Molecular Formulas Organics Nomenclature Molecular Bonding Molecule a

Molecular Formulas Organics Nomenclature
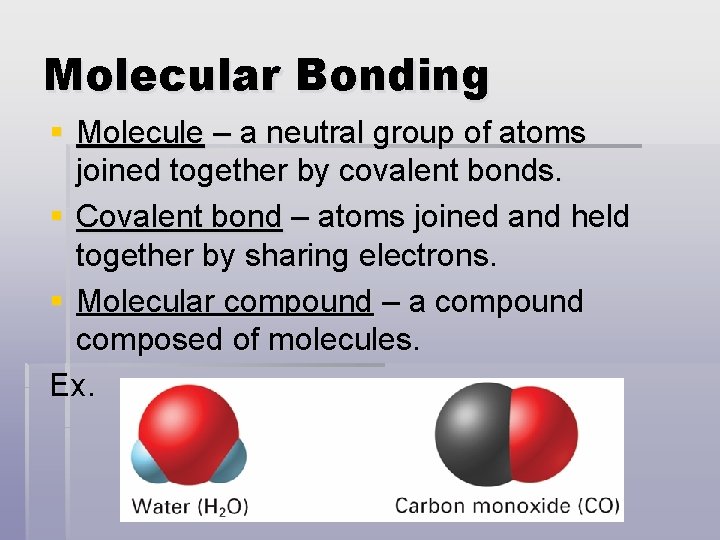
Molecular Bonding § Molecule – a neutral group of atoms joined together by covalent bonds. § Covalent bond – atoms joined and held together by sharing electrons. § Molecular compound – a compound composed of molecules. Ex.

Molecular Bonding § Diatomic molecule – a molecule consisting of 2 atoms. Ex. Oxygen (O 2) § Seven elements exist as diatomic molecules: H 2, O 2, N 2, F 2, Br 2, I 2, Cl 2 § This relates only to these elements as isolated, neutral atoms.

Difference between ionic and molecular

§ Ethane, a component of natural gas, is also a molecular compound.
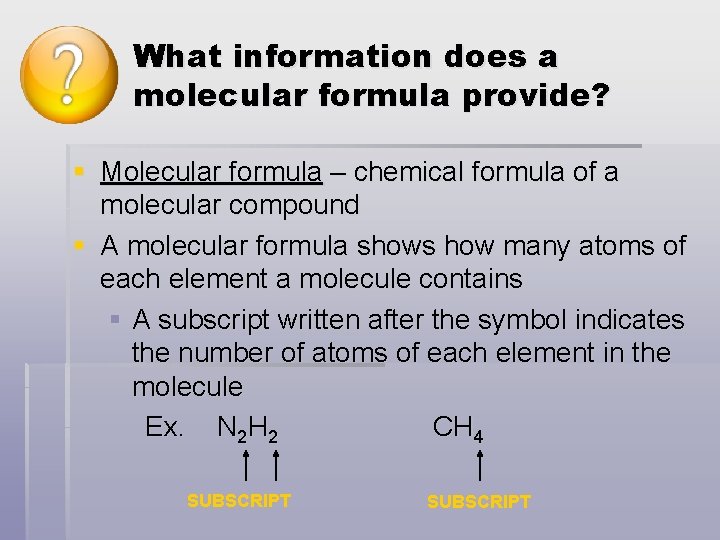
What information does a molecular formula provide? § Molecular formula – chemical formula of a molecular compound § A molecular formula shows how many atoms of each element a molecule contains § A subscript written after the symbol indicates the number of atoms of each element in the molecule Ex. N 2 H 2 CH 4 SUBSCRIPT
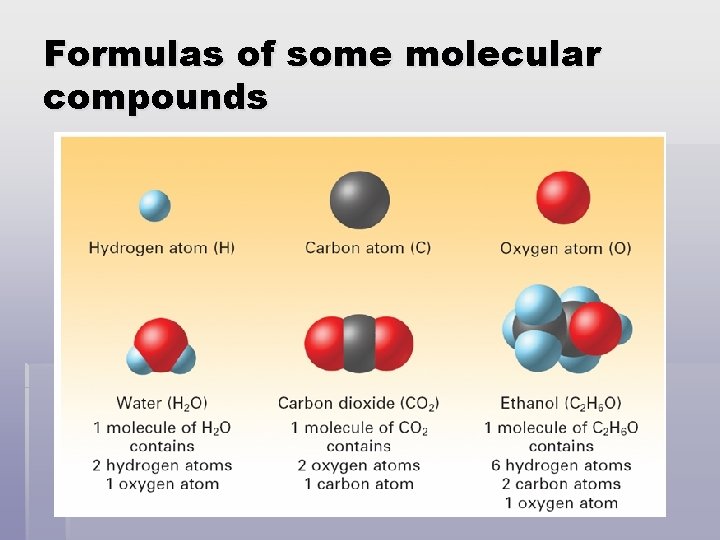
Formulas of some molecular compounds
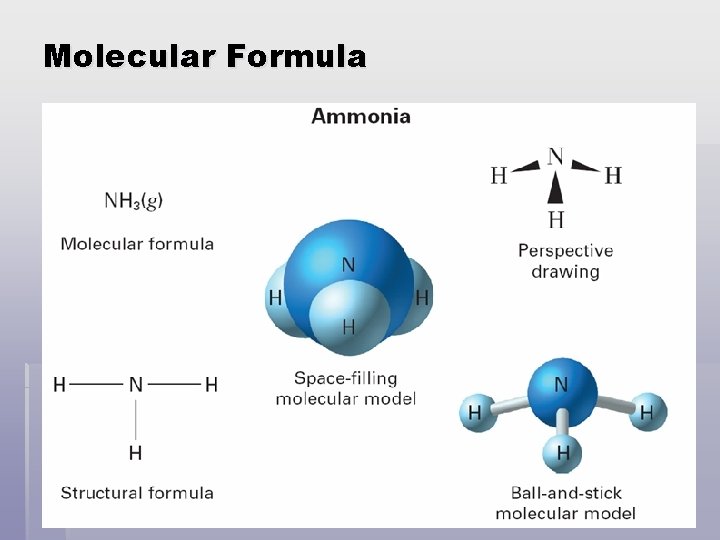
Molecular Formula

1. A molecular formula shows a. how many atoms of each element a molecule contains. b. a molecule's structure. c. which atoms are bonded together. d. how atoms are arranged in space.

