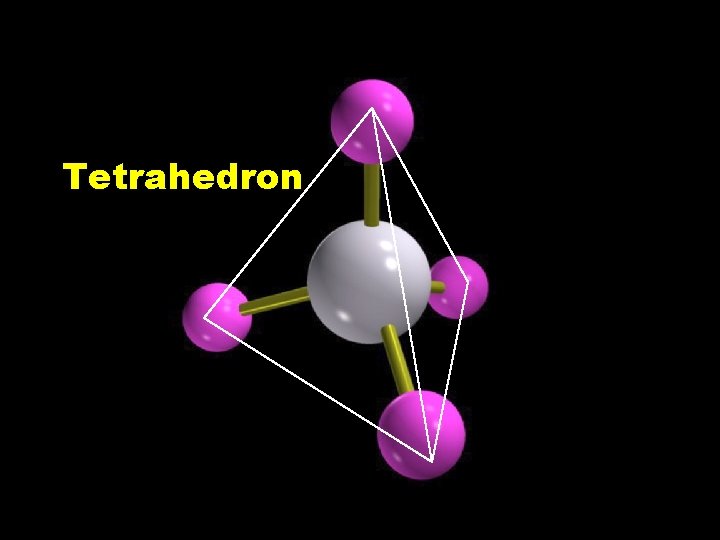
Tetrahedron
Central Atom
Central Atom
Substituents
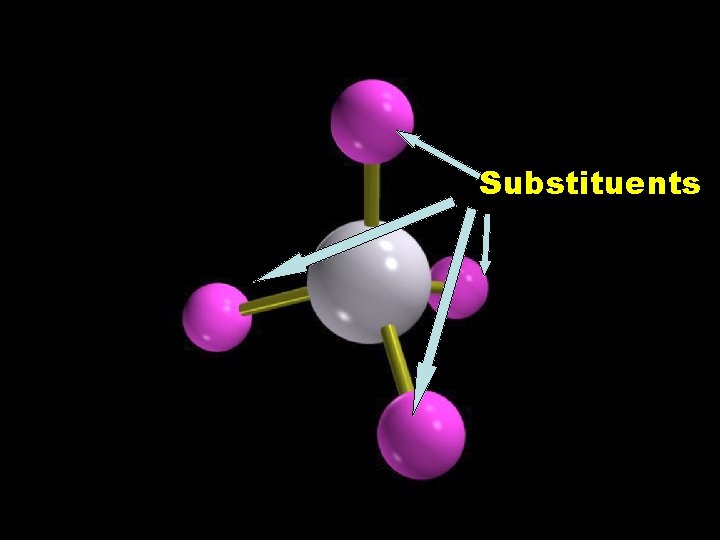
Substituents
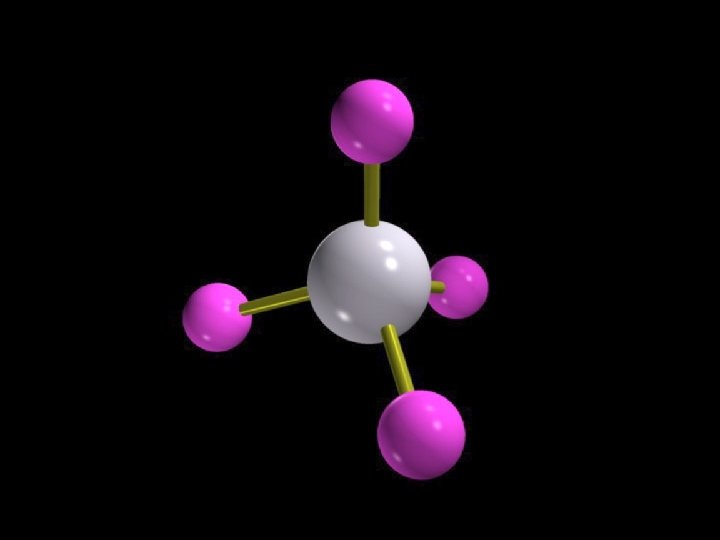
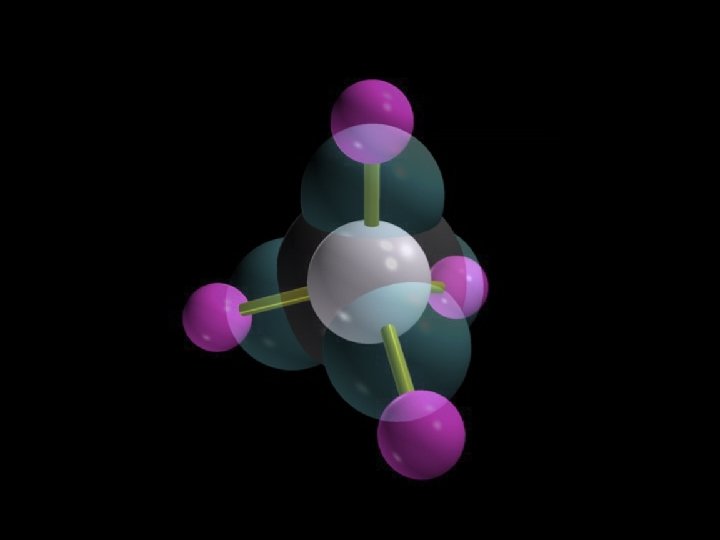
