Molecular Formula A molecular formula shows the elements


















- Slides: 44

Molecular Formula A molecular formula shows the elements that a compound is made out of and the number of atoms of each element present in one molecule of the compound. The numbers are shown written as subscripts. e. g. The molecular formula of butane is C 4 H 10 One molecule of ethane contains 4 carbon atoms and 10 hydrogen atoms. The molecular formula of water is H 2 O One molecule of water contains 2 hydrogen atoms and one oxygen atom. Empirical Formula The empirical formula gives the simplest whole number ratio of each type of atom in a compound e. g. The molecular formula of butane is C 4 H 10 The empirical formula of butane is C 2 H 5

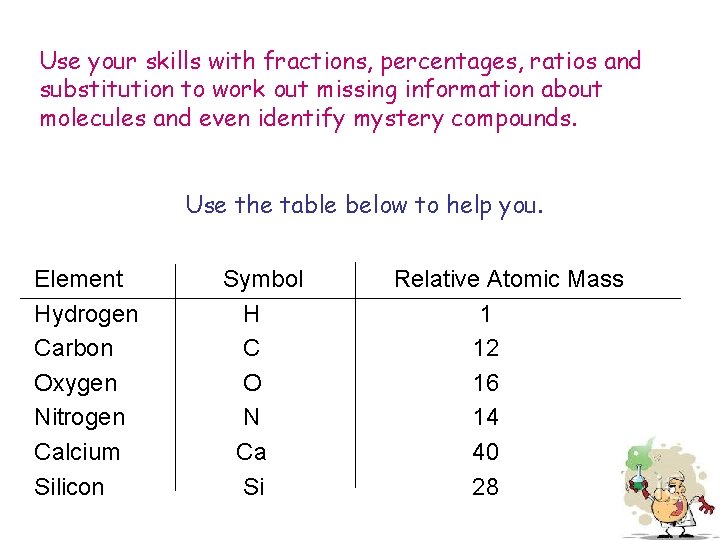
Relative molecular mass Molecules are incredibly light so when discussing how heavy a molecule is Chemists often find the relative molecular mass. This is measured in atomic mass units. The table below shows the relative atomic masses of various elements which can be used to calculate the relative molecular mass of molecules. e. g. The molecular formula of butane is C 4 H 10 The relative molecular mass of ethane is 4 x 12 + 10 x 1 = 58 atomic mass units Element Hydrogen Carbon Oxygen Nitrogen Calcium Silicon Symbol H C O N Ca Si Relative Atomic Mass 1 12 16 14 40 28

Elemental Analysis Is a technique that allows a chemist to workout (as a percentage) how much each element contributes to the total mass of the molecule. If you know these percentages you can work out what the empirical formula of a molecule so it can help Chemists work out what they have made. Ratio Percentages Fractions Substitution

Use your skills with fractions, percentages, ratios and substitution to work out missing information about molecules and even identify mystery compounds. Use the table below to help you. Element Hydrogen Carbon Oxygen Nitrogen Calcium Silicon Symbol H C O N Ca Si Relative Atomic Mass 1 12 16 14 40 28