Octet Rule in Covalent Bonding § In covalent bonds, electron sharing usually occurs so that atoms attain the electron configuration of noble gases § Octet Rule – in forming compounds, atoms tend to achieve the electron configuration of a noble gas § Noble gases: 8 e-’s in its highest occupied energy level (ns 2 np 6) = stable, inert, unreactive
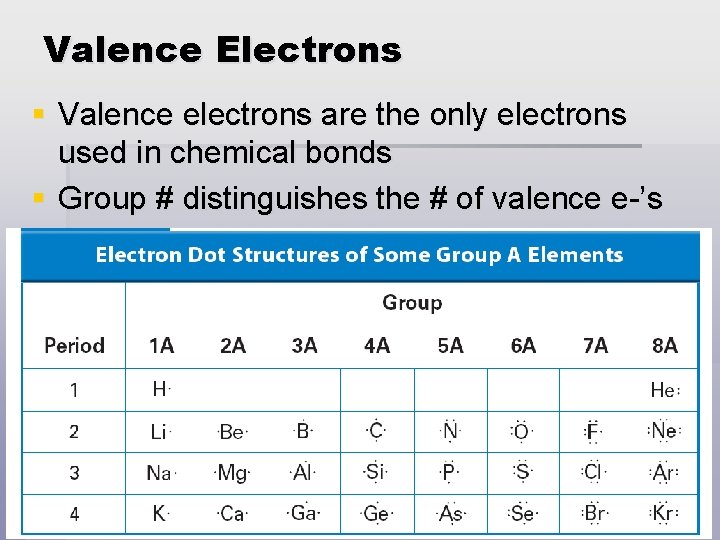
Valence Electrons § Valence electrons are the only electrons used in chemical bonds § Group # distinguishes the # of valence e-’s
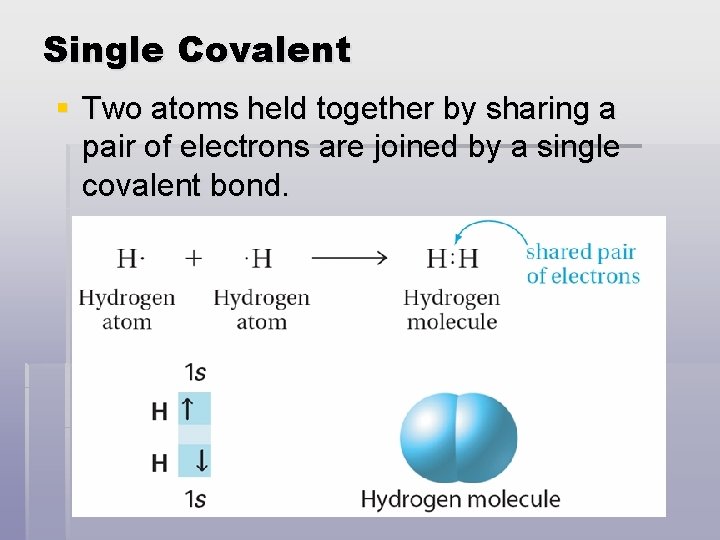
Single Covalent § Two atoms held together by sharing a pair of electrons are joined by a single covalent bond.

Single Covalent
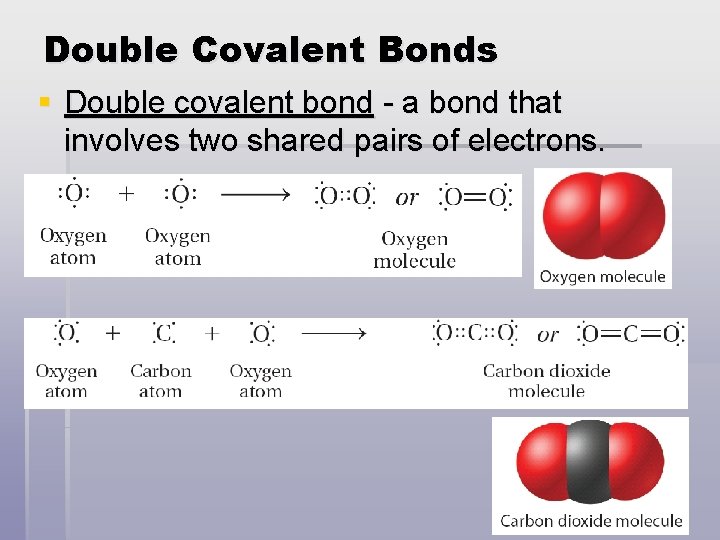
Double Covalent Bonds § Double covalent bond - a bond that involves two shared pairs of electrons.
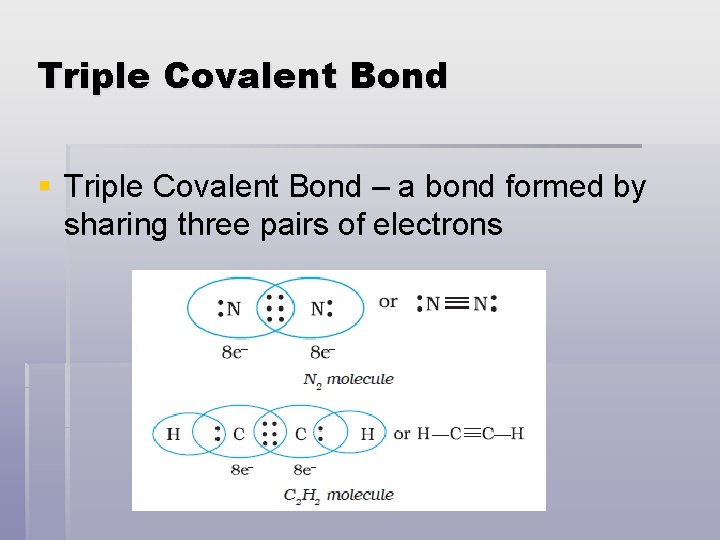
Triple Covalent Bond § Triple Covalent Bond – a bond formed by sharing three pairs of electrons

1. In covalent bonding, atoms attain the configuration of noble gases by a. b. c. d. losing electrons. gaining electrons. transferring electrons. sharing electrons.

2. Electron dot diagrams are superior to molecular formulas in that they a. show which electrons are shared. b. indicate the number of each kind of atom in the molecule. c. show the arrangement of atoms in the molecule. d. are easier to write or draw.

