MOLECULAR FARMING INTRODUCTION Biotechnology in agriculture has two

MOLECULAR FARMING

INTRODUCTION Biotechnology in agriculture has two categories: 1. “Improvements” to existing livestock and crops, and 2. Development of entirely new uses for both animals and plants (biopharming). “Improvements", include ‘input traits’ such as crops with extra resistance to insect attack, improved weed control, increase the plants tolerance to cold, drought and other environmental factor. Example: "Roundup ready" soya "Starlite" corn "Frost-tolerant" tomatoes

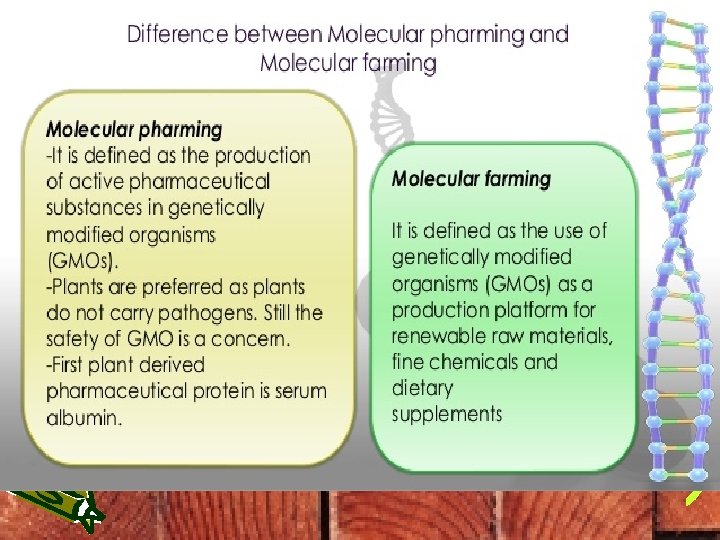
• Plant molecular farming (PMF) is a new branch of plant biotechnology, where plants are engineered to produce recombinant pharmaceutical and industrial proteins in large quantities. • As an emerging subdivision of the biopharmaceutical industry, PMF is still trying to gain comparable social acceptance as the already established production systems that produce these high valued proteins in microbial, yeast or mammalian expression systems.

• Plant molecular farming (PMF) refers to the production of recombinant proteins (including pharmaceuticals and industrial proteins) and other secondary metabolites, in plants. • The first recombinant plant-derived pharmaceutical protein (the human growth hormone) and the first recombinant antibody (expressed in the progeny of the cross of two individual transgenic plants expressingle immunoglobulin gamma and kappa chains) were produced in transgenic plants in 1986 and 1989, respectively.

MOLECULAR FARMING • Molecular farming is a method used to integrate a foreign gene into plants. • Molecular farming is the term for new use plants only (not animals ) and is different in that this does not affect and has nothing to do with Food. • There are two types of Molecular Farming: Medical and non-Medical.

Medical Molecular Farming • The first synthesis of a pharmaceutically-relevant protein, human growth hormone, was described in transgenic tobacco plants in 1986. • Now, molecular farming has become commercially interesting as a method for the production of recombinant pharmaceutical proteins, in particular antibodies.

Non-Medical Molecular Farming includes Industrial Enzymes and Polymers. • Industrial enzymes: for example laccase in transgenic maize, • Technical proteins for research purposes: for example avidin, which is also produced in maize, • Milk proteins such as human beta casein, which is produced in transgenic tomatoes, • Protein polymers: collagens, which are used for medical as well as industrial purposes.

Next… • Potentially the biggest development in this field could be the development of plants growing biodegradable plastics. • Other uses could be as Industrial oils such as hydraulic oil or highg yielding biodiesels, new solid Biofuels, new Fibres and Papers, and as agents for Bioremediation and Phytoremediation, environmentally cleaning up contamination.

