Molecular Dynamics of ACh BP Water in the

Molecular Dynamics of ACh. BP: Water in the Binding Pocket Shiva Amiri http: //sbcb. bioch. ox. ac. uk/amiri. php Biophysical Society Annual Meeting, February, 2006
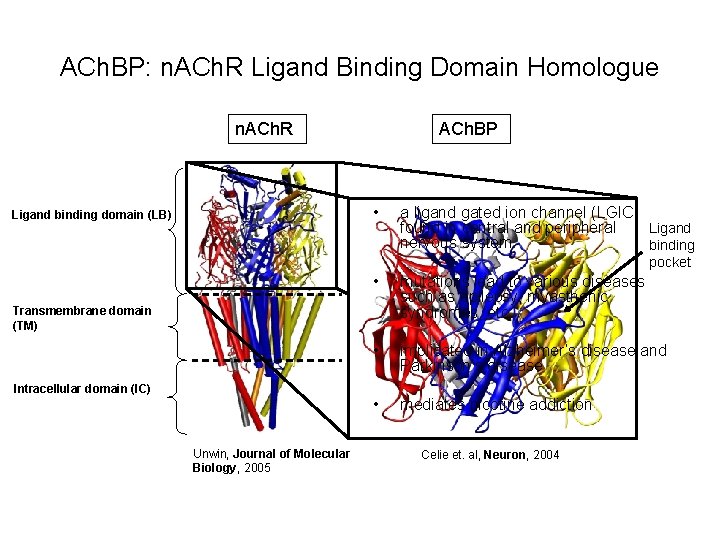
ACh. BP: n. ACh. R Ligand Binding Domain Homologue n. ACh. R ACh. BP • Ligand binding domain (LB) a ligand gated ion channel (LGIC) found in central and peripheral Ligand nervous system binding pocket • mutations lead to various diseases such as epilepsy, myasthenic syndromes, etc. • implicated in Alzheimer’s disease and Parkinson’s disease • mediates nicotine addiction Transmembrane domain (TM) Intracellular domain (IC) Unwin, Journal of Molecular Biology, 2005 Celie et. al, Neuron, 2004
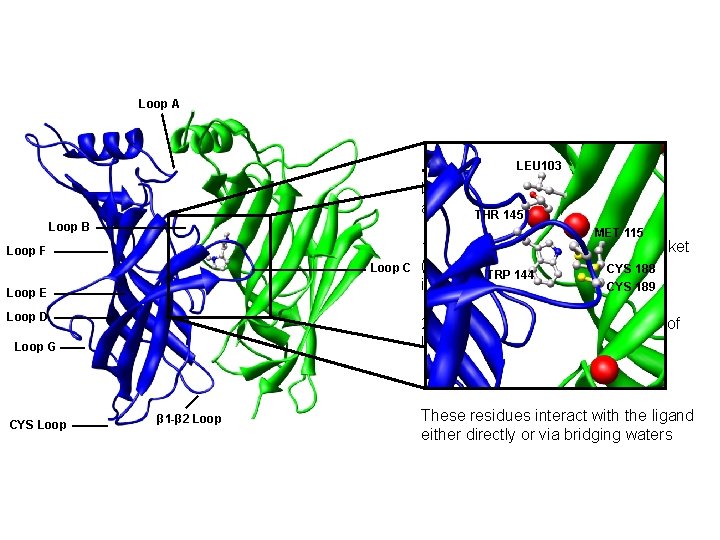
Loop A • Studying the. LEU 103 behaviour of the binding pocket in the presence and absence THR 145 of ligands Loop B MET 115 Loop F Loop C Loop E Loop D 2. The role of water in the binding of ligand to the binding site Loop G CYS Loop 1. The structure of the binding pocket (distances, dihedrals, structural CYS 188 TRP 144 integrity) CYS 189 β 1 -β 2 Loop These residues interact with the ligand either directly or via bridging waters