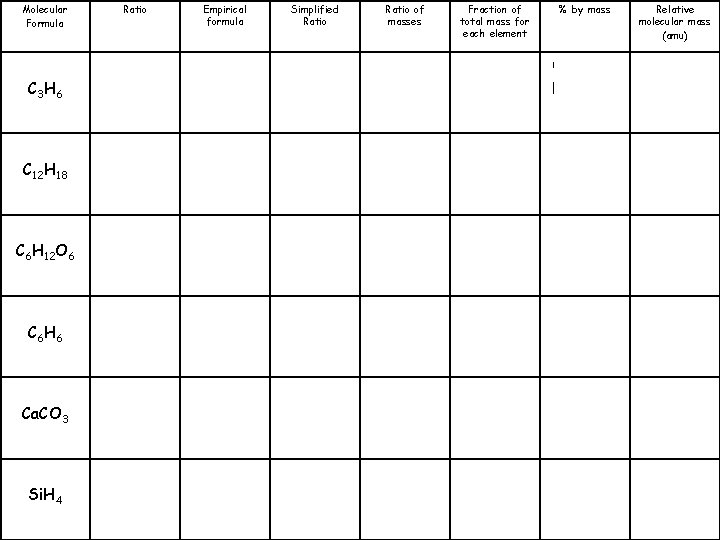
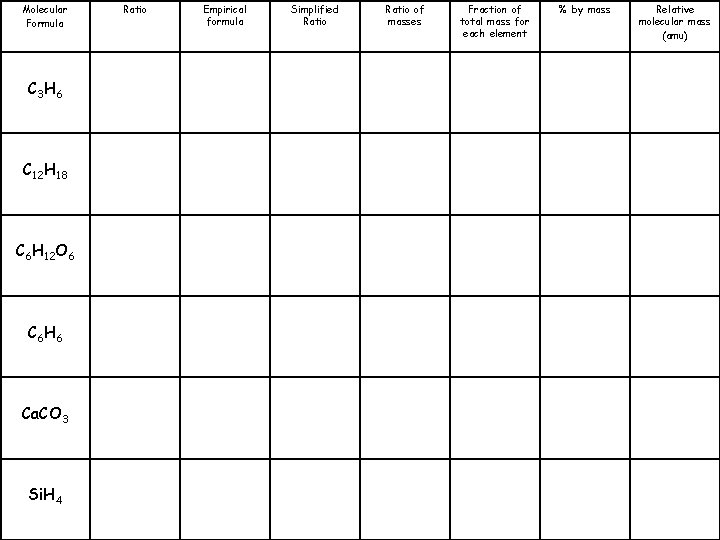
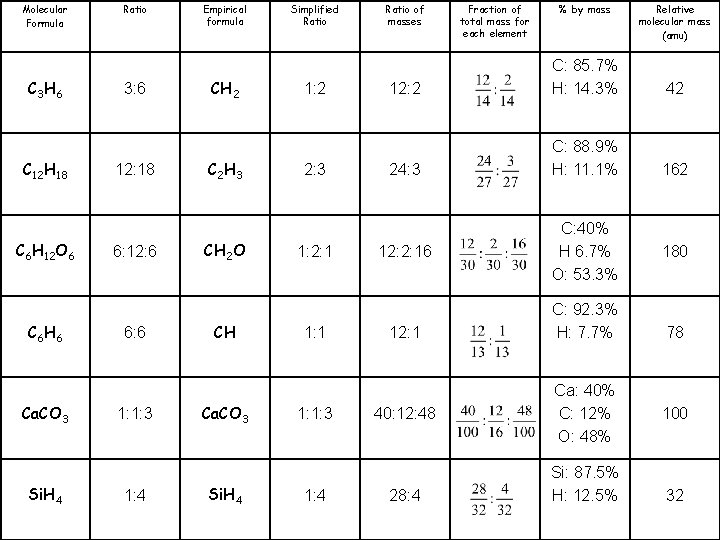
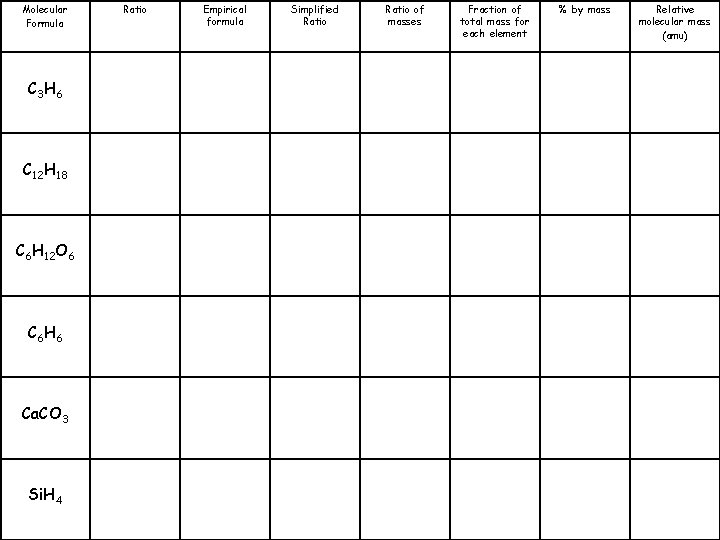
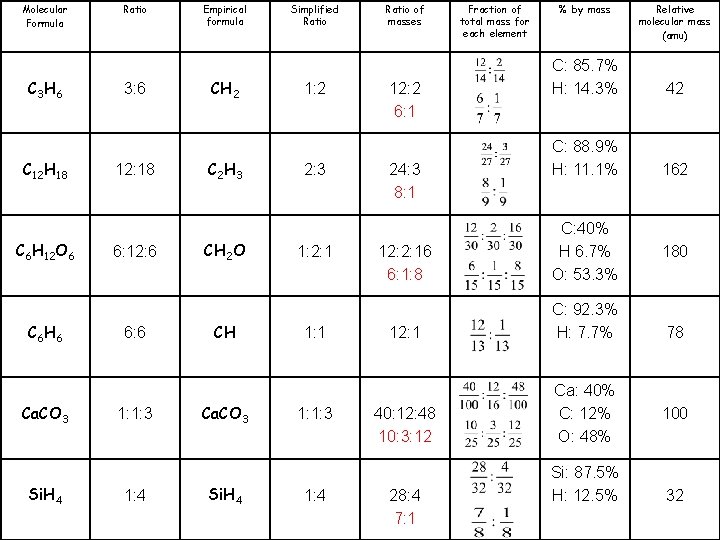
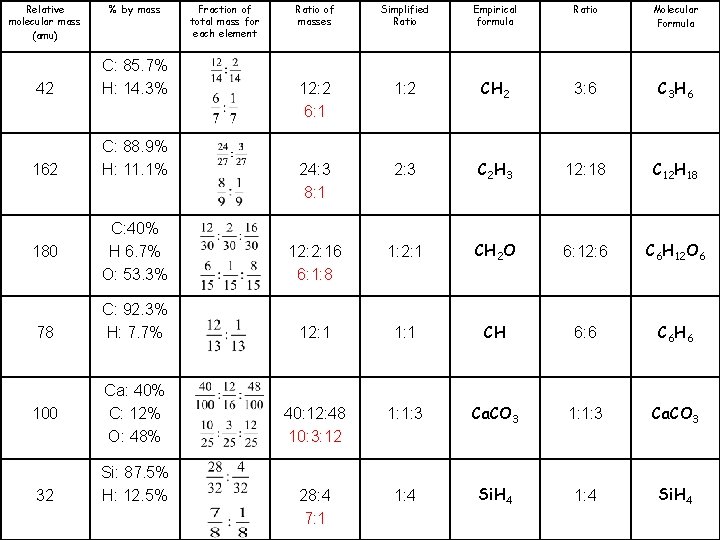
Molecular Formula C 3 H 6 C 12 H 18 C 6 H 12 O 6 C 6 H 6 Ca. CO 3 Si. H 4 Ratio 3: 6 Empirical formula CH 2 Simplified Ratio 1: 2 Ratio of masses 12: 2 Fraction of total mass for each element % by mass C: 85. 7% H: 14. 3% Relative molecular mass (amu) 42

There a few different ways of using this resource: 1. Fill in the gaps in the table (possibly in pairs- each use a different colour) 2. Cut up the completed grid and use it as a card sort (possibly only use one slide rather than two) 3. Print the reversed complete grid with a picture on the back of the page before you cut it out into cards. Put the cards in the correct places on the template grid. Flip over each card to reveal the picture (and hence check the cards are in the correct place.

• Fill in gaps (given formula)

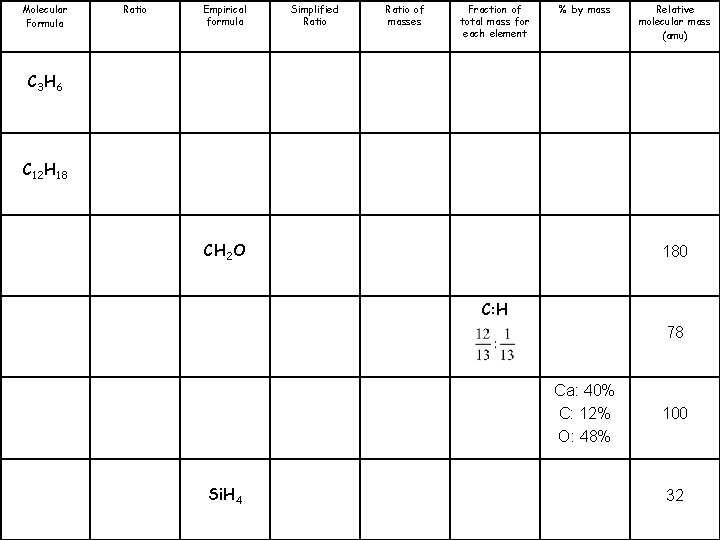
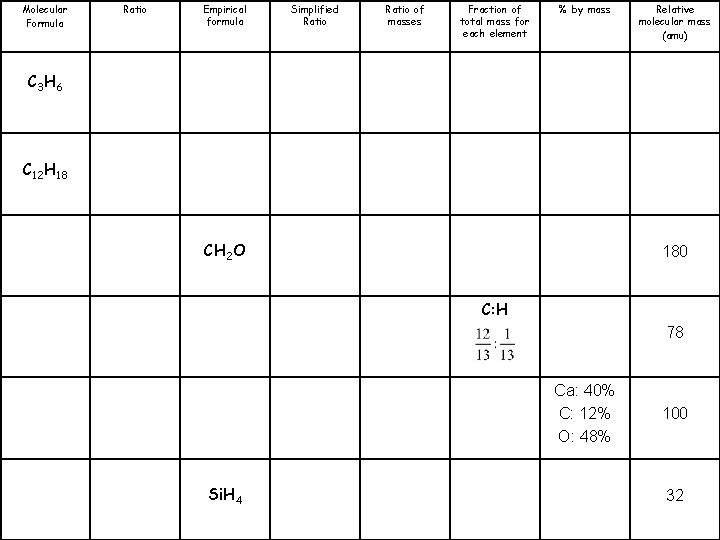
Molecular Formula C 3 H 6 C 12 H 18 C 6 H 12 O 6 C 6 H 6 Ca. CO 3 Si. H 4 Ratio Empirical formula Simplified Ratio of masses Fraction of total mass for each element % by mass Relative molecular mass (amu)