Types of Molecular Farming
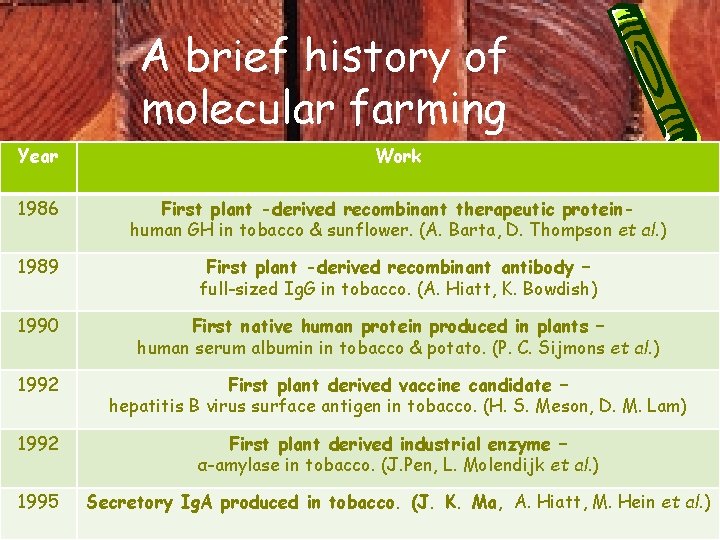
A brief history of molecular farming Year Work 1986 First plant -derived recombinant therapeutic proteinhuman GH in tobacco & sunflower. (A. Barta, D. Thompson et al. ) 1989 First plant -derived recombinant antibody – full-sized Ig. G in tobacco. (A. Hiatt, K. Bowdish) 1990 First native human protein produced in plants – human serum albumin in tobacco & potato. (P. C. Sijmons et al. ) 1992 First plant derived vaccine candidate – hepatitis B virus surface antigen in tobacco. (H. S. Meson, D. M. Lam) 1992 First plant derived industrial enzyme – α-amylase in tobacco. (J. Pen, L. Molendijk et al. ) 1995 Secretory Ig. A produced in tobacco. (J. K. Ma, A. Hiatt, M. Hein et al. )

YEAR WORK 1996 First plant derived protein polymer – artificial elastin in tobacco. (X. Zhang, D. W. Urry, H. Daniel) 1997 First clinical trial using recombinant bacterial antigen delivered in a transgenic potato. (C. O. Tacket et al. ) 1997 Commercial production of avidin in maize. (E. E. Hood et al. ) 2000 Human GH produced in tobacco chloroplast. (J. M. Staub et al. ) 2003 Expression and assembly of a functional antibody in algae. (S. P. Mayfield, S. E. Franklin et al. ) 2003 Commercial production of bovine trypsin in maize.

WHY PLANTS? • Plants are also very flexible and can produce a wide variety of proteins. • Crop plants can synthesize a wide variety of proteins that are free of mammalian toxins and pathogens. • Crop plants produce large amounts of biomass at low cost and require limited facilities. • Crops are therefore well suited for the production of safe low-cost therapeutic proteins.
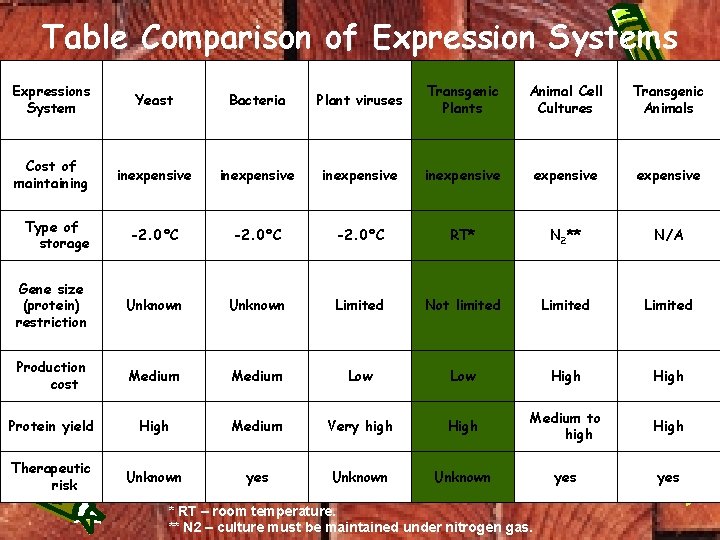
Table Comparison of Expression Systems Expressions System Yeast Bacteria Plant viruses Transgenic Plants Animal Cell Cultures Transgenic Animals Cost of maintaining inexpensive expensive Type of storage -2. 0°C RT* N 2** N/A Gene size (protein) restriction Unknown Limited Not limited Limited Production cost Medium Low High Protein yield High Medium Very high High Medium to high High Therapeutic risk Unknown yes * RT – room temperature. ** N 2 – culture must be maintained under nitrogen gas.