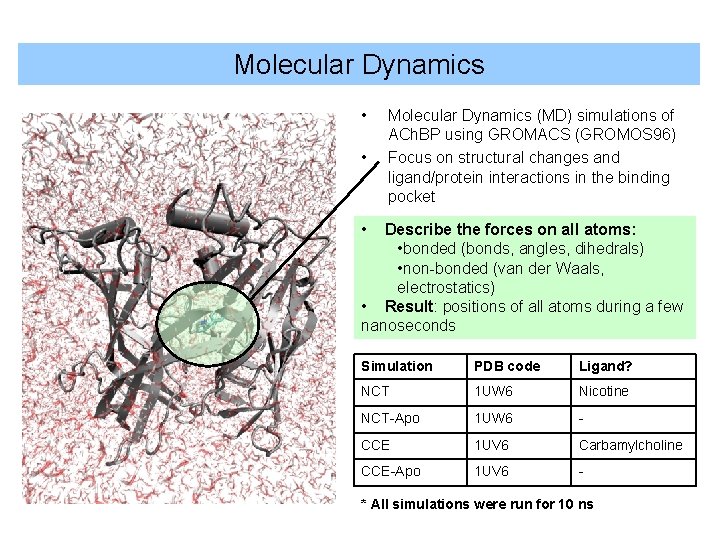
Molecular Dynamics • • Molecular Dynamics (MD) simulations of ACh. BP using GROMACS (GROMOS 96) Focus on structural changes and ligand/protein interactions in the binding pocket • Describe the forces on all atoms: • bonded (bonds, angles, dihedrals) • non-bonded (van der Waals, electrostatics) • Result: positions of all atoms during a few nanoseconds Simulation PDB code Ligand? NCT 1 UW 6 Nicotine NCT-Apo 1 UW 6 - CCE 1 UV 6 Carbamylcholine CCE-Apo 1 UV 6 - * All simulations were run for 10 ns
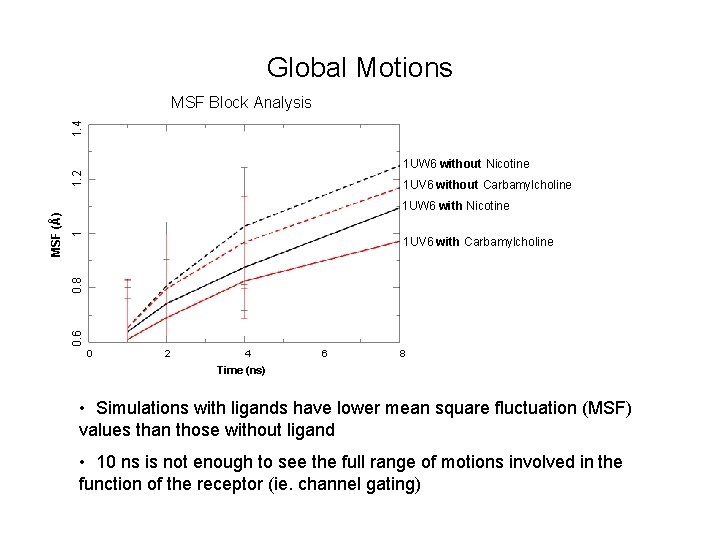
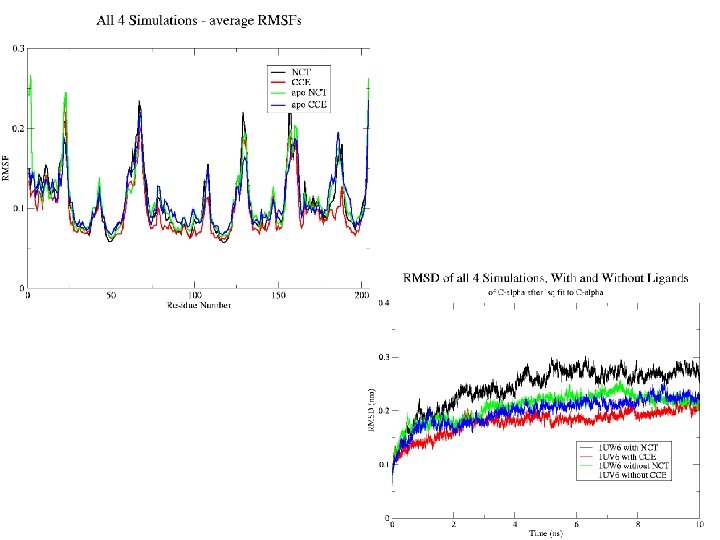
Global Motions 1. 4 MSF Block Analysis 1 UV 6 without Carbamylcholine 1 1 UW 6 with Nicotine 0. 8 1 UV 6 with Carbamylcholine 0. 6 MSF (Å) 1. 2 1 UW 6 without Nicotine 0 2 4 6 8 Time (ns) • Simulations with ligands have lower mean square fluctuation (MSF) values than those without ligand • 10 ns is not enough to see the full range of motions involved in the function of the receptor (ie. channel gating)
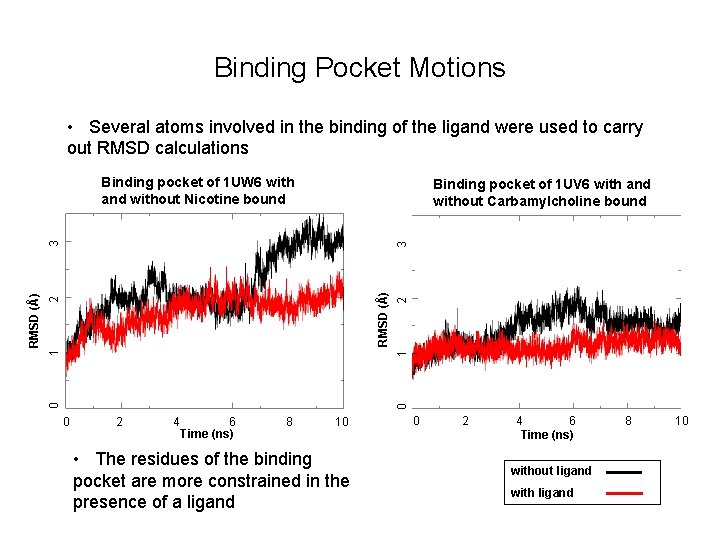