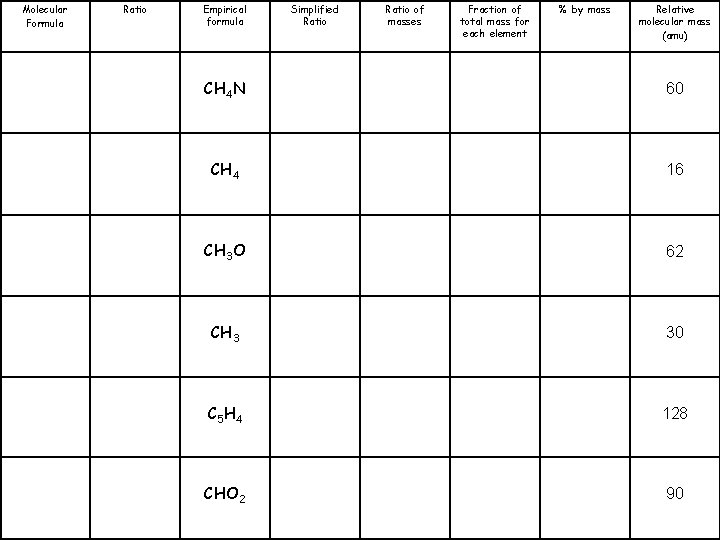
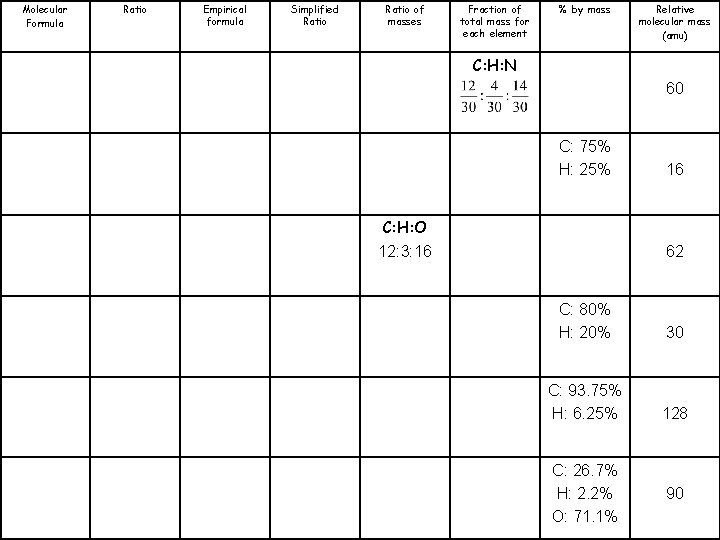
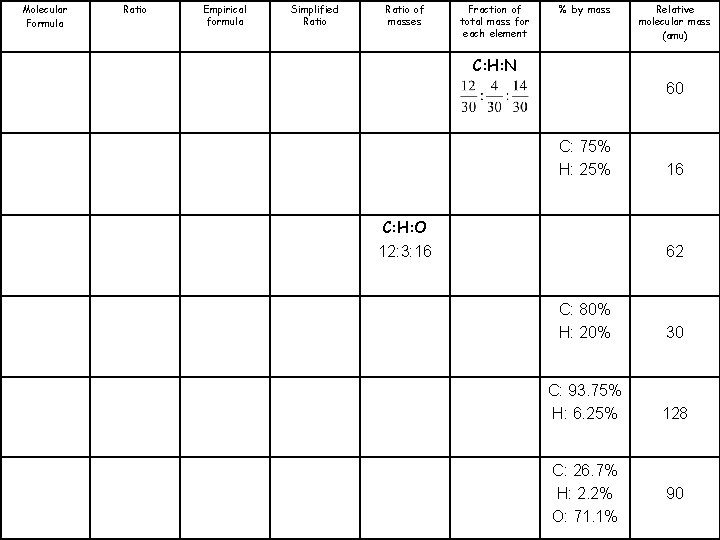
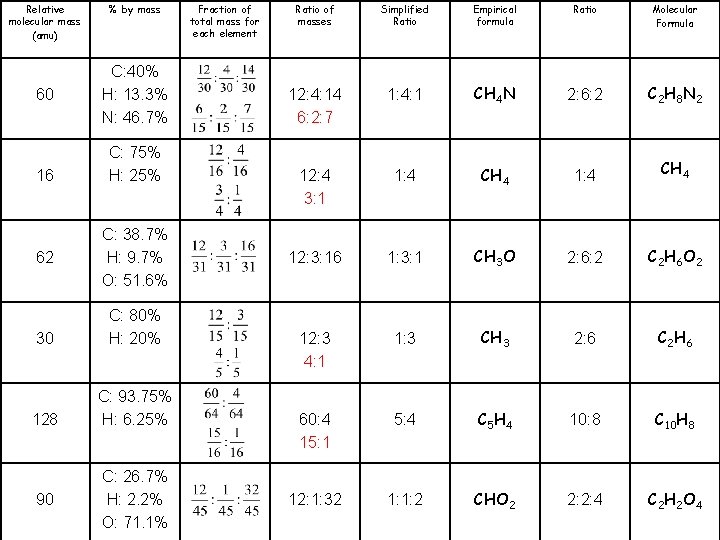
Molecular Formula Ratio Empirical formula Simplified Ratio of masses Fraction of total mass for each element % by mass Relative molecular mass (amu) CH 4 N 60 CH 4 16 CH 3 O 62 CH 3 30 C 5 H 4 128 CHO 2 90