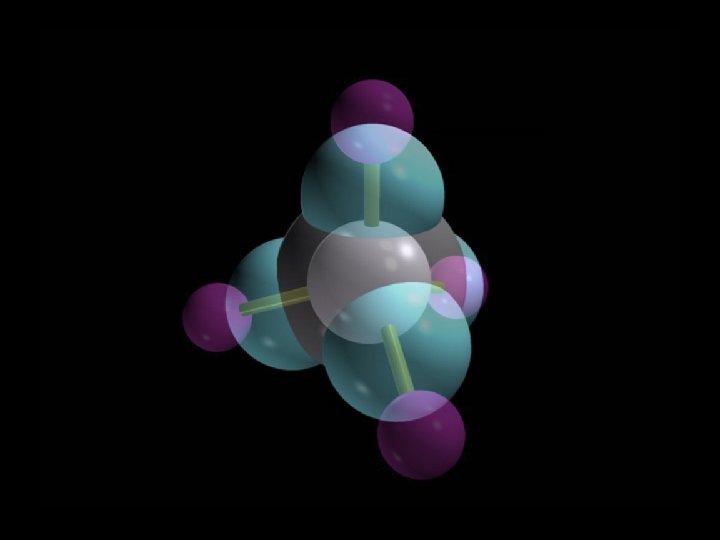
Methane, CH 4
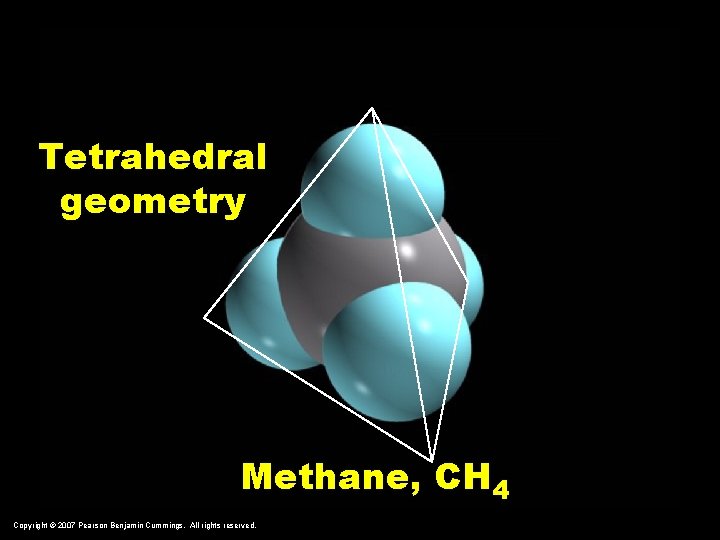
Tetrahedral geometry Methane, CH 4 Copyright © 2007 Pearson Benjamin Cummings. All rights reserved.