Binding Pocket Motions • Several atoms involved in the binding of the ligand were used to carry out RMSD calculations Binding pocket of 1 UW 6 with and without Nicotine bound 3 1 2 RMSD (Å) 2 1 0 0 RMSD (Å) 3 Binding pocket of 1 UV 6 with and without Carbamylcholine bound 0 2 4 6 Time (ns) 8 10 • The residues of the binding pocket are more constrained in the presence of a ligand 0 2 4 6 Time (ns) without ligand with ligand 8 10
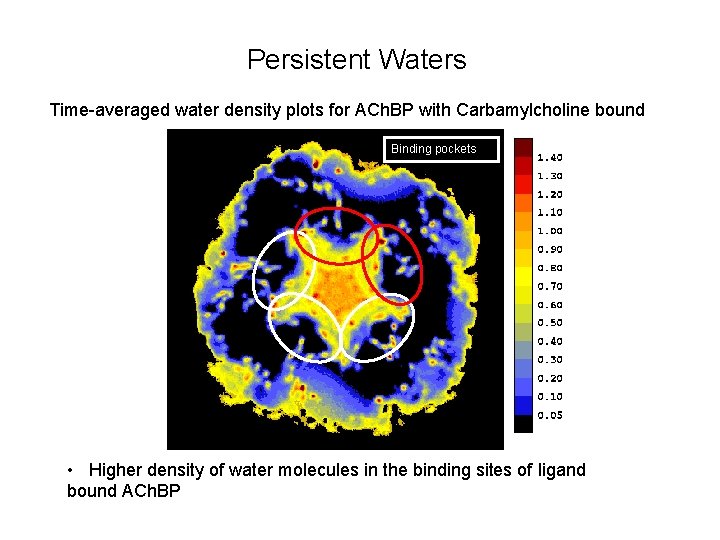
Persistent Waters Time-averaged water density plots for ACh. BP with Carbamylcholine bound Binding pockets • Higher density of water molecules in the binding sites of ligand bound ACh. BP
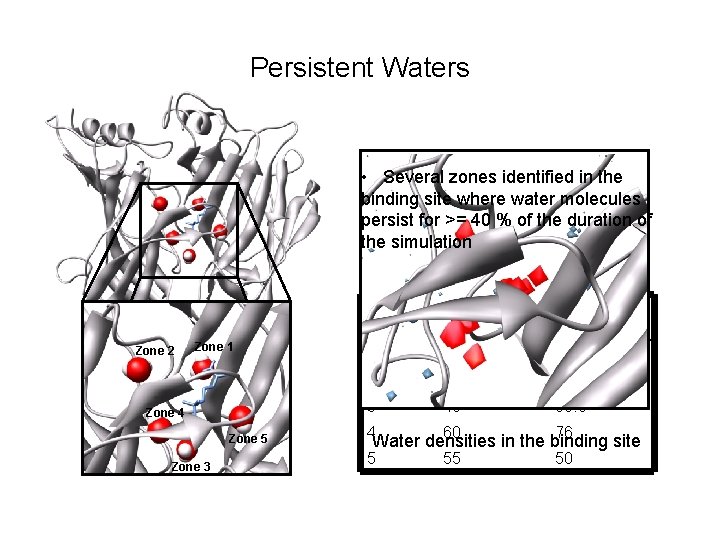
Persistent Waters • Several zones identified in the binding site where water molecules persist for >= 40 % of the duration of the simulation Zone 2 Zone 1 Zone 4 Zone 5 Zone 3 ZONE Average % for NCT Average % for CCE 1 92 92. 5 2 45 79. 5 3 40 89. 5 4 60 76 5 55 50 Water densities in the binding site

Water molecules which remain in their position in the binding pocket with Nicotine bound

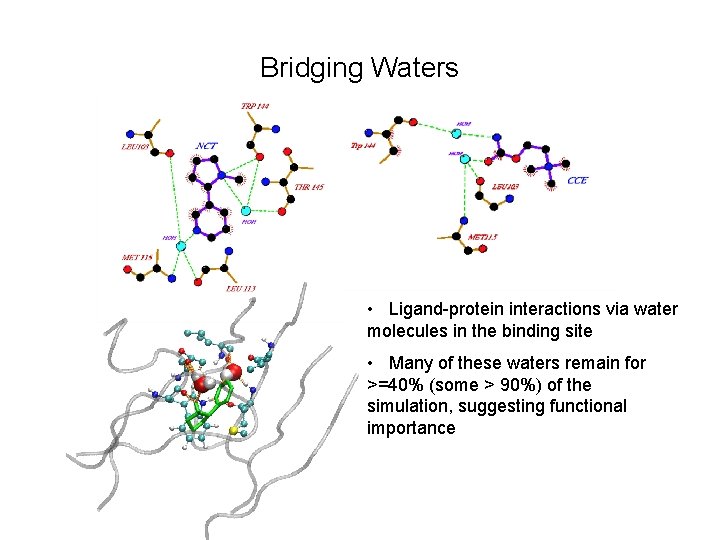
Bridging Waters • Ligand-protein interactions via water molecules in the binding site • Many of these waters remain for >=40% (some > 90%) of the simulation, suggesting functional importance