3. Which of the following molecules would contain a bond formed when atoms share three pairs of electrons? a. b. c. d. Se 2 As 2 Br 2 Te 2
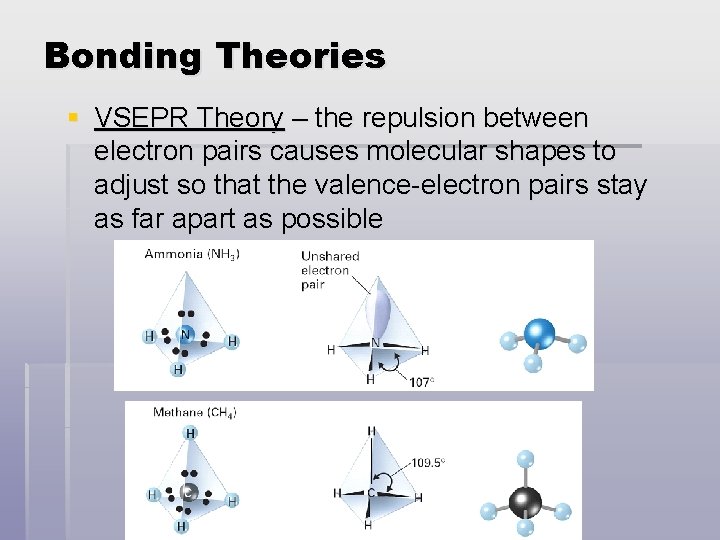
Bonding Theories § VSEPR Theory – the repulsion between electron pairs causes molecular shapes to adjust so that the valence-electron pairs stay as far apart as possible
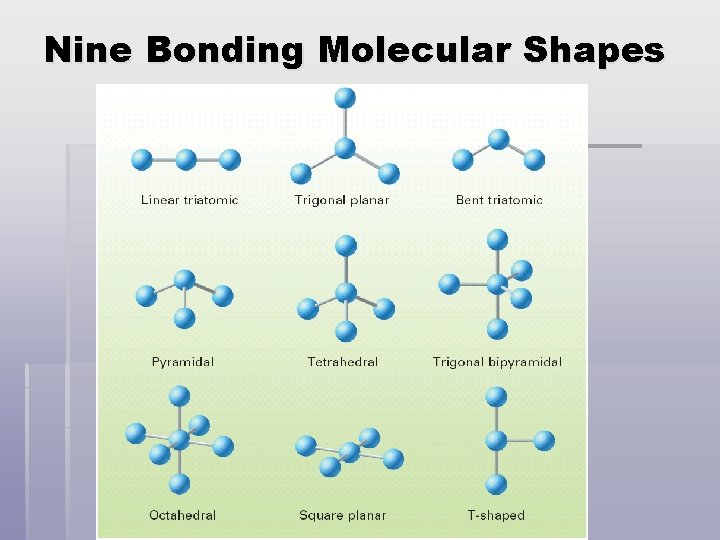
Nine Bonding Molecular Shapes

Organics - Hydrocarbons § Hydrocarbons – simplest organic compounds that contain only carbon & hydrogen Ex. methane (CH 4) & ethane (C 2 H 6) § One carbon atom can form a single covalent bond with four hydrogen atoms. § Principle of organics: carbon has 4 valence ’s; will always form 4 covalent bonds e-
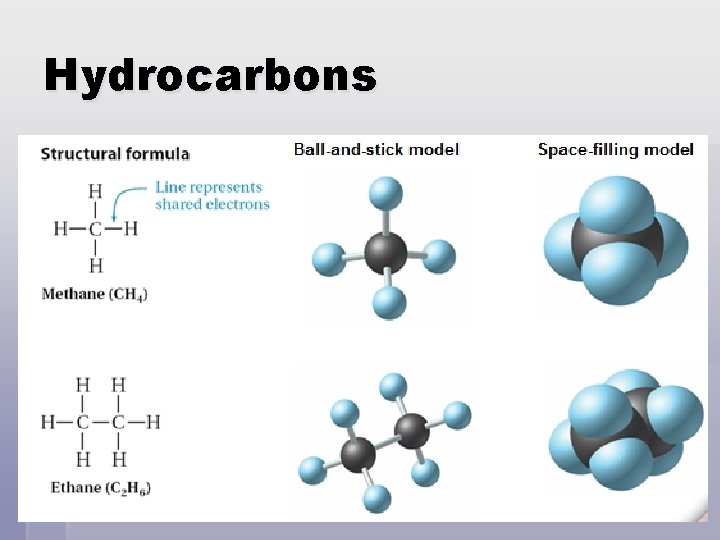
Hydrocarbons

Hydrocarbon - Alkanes § Alkane – hydrocarbons with only single covalent bonds. § Arranged in a straight chain or in a chain that has branches. § Cn. H 2 n+2 results in the highest ratio of H/C ratio for a stable hydrocarbon. § Saturated hydrocarbons – H/C ratio is at a max.
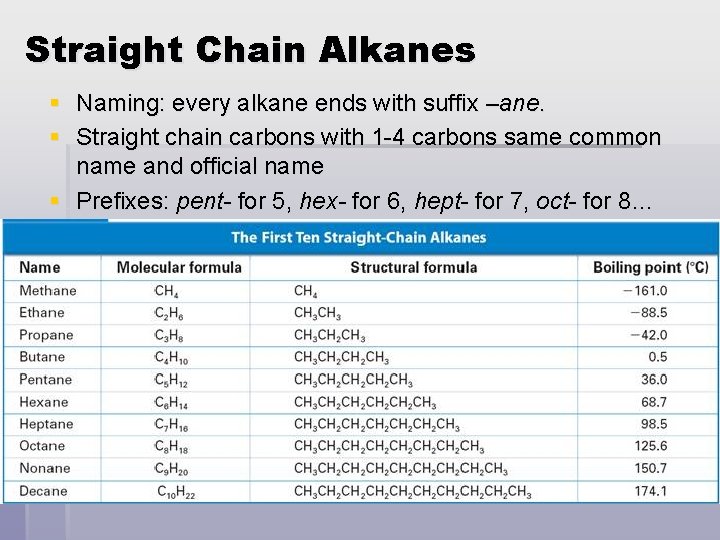
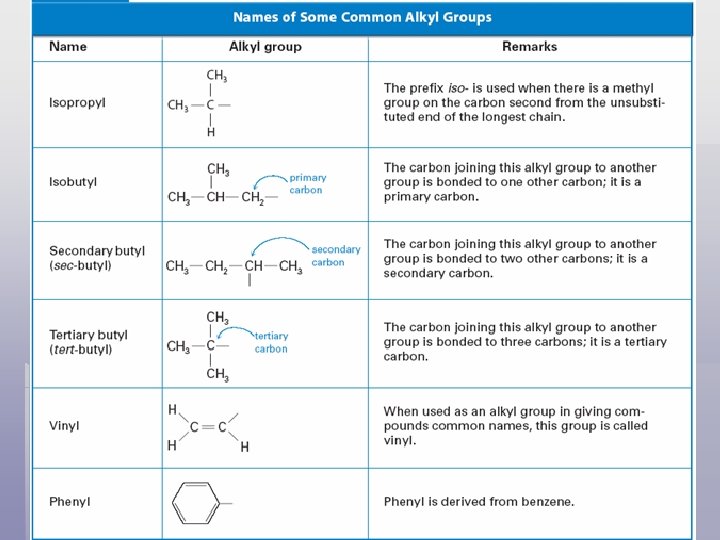
Straight Chain Alkanes § Naming: every alkane ends with suffix –ane. § Straight chain carbons with 1 -4 carbons same common name and official name § Prefixes: pent- for 5, hex- for 6, hept- for 7, oct- for 8…