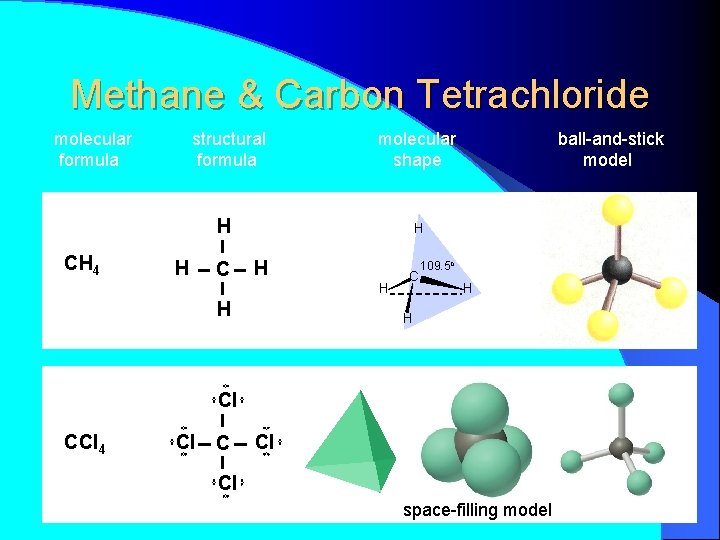
Methane & Carbon Tetrachloride molecular formula structural formula molecular shape H CH 4 H C ball-and-stick model H H C 109. 5 o H H Cl CCl 4 Cl Cl space-filling model
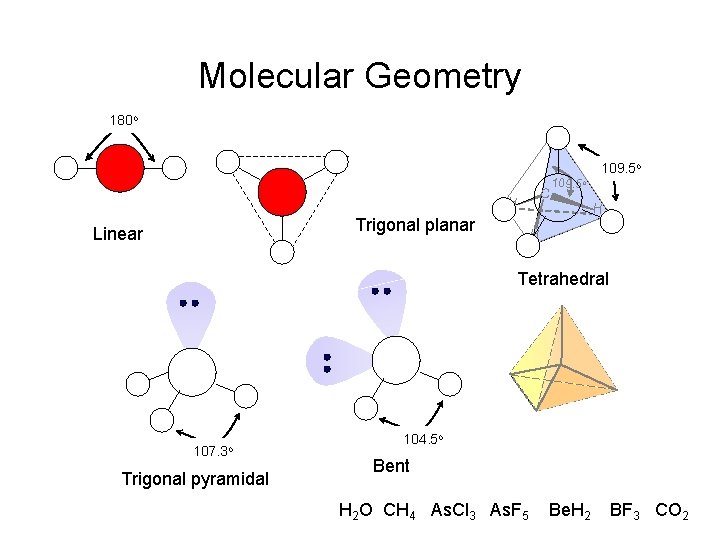
Molecular Geometry 180 o H H Trigonal planar Linear C 109. 5 o H H Tetrahedral 107. 3 o Trigonal pyramidal 104. 5 o Bent H 2 O CH 4 As. Cl 3 As. F 5 Be. H 2 BF 3 CO 2

A Lone Pair Pear
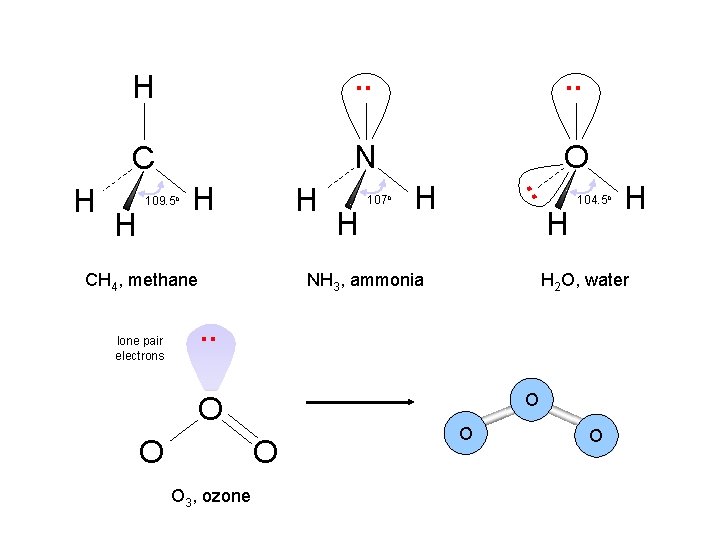
. . C N O H 109. 5 o H H CH 4, methane lone pair electrons H 107 o H . . H H H NH 3, ammonia 104. 5 o H 2 O, water . . O O O 3, ozone H O O
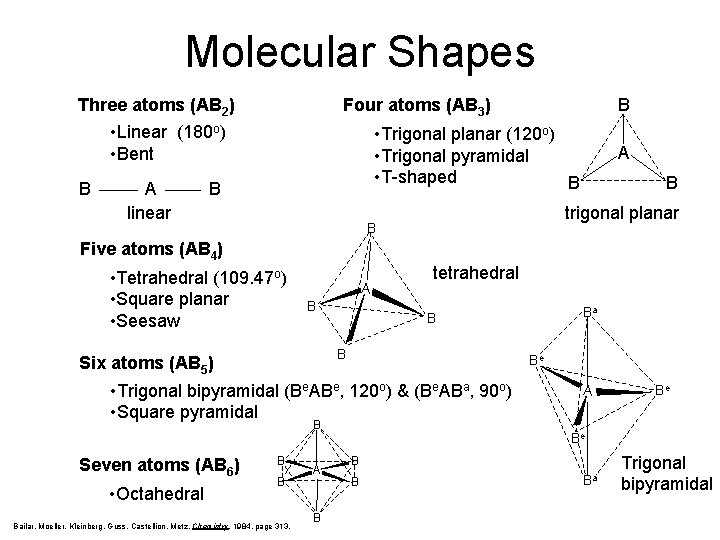
Molecular Shapes Three atoms (AB 2) Four atoms (AB 3) • Linear (180 o) • Bent B A linear B • Trigonal planar (120 o) • Trigonal pyramidal • T-shaped B B A B trigonal planar B Five atoms (AB 4) • Tetrahedral (109. 