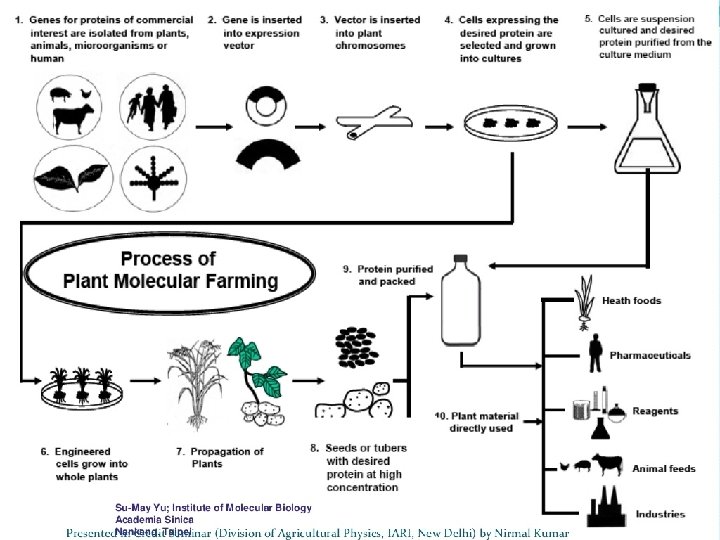
PMF STRATERGY • • • Clone a gene of interest Transform the host platform species Grow the host species, recover biomass Process biomass Purify product of interest Deliver product of interest


HOW IS IT DONE? • Genetic enhancement is used to introduce and express genes coding for the high value proteins. • This involves the growing, harvesting, transport, storage, and downstream processing of extraction and purification of the protein.

PRODUCTION OF BRYODIN IN TOBACCO PLANT • Tobacco plants that are able to produce bryodin. • This protein, which is produced in the roots of bryonia, deactivates ribosomes and is being tested for its effect against HIV infection.

Expression systems for PMF 1. 2. 3. 4. 5. Transgenic plants Plant -cell -suspension culture Transplastomic plants Transient expression system Hydroponic cultures

1. Transgenic plants – Foreign DNA incorporated into the nuclear genome using. Agrobacterium tumefaciens – Particle bombardment • • • Most common Long term non-refrigerated storage Scalability More ‘gene to protein’ time Biosafety concerns

2. Plant cell suspension culture • Culture derived from transgenic explants • Transformation after desegregation • Recombinant protein localization depends on. Presence of targeting / leader peptides in the recombinant protein • Permeability of plant cell wall for macromolecules • Containment & production under GMP procedure • Low scale up capacity

3. Transplastomic plants: • • DNA introduced into chloroplast genome High transgene copy number No gene silencing Recombinant protein accumulate in chloroplast Natural transgene containment Long term storage not possible Limitations: Long development time, Limited use for production of therapeutic glycoproteins.

4. Transient expression system Approaches: – Biolistic delivery of ‘naked DNA’Usually reaches only a few cells. Can be used for a rapid test for protein expression – Agroinfiltration: Delivery of Agrobacteriumin intact leaf tissue by vacuum infiltration – Targets many more cells in a leaf – Infection with modified viral vectors

5. Hydroponic culture • A signal peptide is attached to the recombinant protein directing it to the secretory pathway • Protein can be recovered from the root exudates (Rhizosecretion) or leaf guttation fluid (Phylosecretion) • Technology being developed by the US biotechnology company Phytomedics Inc. • Purification is easier • Reduced fear of unintentional environmental release • Expensive to operate hydroponic facilities




THANKS
- Slides: 30