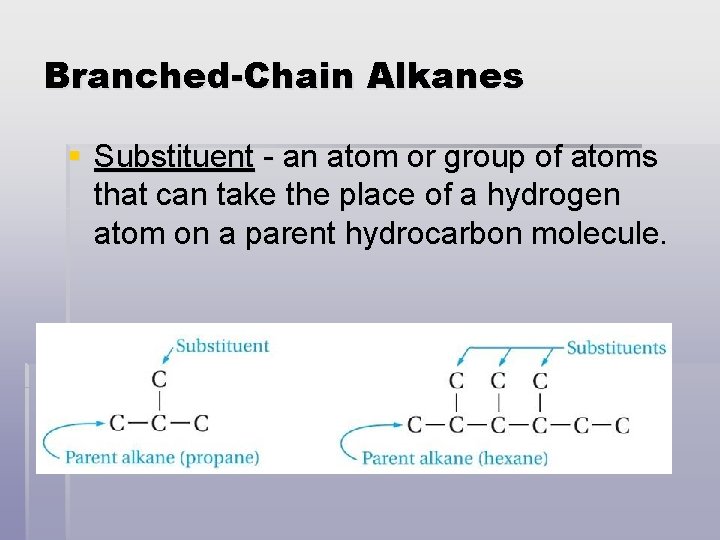
Branched-Chain Alkanes § Substituent - an atom or group of atoms that can take the place of a hydrogen atom on a parent hydrocarbon molecule.
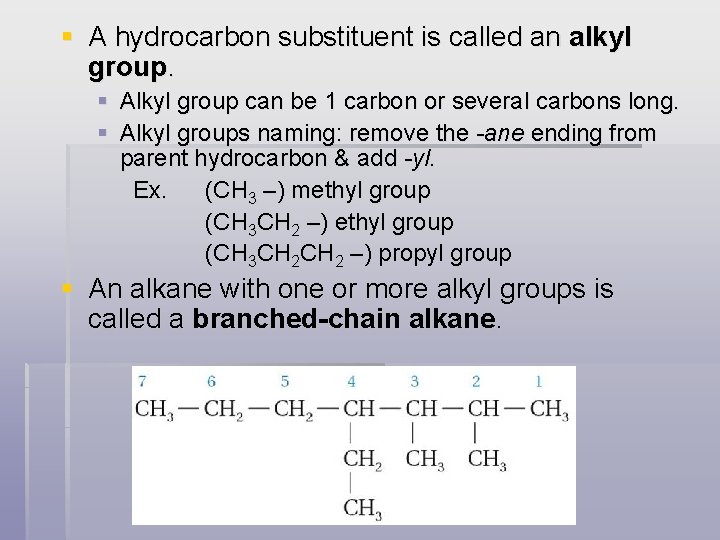
§ A hydrocarbon substituent is called an alkyl group. § Alkyl group can be 1 carbon or several carbons long. § Alkyl groups naming: remove the -ane ending from parent hydrocarbon & add -yl. Ex. (CH 3 –) methyl group (CH 3 CH 2 –) propyl group § An alkane with one or more alkyl groups is called a branched-chain alkane.

Naming Branched-Chain Alkanes § Name this compound using the IUPAC system. Notice that the longest chain is not written in a straight line. 1 2 3 4 5 6 3, 3 dimethylhexane
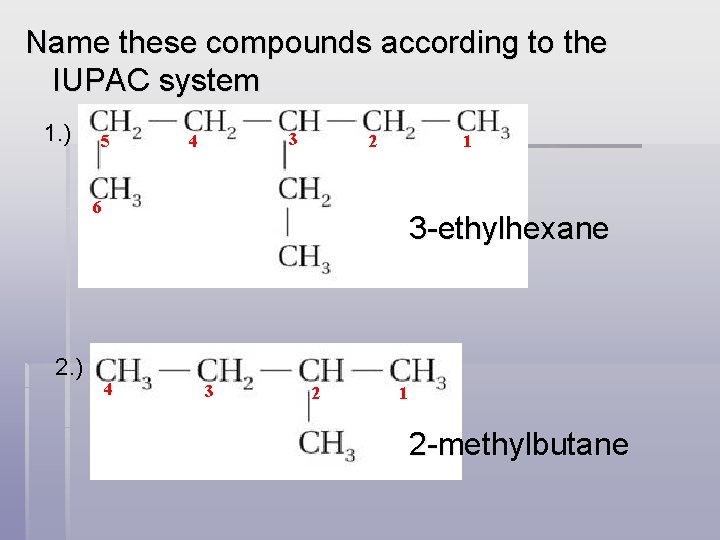
Name these compounds according to the IUPAC system 1. ) 5 3 4 2 1 6 2. ) 3 -ethylhexane 4 3 2 1 2 -methylbutane
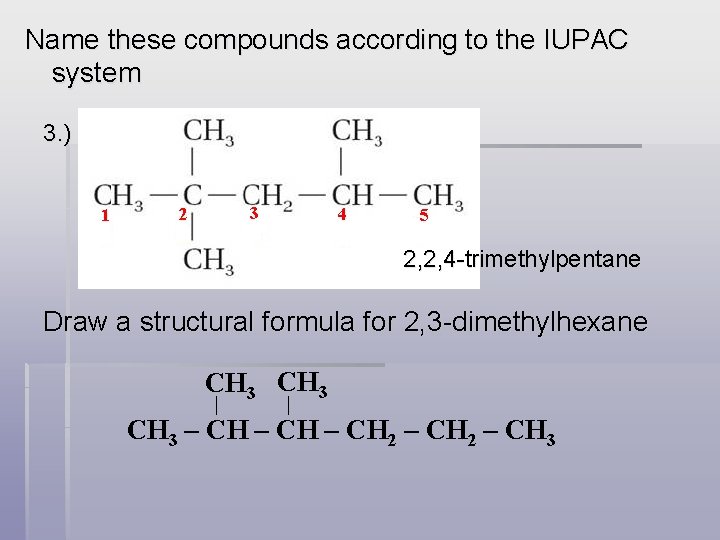
Name these compounds according to the IUPAC system 3. ) 1 2 3 4 5 2, 2, 4 -trimethylpentane Draw a structural formula for 2, 3 -dimethylhexane CH 3 – CH 2 – CH 3

1. Choose the correct words for the spaces. Because carbon has ______ valence electrons, it can form _______ bonds. a. b. c. d. four, four covalent four, four ionic six, six covalent six, four or fewer covalent

2. Alkanes are hydrocarbons that contain only ______ bonds. a. b. c. d. carbon-carbon single covalent carbon-hydrogen ionic

Hydrocarbons - Alkenes § Alkenes – hydrocarbons that contain one or more carbon-carbon double covalent bonds. § Cn. H 2 n unsaturated hydrocarbon. § Naming: same IUPAC rules, ending in –ene.