• answers

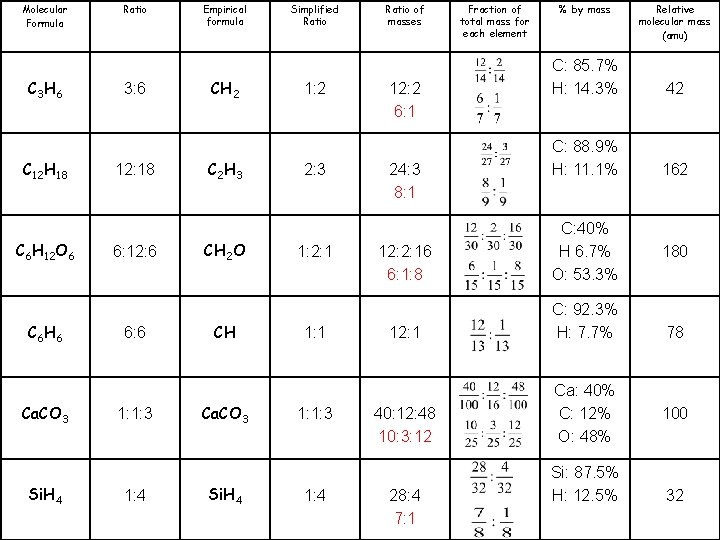
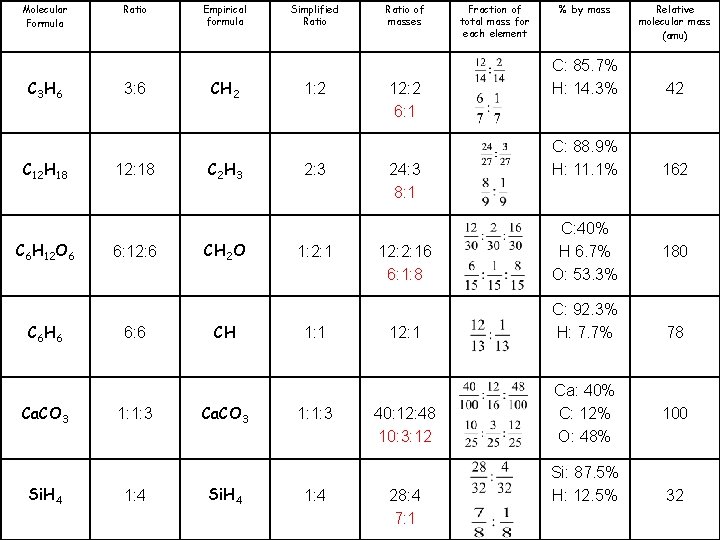
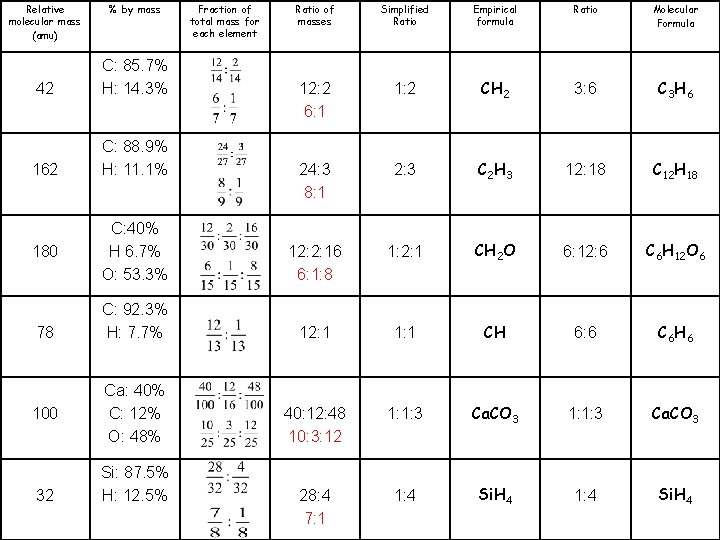
Molecular Formula C 3 H 6 C 12 H 18 C 6 H 12 O 6 C 6 H 6 Ca. CO 3 Si. H 4 Ratio 3: 6 12: 18 6: 12: 6 6: 6 1: 1: 3 1: 4 Empirical formula CH 2 C 2 H 3 CH 2 O CH Ca. CO 3 Si. H 4 Simplified Ratio 1: 2 2: 3 1: 2: 1 1: 1: 3 1: 4 Ratio of masses Fraction of total mass for each element % by mass Relative molecular mass (amu) 12: 2 C: 85. 7% H: 14. 3% 42 24: 3 C: 88. 9% H: 11. 1% 162 12: 2: 16 12: 1 40: 12: 48 28: 4 C: 40% H 6. 7% O: 53. 3% C: 92. 3% H: 7. 7% Ca: 40% C: 12% O: 48% Si: 87. 5% H: 12. 5% 180 78 100 32