47 o) • Square planar • Seesaw A B tetrahedral B Six atoms (AB 5) Be • Trigonal bipyramidal (Be. ABe, 120 o) & (Be. ABa, 90 o) • Square pyramidal B Seven atoms (AB 6) • Octahedral B B Bailar, Moeller, Kleinberg, Guss, Castellion, Metz, Chemistry, 1984, page 313. A B Ba B A Be Be B B Ba Trigonal bipyramidal

Bonding and Shape of Molecules Number of Unshared Pairs Covalent Structure Shape Examples -Be- Linear Be. Cl 2 Trigonal planar BF 3 2 0 3 0 4 0 C Tetrahedral CH 4, Si. Cl 4 3 1 : Number of Bonds Pyramidal NH 3, PCl 3 2 2 Bent H 2 O, H 2 S, SCl 2 B : N O:
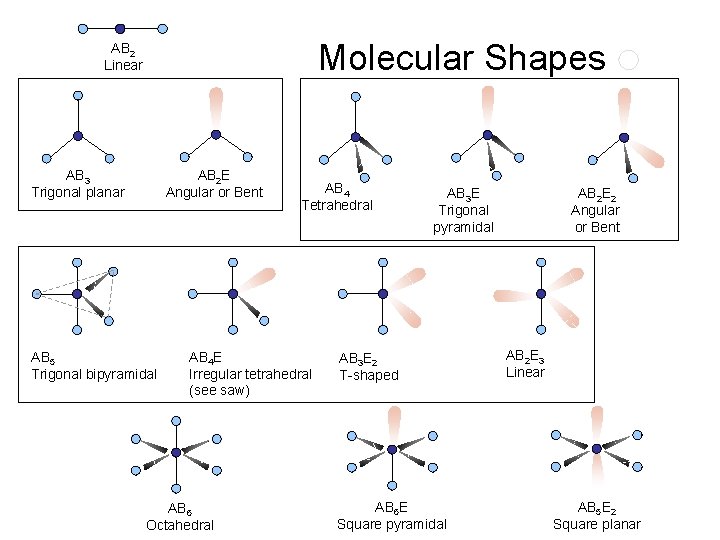
Molecular Shapes AB 2 Linear AB 3 Trigonal planar AB 2 E Angular or Bent AB 5 Trigonal bipyramidal AB 4 Tetrahedral AB 4 E Irregular tetrahedral (see saw) AB 6 Octahedral AB 3 E Trigonal pyramidal AB 3 E 2 T-shaped AB 6 E Square pyramidal AB 2 E 2 Angular or Bent AB 2 E 3 Linear AB 5 E 2 Square planar
- Slides: 23