Hydrocarbons - Alkynes § Alkynes – hydrocarbons that contain one or more carbon triple covalent bonds. § Cn. H 2 n-2 unsaturated hydrocarbon. § Naming: same IUPAC rules, ending in -yne Name Chemical Formula Ethyne C 2 H 2 Propyne C 3 H 4 Butyne C 4 H 6 Pentyne C 5 H 8 Hexyne C 6 H 10 Heptyne C 7 H 12 Octyne C 8 H 14 Nonyne C 9 H 16 Decyne C 10 H 18

1. What is the correct name for a compound with a C-C-C=C-C carbon skeleton? a. b. c. d. 3 -pentyne 2 -pentene 3 -pentene 4 -pentene

2. Which of the following substances is an alkyne? a. b. c. d. C 6 H 12 C 2 H 4 C 2 H 2 C 4 H 8

Isomers § Isomers – compounds that have the same molecular formula but different molecular structures § Structural isomers differ in physical properties such as boiling point and melting point. They also have different chemical reactivities.
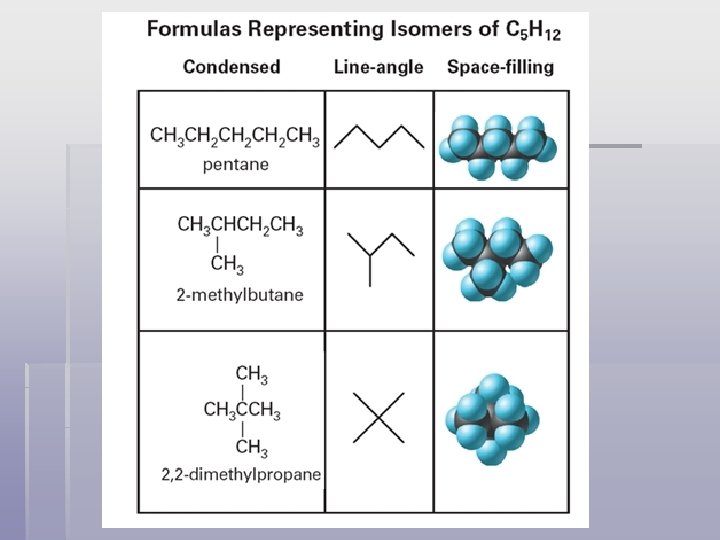
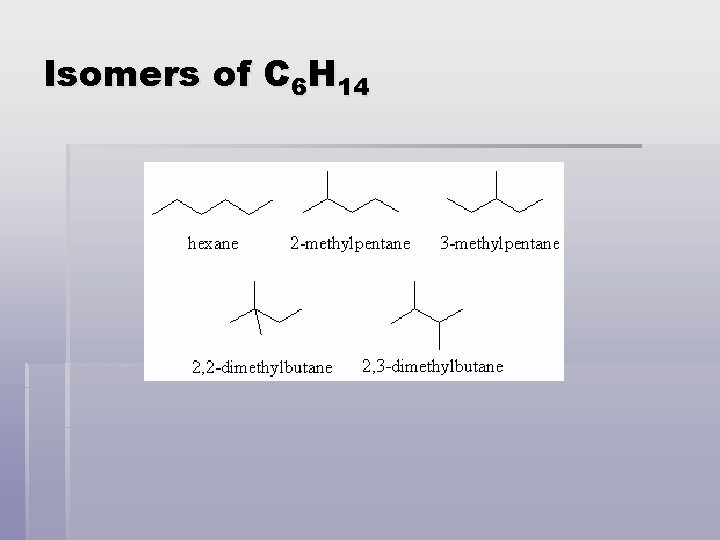
Isomers of C 6 H 14

Stereoisomers § Stereoisomers – molecules in which the atoms are joined in the same order, but the positions of the atoms in space are different. § 2 types of stereoisomers 1. ) geometric isomers 2. ) optical isomers
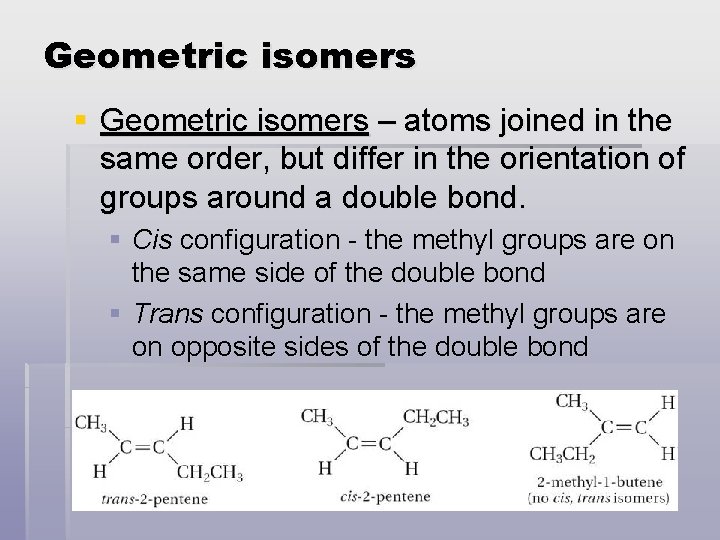
Geometric isomers § Geometric isomers – atoms joined in the same order, but differ in the orientation of groups around a double bond. § Cis configuration - the methyl groups are on the same side of the double bond § Trans configuration - the methyl groups are on opposite sides of the double bond
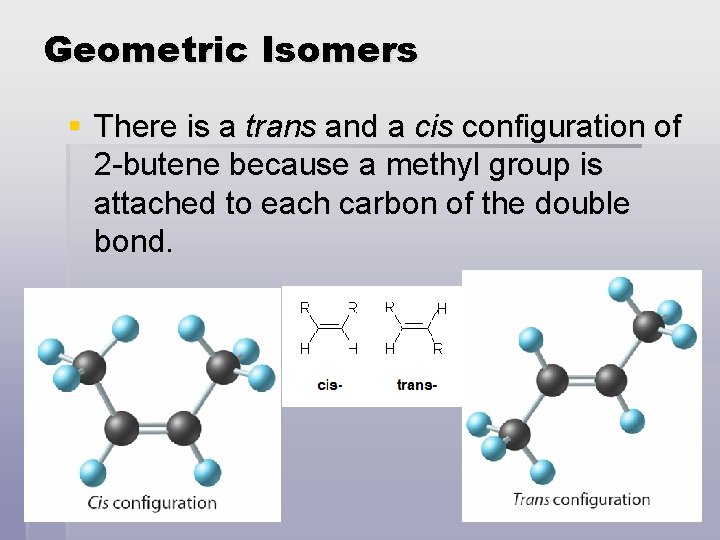
Geometric Isomers § There is a trans and a cis configuration of 2 -butene because a methyl group is attached to each carbon of the double bond.
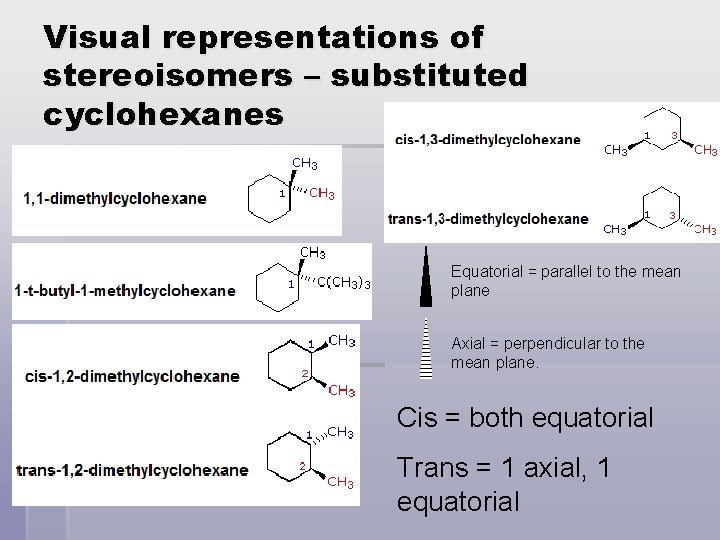
Visual representations of stereoisomers – substituted cyclohexanes Equatorial = parallel to the mean plane Axial = perpendicular to the mean plane. Cis = both equatorial Trans = 1 axial, 1 equatorial