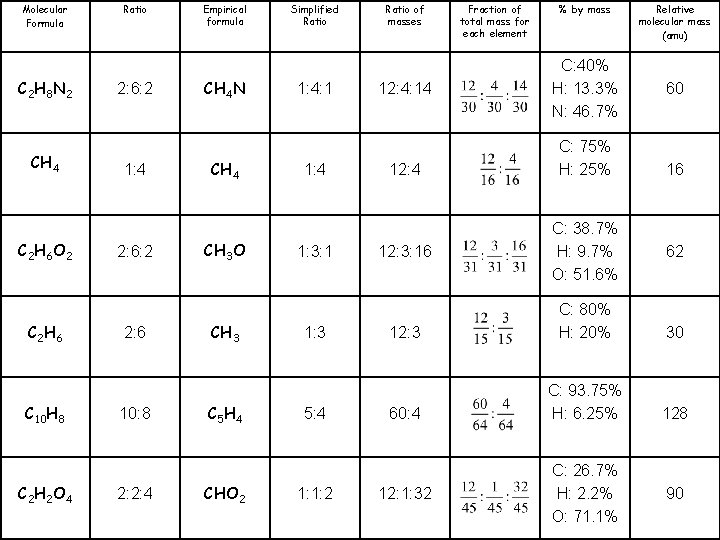
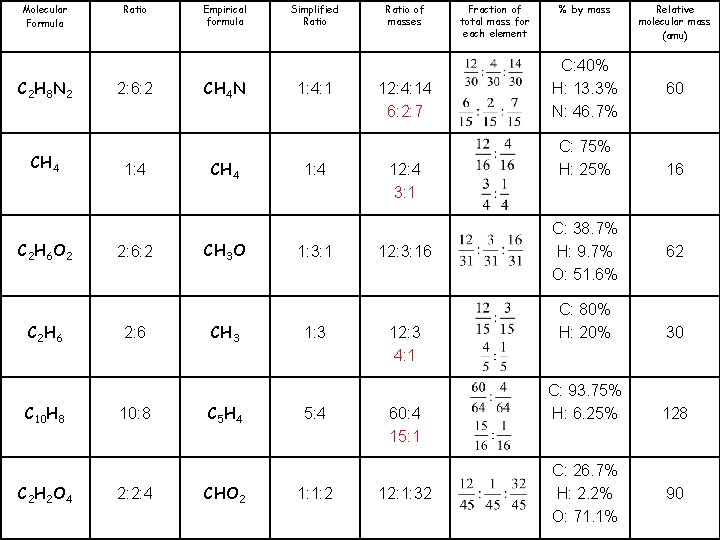
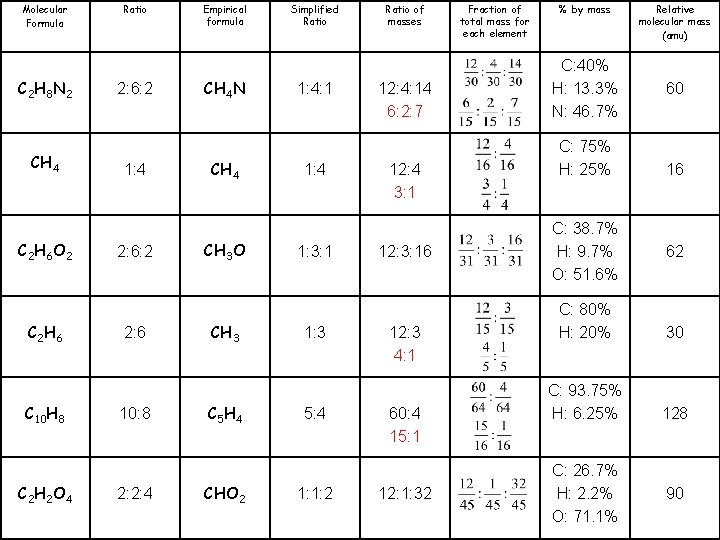
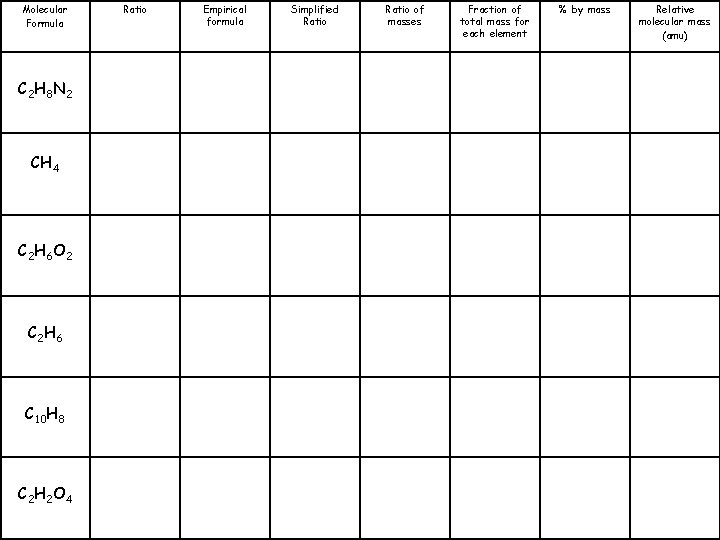
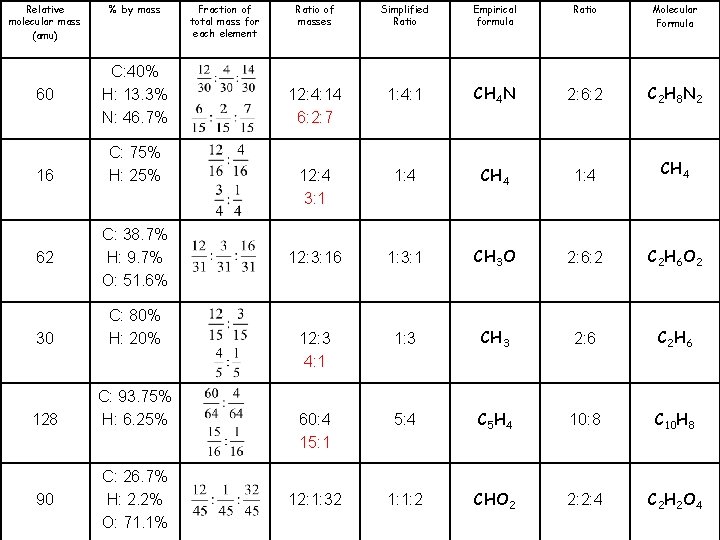
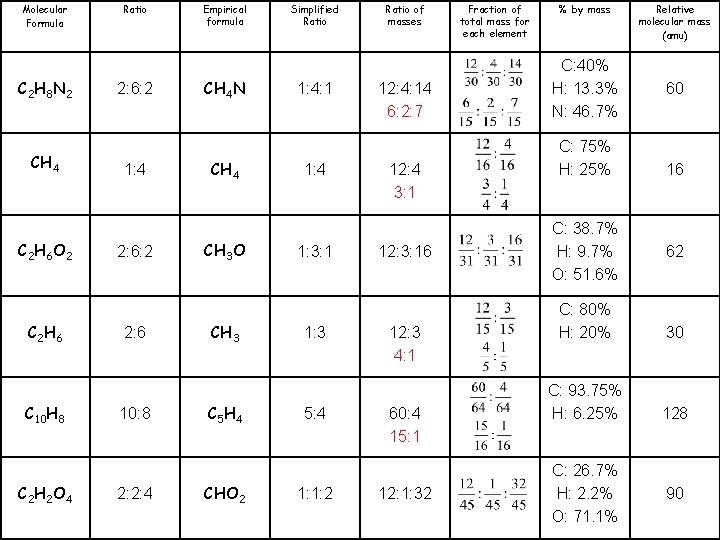
Molecular Formula C 2 H 8 N 2 CH 4 C 2 H 6 O 2 C 2 H 6 C 10 H 8 C 2 H 2 O 4 Ratio 2: 6: 2 1: 4 2: 6: 2 2: 6 10: 8 2: 2: 4 Empirical formula CH 4 N CH 4 CH 3 O CH 3 C 5 H 4 CHO 2 Simplified Ratio 1: 4: 1 1: 4 1: 3: 1 1: 3 5: 4 1: 1: 2 Ratio of masses 12: 4: 14 12: 3: 16 Fraction of total mass for each element % by mass C: 40% H: 13. 3% N: 46. 7% C: 75% H: 25% C: 38. 7% H: 9. 7% O: 51. 6% Relative molecular mass (amu) 60 16 62 12: 3 C: 80% H: 20% 30 60: 4 C: 93. 75% H: 6. 25% 128 12: 1: 32 C: 26. 7% H: 2. 2% O: 71. 1% 90

• Fill in gaps (harder)

Molecular Formula Ratio Empirical formula Simplified Ratio of masses Fraction of total mass for each element % by mass Relative molecular mass (amu) C 3 H 6 C 12 H 18 CH 2 O 180 C: H 78 Ca: 40% C: 12% O: 48% Si. H 4 100 32