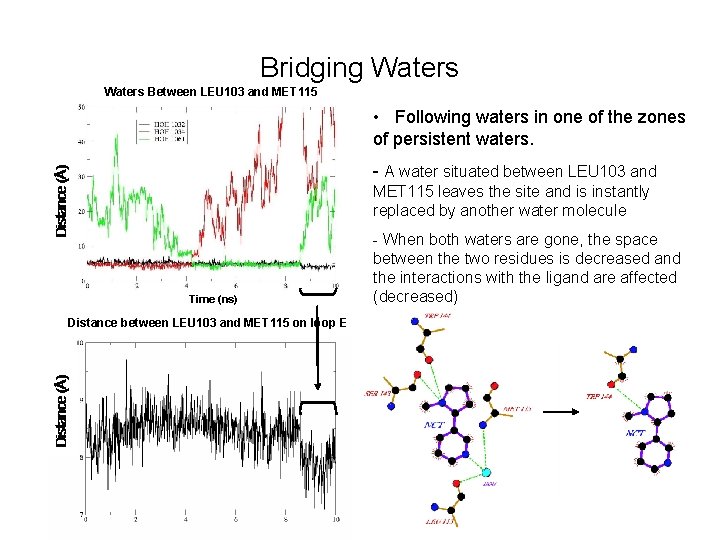
Bridging Waters Between LEU 103 and MET 115 • Following waters in one of the zones of persistent waters. - A water situated between LEU 103 and MET 115 leaves the site and is instantly replaced by another water molecule Time (ns) Distance between LEU 103 and MET 115 on loop E Time (ns) - When both waters are gone, the space between the two residues is decreased and the interactions with the ligand are affected (decreased)

Conclusions • ACh. BP has greater global flexibility in the non-ligand bound state - the binding of a ligand adds structural integrity to the ion channel • The binding pocket is less flexible in the presence of a ligand • There are positionally conserved waters in the binding pocket, higher in quantity and more persistent in the presence of a ligand • Several water molecules bridge the ligand to neighbouring residues in the binding site • These waters plays a structural role in the binding pocket, adding rigidity that may extend beyond the binding site to functionally relevant loops