Optical Isomers § A carbon with four different atoms or groups attached is an asymmetric carbon. § Pairs of molecules that differ only in the way that four different groups are arranged around a central carbon atom are called optical isomers. H, F, Cl, and Br are attached to a single carbon atom • Mirror images are non superimposable

Which compound has an asymmetric carbon? 1. ) 2. ) 3. ) 4. ) Not asymmetric, central carbon has two CH 3 groups attached. Central carbon is asymmetric, one H, one OH, one CH 3, and one CH 2 CH 3 group attached Central carbon is asymmetric, one Cl, one H, one CH 3, and one CHO Not asymmetric, central carbon has two CH 3 groups attached.

1. Structural isomers have different properties because they have a. b. c. d. a different number of bonds. different types of bonds. different substituents. a different order of atoms.

2. Substituent groups on opposite sides of a double bond are said to be in the _______ configuration. If the groups are on the same side of the bond, then they are in the _______ configuration. a. b. c. d. cis, trans, cis trans, iso stereo, cis

3. A carbon atom that has four different substituents always forms a. b. c. d. an optical isomer. a saturated hydrocarbon. a geometric isomer. an unsaturated hydrocarbon.

Hydrocarbon Rings § Cyclic hydrocarbons – compounds that contain a hydrocarbon ring § Saturated hydrocarbons § Cn. H 2 n : (CH 2)n

Hydrocarbon Rings
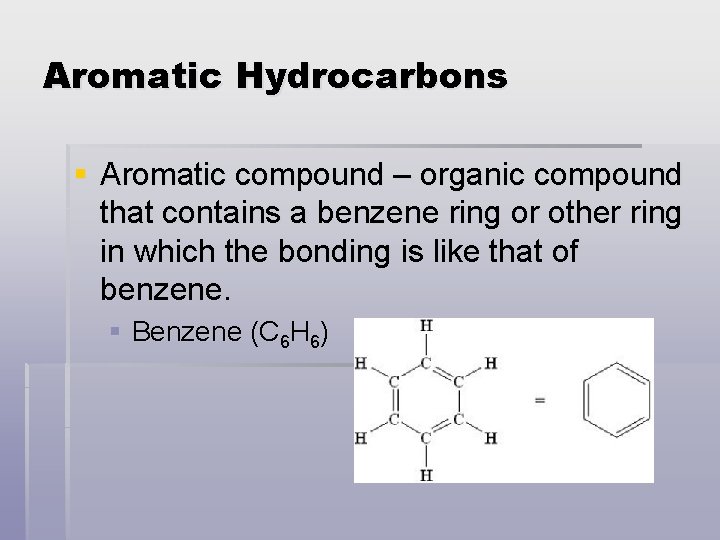
Aromatic Hydrocarbons § Aromatic compound – organic compound that contains a benzene ring or other ring in which the bonding is like that of benzene. § Benzene (C 6 H 6)
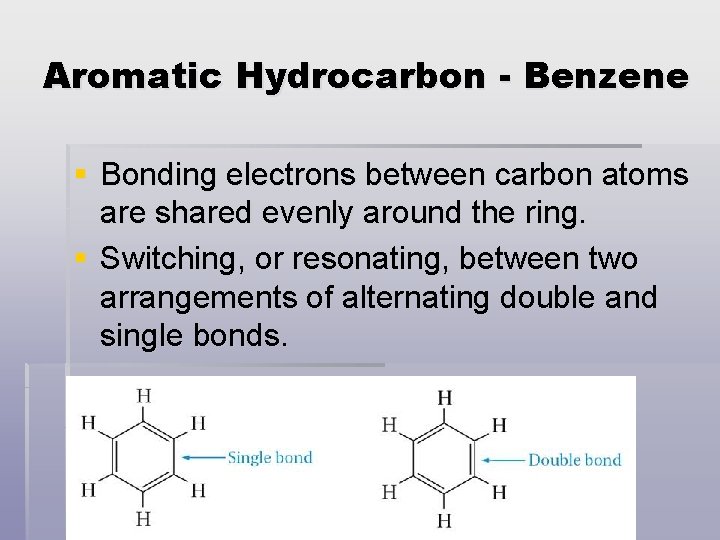
Aromatic Hydrocarbon - Benzene § Bonding electrons between carbon atoms are shared evenly around the ring. § Switching, or resonating, between two arrangements of alternating double and single bonds.
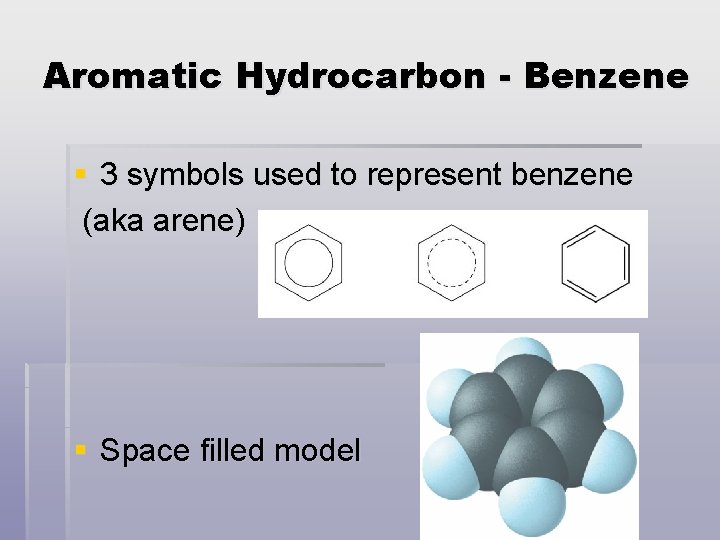
Aromatic Hydrocarbon - Benzene § 3 symbols used to represent benzene (aka arene) § Space filled model

Substituted Aromatic Compounds § Compounds with a substituent on a benzene ring are named as a derivative of benzene. When benzene is a substituent, it is called a phenyl group
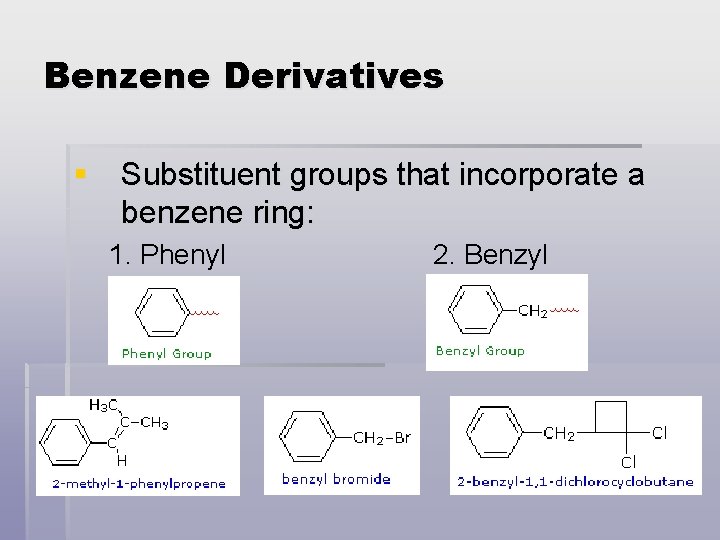
Benzene Derivatives § Substituent groups that incorporate a benzene ring: 1. Phenyl 2. Benzyl