Molecular Formula Ratio Empirical formula Simplified Ratio of masses Fraction of total mass for each element % by mass Relative molecular mass (amu) C: H: N 60 C: 75% H: 25% C: H: O 12: 3: 16 16 62 C: 80% H: 20% 30 C: 93. 75% H: 6. 25% 128 C: 26. 7% H: 2. 2% O: 71. 1% 90

• answers

Molecular Formula C 3 H 6 C 12 H 18 C 6 H 12 O 6 C 6 H 6 Ca. CO 3 Si. H 4 Ratio 3: 6 12: 18 6: 12: 6 6: 6 1: 1: 3 1: 4 Empirical formula CH 2 C 2 H 3 CH 2 O CH Ca. CO 3 Si. H 4 Simplified Ratio 1: 2 2: 3 1: 2: 1 1: 1: 3 1: 4 Ratio of masses 12: 2 6: 1 24: 3 8: 1 Fraction of total mass for each element % by mass C: 85. 7% H: 14. 3% 42 C: 88. 9% H: 11. 1% 162 12: 2: 16 6: 1: 8 C: 40% H 6. 7% O: 53. 3% 12: 1 C: 92. 3% H: 7. 7% 40: 12: 48 10: 3: 12 Ca: 40% C: 12% O: 48% 28: 4 7: 1 Relative molecular mass (amu) Si: 87. 5% H: 12. 5% 180 78 100 32

Molecular Formula C 2 H 8 N 2 CH 4 C 2 H 6 O 2 C 2 H 6 C 10 H 8 C 2 H 2 O 4 Ratio 2: 6: 2 1: 4 2: 6: 2 2: 6 10: 8 2: 2: 4 Empirical formula CH 4 N CH 4 CH 3 O CH 3 C 5 H 4 CHO 2 Simplified Ratio 1: 4: 1 1: 4 1: 3: 1 1: 3 5: 4 1: 1: 2 Ratio of masses 12: 4: 14 6: 2: 7 12: 4 3: 1 12: 3: 16 12: 3 4: 1 60: 4 15: 1 12: 1: 32 Fraction of total mass for each element % by mass C: 40% H: 13. 3% N: 46. 7% C: 75% H: 25% C: 38. 7% H: 9. 7% O: 51. 6% Relative molecular mass (amu) 60 16 62 C: 80% H: 20% 30 C: 93. 75% H: 6. 25% 128 C: 26. 7% H: 2. 2% O: 71. 1% 90

• Card sort

Molecular Formula C 3 H 6 C 12 H 18 C 6 H 12 O 6 C 6 H 6 Ca. CO 3 Si. H 4 Ratio 3: 6 12: 18 6: 12: 6 6: 6 1: 1: 3 1: 4 Empirical formula CH 2 C 2 H 3 CH 2 O CH Ca. CO 3 Si. H 4 Simplified Ratio 1: 2 2: 3 1: 2: 1 1: 1: 3 1: 4 Ratio of masses 12: 2 6: 1 24: 3 8: 1 Fraction of total mass for each element % by mass C: 85. 7% H: 14. 3% 42 C: 88. 9% H: 11. 1% 162 12: 2: 16 6: 1: 8 C: 40% H 6. 7% O: 53. 3% 12: 1 C: 92. 3% H: 7. 7% 40: 12: 48 10: 3: 12 Ca: 40% C: 12% O: 48% 28: 4 7: 1 Relative molecular mass (amu) Si: 87. 5% H: 12. 5% 180 78 100 32

Molecular Formula C 2 H 8 N 2 CH 4 C 2 H 6 O 2 C 2 H 6 C 10 H 8 C 2 H 2 O 4 Ratio 2: 6: 2 1: 4 2: 6: 2 2: 6 10: 8 2: 2: 4 Empirical formula CH 4 N CH 4 CH 3 O CH 3 C 5 H 4 CHO 2 Simplified Ratio 1: 4: 1 1: 4 1: 3: 1 1: 3 5: 4 1: 1: 2 Ratio of masses 12: 4: 14 6: 2: 7 12: 4 3: 1 12: 3: 16 12: 3 4: 1 60: 4 15: 1 12: 1: 32 Fraction of total mass for each element % by mass C: 40% H: 13. 3% N: 46. 7% C: 75% H: 25% C: 38. 7% H: 9. 7% O: 51. 6% Relative molecular mass (amu) 60 16 62 C: 80% H: 20% 30 C: 93. 75% H: 6. 25% 128 C: 26. 7% H: 2. 2% O: 71. 1% 90

• Card sort with template (given formula)

Molecular Formula C 3 H 6 C 12 H 18 C 6 H 12 O 6 C 6 H 6 Ca. CO 3 Si. H 4 Ratio Empirical formula Simplified Ratio of masses Fraction of total mass for each element % by mass Relative molecular mass (amu)

Molecular Formula C 2 H 8 N 2 CH 4 C 2 H 6 O 2 C 2 H 6 C 10 H 8 C 2 H 2 O 4 Ratio Empirical formula Simplified Ratio of masses Fraction of total mass for each element % by mass Relative molecular mass (amu)

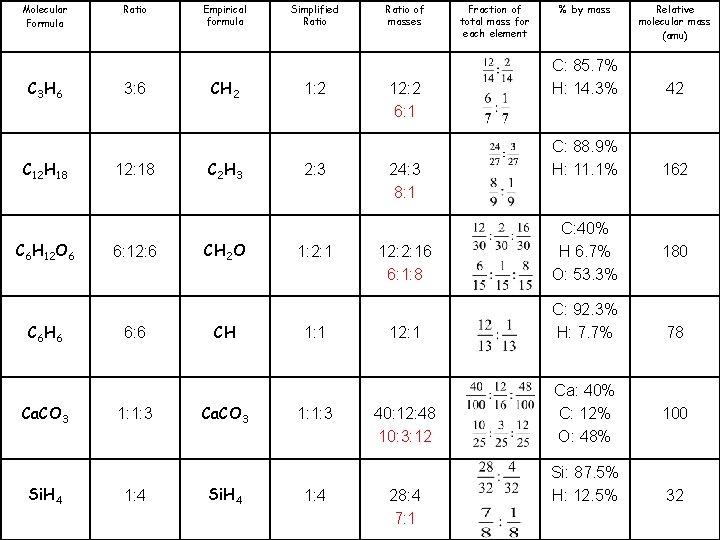
Relative molecular mass (amu) % by mass Ratio of masses Simplified Ratio Empirical formula Ratio Molecular Formula 12: 2 6: 1 1: 2 CH 2 3: 6 C 3 H 6 24: 3 8: 1 2: 3 C 2 H 3 12: 18 C 12 H 18 C: 40% H 6. 7% O: 53. 3% 12: 2: 16 6: 1: 8 1: 2: 1 CH 2 O 6: 12: 6 C 6 H 12 O 6 C: 92. 3% H: 7. 7% 12: 1 1: 1 CH 6: 6 C 6 H 6 Ca: 40% C: 12% O: 48% 40: 12: 48 10: 3: 12 1: 1: 3 Ca. CO 3 28: 4 7: 1 1: 4 Si. H 4 42 C: 85. 7% H: 14. 3% 162 C: 88. 9% H: 11. 1% 180 78 100 32 Si: 87. 5% H: 12. 5% Fraction of total mass for each element