Benzene Derivatives § Disubstituted benzenes: the following prefixes are used: § Ortho- for 1, 2 - substituents § Meta- for 1, 3 - substituents § Para- for 1, 4 - substituents
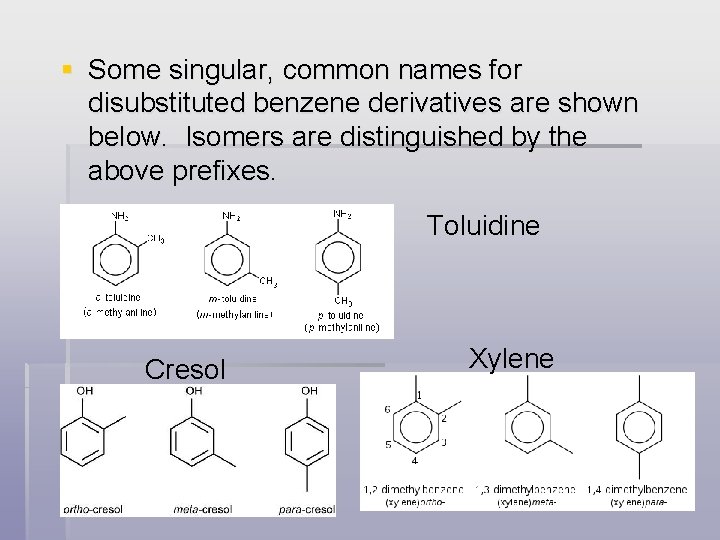
§ Some singular, common names for disubstituted benzene derivatives are shown below. Isomers are distinguished by the above prefixes. Toluidine Cresol Xylene

1. When an alkane has a benzene ring as a substituent, the ring is called _______ group. a. b. c. d. a benzene a toluene a phenyl an arene

Review of Some Terms § Alkane – a hydrocarbon containing only single covalent bonds; saturated hydrocarbons. § Alkene – a hydrocarbon containing one or more carbon-carbon double bonds; unsaturated hydrocarbons. § Alkyne – a hydrocarbon containing a carbon-carbon triple bond, unsaturated carbon. § Cycloalkane – an alicyclic hydrocarbon with a saturated ring. § Aliphatic – any straight-chain or branched-chain alkane, alkene, or alkyne. § Aromatic – an organic compound that contains a benzene ring or other ring in which the bonding is like that of benzene. § Alkyl group – a hydrocarbon substituent. Ex. the methyl group (– CH 3)

How are organic compounds classified? § functional group is a specific arrangement of atoms in an organic compound that is capable of characteristic chemical reactions
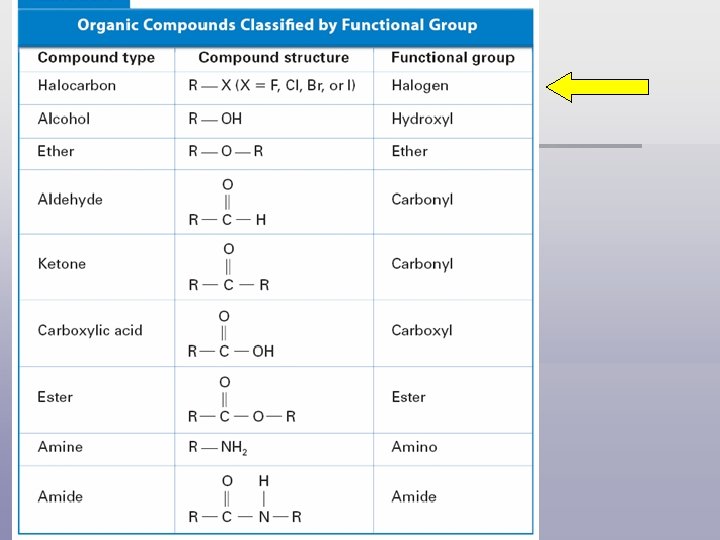

Halogen Substituents § Halocarbons – a class of organic compounds containing covalently bonded F, Cl, Br, or I; all halogens (Group 7 nonmetals) § IUPAC: named as substituents on parent hydrocarbon Fluorine Fluoro. Chlorine Chloro- Bromine Bromo- Iodine Iodo- § Common name: Consists of 2 parts 1. Name the hydrocarbon portion of the molecule as an alkyl group, such as methyl- or ethyl 2. Give the halogen an –ide ending.

Halogen Substituents § Alkyl halides - halocarbons in which a halogen is attached to a carbon of an aliphatic chain § Aryl halides - Halocarbons in which a halogen is attached to a carbon of an aromatic (arene) ring
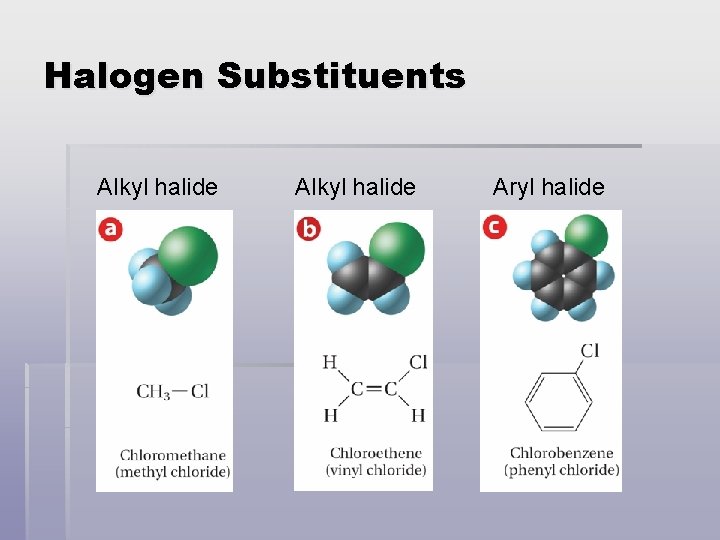
Halogen Substituents Alkyl halide Aryl halide

How are halocarbons prepared? § A common type of organic reaction is a substitution reaction, in which an atom, or a group of atoms, replaces another atom or group of atoms. § A halogen can replace a hydrogen atom on an alkane to produce a halocarbon.

Substitution Rx. § A halogen can replace a hydrogen atom on an alkane to produce a halocarbon. § A catalyst is needed to break relatively strong covalent bonds.