(insert picture of your own choice)

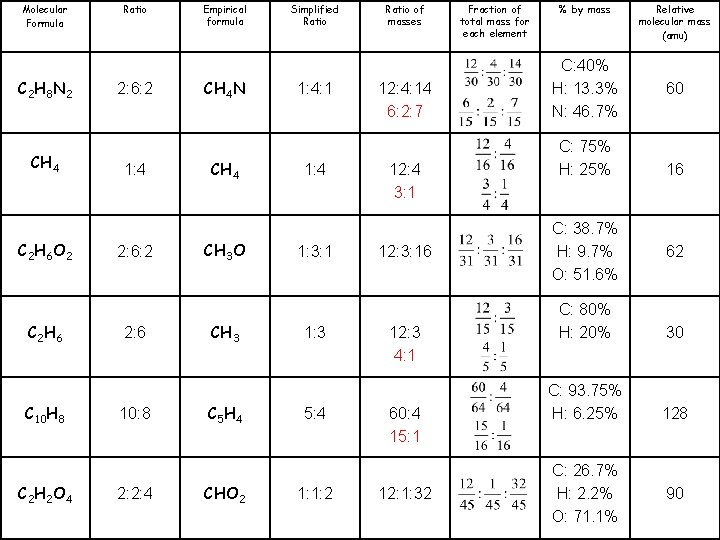
Relative molecular mass (amu) 60 16 62 % by mass C: 40% H: 13. 3% N: 46. 7% C: 75% H: 25% C: 38. 7% H: 9. 7% O: 51. 6% 30 C: 80% H: 20% 128 C: 93. 75% H: 6. 25% 90 C: 26. 7% H: 2. 2% O: 71. 1% Fraction of total mass for each element Ratio of masses Simplified Ratio Empirical formula Ratio Molecular Formula 12: 4: 14 6: 2: 7 1: 4: 1 CH 4 N 2: 6: 2 C 2 H 8 N 2 12: 4 3: 1 1: 4 CH 4 12: 3: 16 1: 3: 1 CH 3 O 2: 6: 2 C 2 H 6 O 2 12: 3 4: 1 1: 3 CH 3 2: 6 C 2 H 6 60: 4 15: 1 5: 4 C 5 H 4 10: 8 C 10 H 8 12: 1: 32 1: 1: 2 CHO 2 2: 2: 4 C 2 H 2 O 4

(insert picture of your own choice)

• Card sort with template (harder)

Molecular Formula Ratio Empirical formula Simplified Ratio of masses Fraction of total mass for each element % by mass Relative molecular mass (amu) C 3 H 6 C 12 H 18 CH 2 O 180 C: H 78 Ca: 40% C: 12% O: 48% Si. H 4 100 32

Molecular Formula Ratio Empirical formula Simplified Ratio of masses Fraction of total mass for each element % by mass Relative molecular mass (amu) C: H: N 60 C: 75% H: 25% C: H: O 12: 3: 16 16 62 C: 80% H: 20% 30 C: 93. 75% H: 6. 25% 128 C: 26. 7% H: 2. 2% O: 71. 1% 90

Molecular Formula C 3 H 6 C 12 H 18 C 6 H 12 O 6 C 6 H 6 Ca. CO 3 Si. H 4 Ratio 3: 6 12: 18 6: 12: 6 6: 6 1: 1: 3 1: 4 Empirical formula CH 2 C 2 H 3 CH 2 O CH Ca. CO 3 Si. H 4 Simplified Ratio 1: 2 2: 3 1: 2: 1 1: 1: 3 1: 4 Ratio of masses 12: 2 6: 1 24: 3 8: 1 Fraction of total mass for each element % by mass C: 85. 7% H: 14. 3% 42 C: 88. 9% H: 11. 1% 162 12: 2: 16 6: 1: 8 C: 40% H 6. 7% O: 53. 3% 12: 1 C: 92. 3% H: 7. 7% 40: 12: 48 10: 3: 12 Ca: 40% C: 12% O: 48% 28: 4 7: 1 Relative molecular mass (amu) Si: 87. 5% H: 12. 5% 180 78 100 32

Molecular Formula C 2 H 8 N 2 CH 4 C 2 H 6 O 2 C 2 H 6 C 10 H 8 C 2 H 2 O 4 Ratio 2: 6: 2 1: 4 2: 6: 2 2: 6 10: 8 2: 2: 4 Empirical formula CH 4 N CH 4 CH 3 O CH 3 C 5 H 4 CHO 2 Simplified Ratio 1: 4: 1 1: 4 1: 3: 1 1: 3 5: 4 1: 1: 2 Ratio of masses 12: 4: 14 6: 2: 7 12: 4 3: 1 12: 3: 16 12: 3 4: 1 60: 4 15: 1 12: 1: 32 Fraction of total mass for each element % by mass C: 40% H: 13. 3% N: 46. 7% C: 75% H: 25% C: 38. 7% H: 9. 7% O: 51. 6% Relative molecular mass (amu) 60 16 62 C: 80% H: 20% 30 C: 93. 75% H: 6. 25% 128 C: 26. 7% H: 2. 2% O: 71. 1% 90

Relative molecular mass (amu) % by mass Ratio of masses Simplified Ratio Empirical formula Ratio Molecular Formula 12: 2 6: 1 1: 2 CH 2 3: 6 C 3 H 6 24: 3 8: 1 2: 3 C 2 H 3 12: 18 C 12 H 18 C: 40% H 6. 7% O: 53. 3% 12: 2: 16 6: 1: 8 1: 2: 1 CH 2 O 6: 12: 6 C 6 H 12 O 6 C: 92. 3% H: 7. 7% 12: 1 1: 1 CH 6: 6 C 6 H 6 Ca: 40% C: 12% O: 48% 40: 12: 48 10: 3: 12 1: 1: 3 Ca. CO 3 28: 4 7: 1 1: 4 Si. H 4 42 C: 85. 7% H: 14. 3% 162 C: 88. 9% H: 11. 1% 180 78 100 32 Si: 87. 5% H: 12. 5% Fraction of total mass for each element