Substitution Rx. § Simple halogenation reactions can produce a mixture of mono-, di-, tri-, and tetrachloromethanes. § Common names are in parenthesis
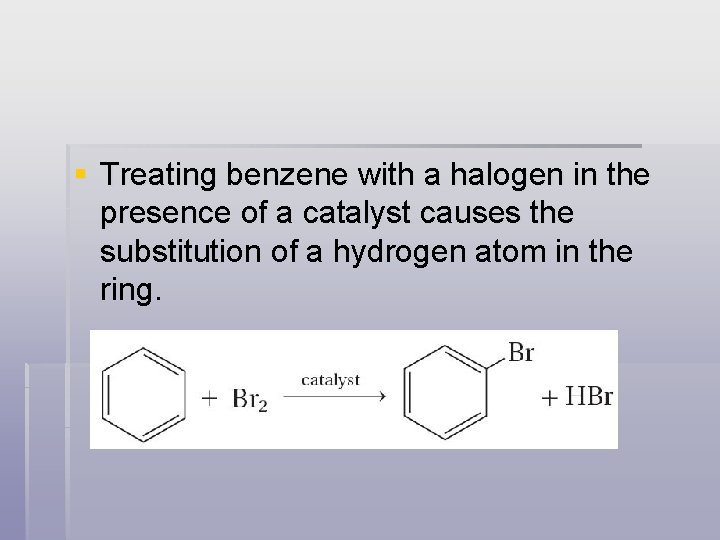
§ Treating benzene with a halogen in the presence of a catalyst causes the substitution of a hydrogen atom in the ring.

1. Organic molecules are classified according to their a. b. c. d. functional groups. longest chain. derivatives. number of rings.

2. What is the correct IUPAC name for the compound CH 2 Br? a. b. c. d. methylbromoethylbromide dibromopropane 1, 3 -dibromopropane propyl-1, 3 -dibromide

3. Which reaction will produce a halocarbon? a. b. hydrogenation of an alkyl halide substitution reaction of a halogen with an alkane c. reaction of potassium hydroxide with an aryl halide d. reaction of sodium hydroxide with an alkyl halide

How are functional groups introduced to alkenes? § Addition reactions of alkenes are an important method of introducing new functional groups into organic molecules. § In an addition reaction, a substance is added at the double or triple bond of an alkene or alkyne. § Double and triple bonds between carbon atoms are much more reactive than single bonds between carbon atoms.
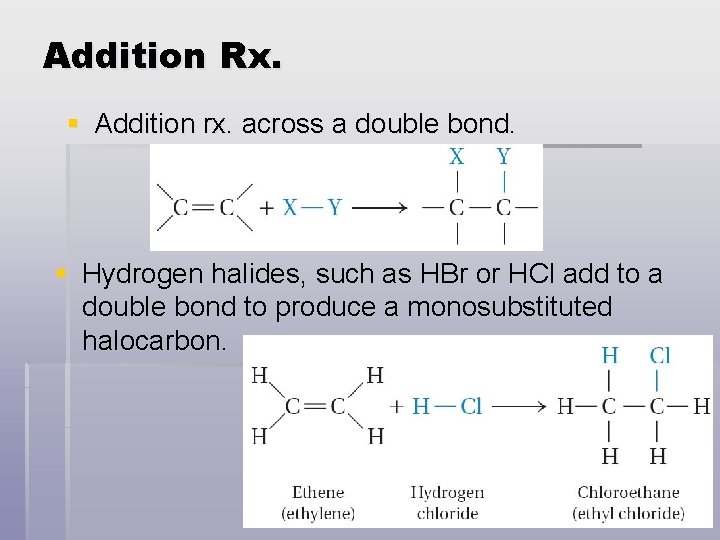
Addition Rx. § Addition rx. across a double bond. § Hydrogen halides, such as HBr or HCl add to a double bond to produce a monosubstituted halocarbon.

Addition Rx. § When the reagent X-Y is a halogen molecule such as chlorine or bromine, the product of the rx is a disubstituted halocarbon.
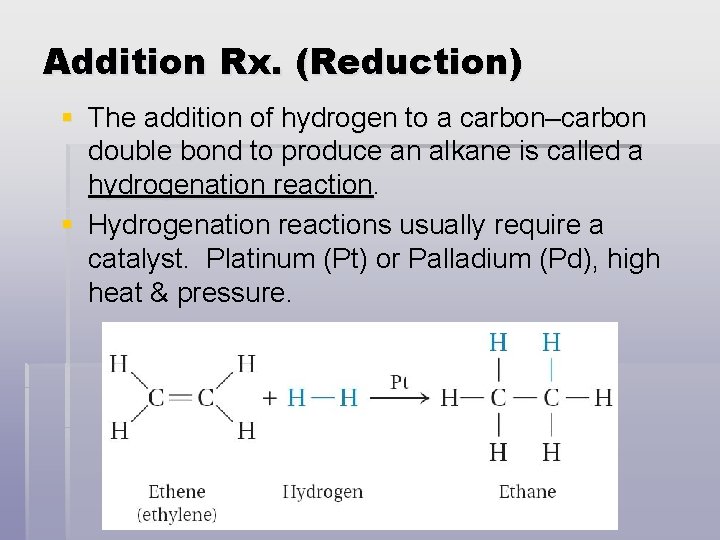
Addition Rx. (Reduction) § The addition of hydrogen to a carbon–carbon double bond to produce an alkane is called a hydrogenation reaction. § Hydrogenation reactions usually require a catalyst. Platinum (Pt) or Palladium (Pd), high heat & pressure.
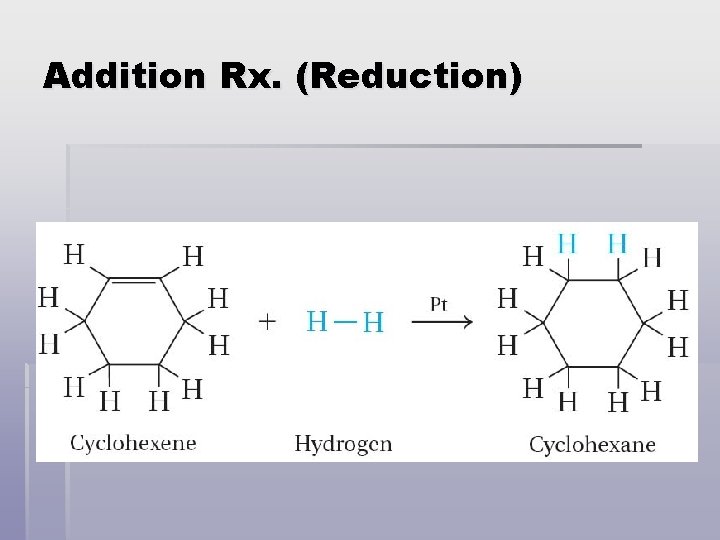
Addition Rx. (Reduction)

1. What type of reaction would convert CH 2=CH—CH 3 into CHBr— CH 3? a. b. c. d. substitution hydrogenation addition hydration
- Slides: 78