(insert picture of your own choice)

Relative molecular mass (amu) 60 16 62 % by mass C: 40% H: 13. 3% N: 46. 7% C: 75% H: 25% C: 38. 7% H: 9. 7% O: 51. 6% 30 C: 80% H: 20% 128 C: 93. 75% H: 6. 25% 90 C: 26. 7% H: 2. 2% O: 71. 1% Fraction of total mass for each element Ratio of masses Simplified Ratio Empirical formula Ratio Molecular Formula 12: 4: 14 6: 2: 7 1: 4: 1 CH 4 N 2: 6: 2 C 2 H 8 N 2 12: 4 3: 1 1: 4 CH 4 12: 3: 16 1: 3: 1 CH 3 O 2: 6: 2 C 2 H 6 O 2 12: 3 4: 1 1: 3 CH 3 2: 6 C 2 H 6 60: 4 15: 1 5: 4 C 5 H 4 10: 8 C 10 H 8 12: 1: 32 1: 1: 2 CHO 2 2: 2: 4 C 2 H 2 O 4

(insert picture of your own choice)


Molecular Formula C 3 H 6 C 12 H 18 C 6 H 12 O 6 C 6 H 6 Ca. CO 3 Si. H 4 Ratio 3: 6 12: 18 6: 12: 6 6: 6 1: 1: 3 1: 4 Empirical formula CH 2 C 2 H 3 CH 2 O CH Ca. CO 3 Si. H 4 Simplified Ratio 1: 2 2: 3 1: 2: 1 1: 1: 3 1: 4 Ratio of masses 12: 2 6: 1 24: 3 8: 1 Fraction of total mass for each element % by mass C: 85. 7% H: 14. 3% 42 C: 88. 9% H: 11. 1% 162 12: 2: 16 6: 1: 8 C: 40% H 6. 7% O: 53. 3% 12: 1 C: 92. 3% H: 7. 7% 40: 12: 48 10: 3: 12 Ca: 40% C: 12% O: 48% 28: 4 7: 1 Relative molecular mass (amu) Si: 87. 5% H: 12. 5% 180 78 100 32

Molecular Formula C 2 H 8 N 2 CH 4 C 2 H 6 O 2 C 2 H 6 C 10 H 8 C 2 H 2 O 4 Ratio 2: 6: 2 1: 4 2: 6: 2 2: 6 10: 8 2: 2: 4 Empirical formula CH 4 N CH 4 CH 3 O CH 3 C 5 H 4 CHO 2 Simplified Ratio 1: 4: 1 1: 4 1: 3: 1 1: 3 5: 4 1: 1: 2 Ratio of masses 12: 4: 14 6: 2: 7 12: 4 3: 1 12: 3: 16 12: 3 4: 1 60: 4 15: 1 12: 1: 32 Fraction of total mass for each element % by mass C: 40% H: 13. 3% N: 46. 7% C: 75% H: 25% C: 38. 7% H: 9. 7% O: 51. 6% Relative molecular mass (amu) 60 16 62 C: 80% H: 20% 30 C: 93. 75% H: 6. 25% 128 C: 26. 7% H: 2. 2% O: 71. 1% 90

Alternative ratio activity

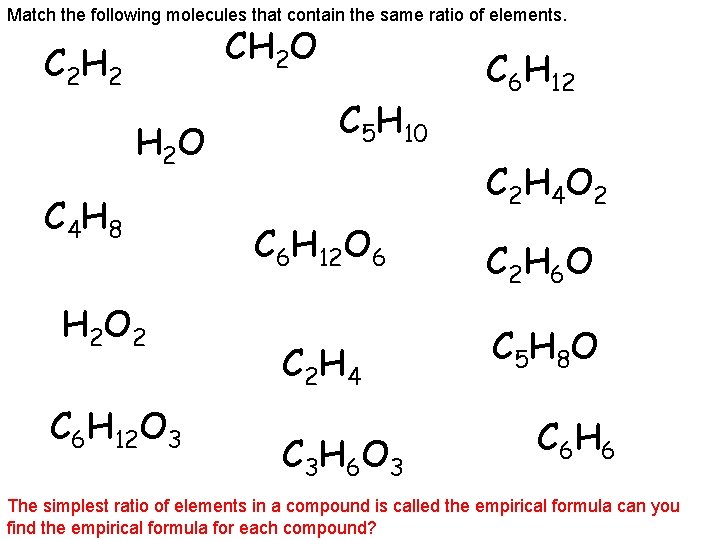
Match the following molecules that contain the same ratio of elements. CH 2 O C 2 H 2 O C 4 H 8 H 2 O 2 C 6 H 12 O 3 C 5 H 10 C 6 H 12 C 2 H 4 O 2 C 6 H 12 O 6 C 2 H 6 O C 2 H 4 C 5 H 8 O C 3 H 6 O 3 C 6 H 6 The simplest ratio of elements in a compound is called the empirical formula can you find the empirical formula for each compound?

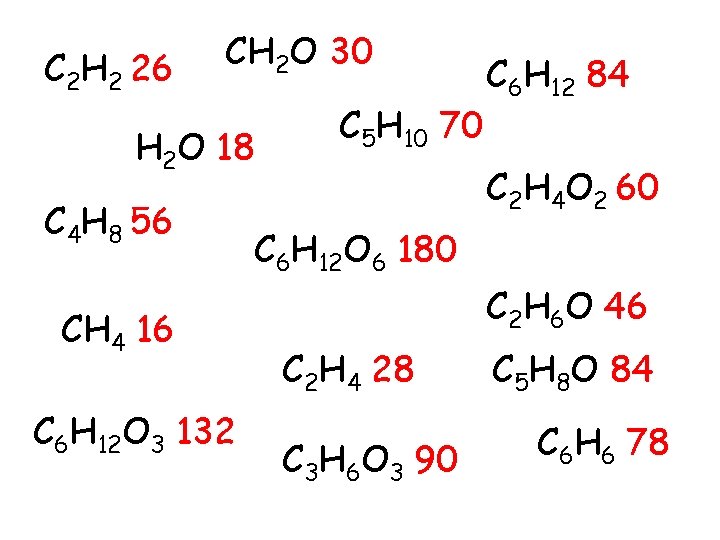
Extension • The mass of each of the compound is shown on the next slide. • Can you work out the mass of the following atoms H (hydrogen) C (carbon O (oxygen)

C 2 H 2 26 CH 2 O 30 H 2 O 18 C 4 H 8 56 CH 4 16 C 6 H 12 O 3 132 C 5 H 10 70 C 6 H 12 84 C 2 H 4 O 2 60 C 6 H 12 O 6 180 C 2 H 6 O 46 C 2 H 4 28 C 3 H 6 O 3 90 C 5 H 8 O 84 C 6 H 6 78