Acknowledgements • • Prof. Mark S. P. Sansom Dr. Philip C. Biggin • • • • • Dr. Alessandro Grottesi Dr. Kaihsu Tai Dr. Zara Sands Dr. Oliver Beckstein Dr. Jorge Pikunic Dr. Andy Hung Dr. Shozeb Haider Dr. Syma Khalid Dr. Pete Bond Dr. Kia Balali-Mood Dr. Hiunji Kim Dr. Martin Ulmschneider Dr. Daniele Bemporad Dr. Bing Wu Sundeep Deol Yalini Pathy Jonathan Cuthbertson former members • • • • Jennifer Johnston Katherine Cox Robert D’Rozario Jeff Campbell Loredana Vaccaro John Holyoake Tony Ivetac Samantha Kaye Sylvanna Ho Benjamin Hall Tim Carpenter Emi Psachoulia Chze Ling Wee Ranjit Vijayan Michael Kohl
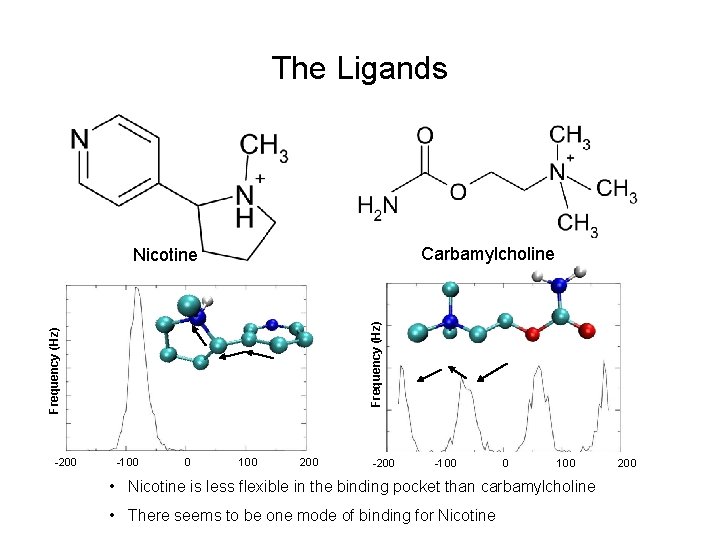
The Ligands Carbamylcholine Frequency (Hz) Nicotine -200 -100 0 100 • Nicotine is less flexible in the binding pocket than carbamylcholine • There seems to be one mode of binding for Nicotine 200
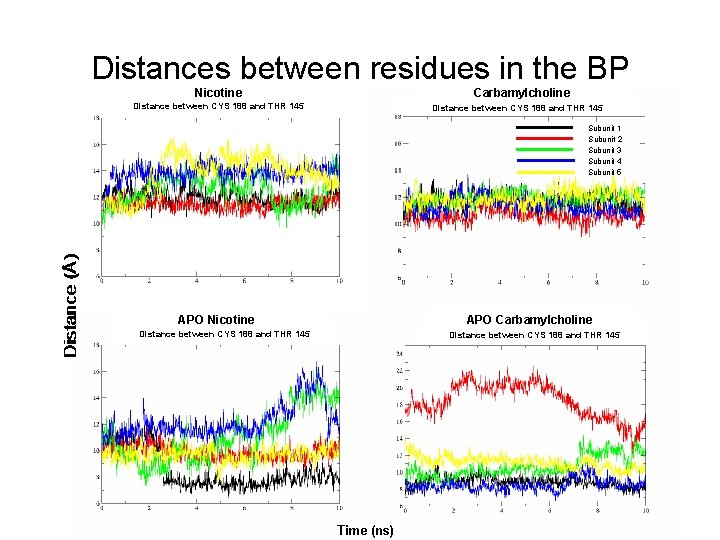
Distances between residues in the BP Nicotine Carbamylcholine Distance between CYS 188 and THR 145 Subunit 1 Subunit 2 Subunit 3 Subunit 4 Subunit 5 APO Nicotine APO Carbamylcholine Distance between CYS 188 and THR 145 Time (ns)
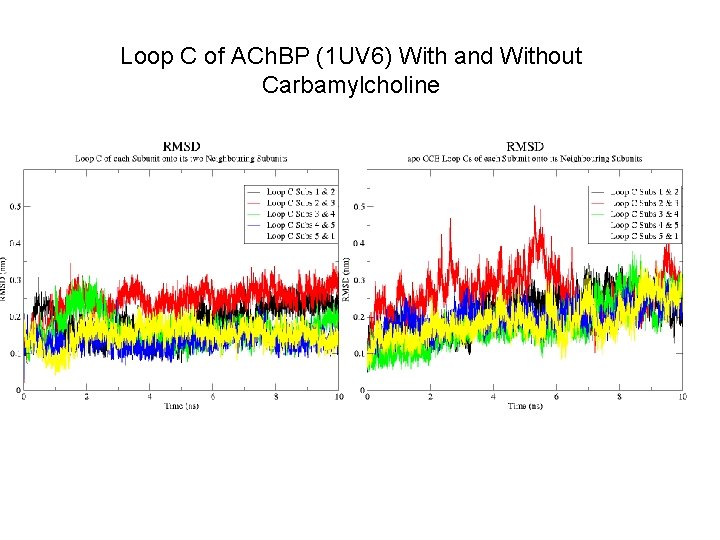
Loop C of ACh. BP (1 UV 6) With and Without Carbamylcholine
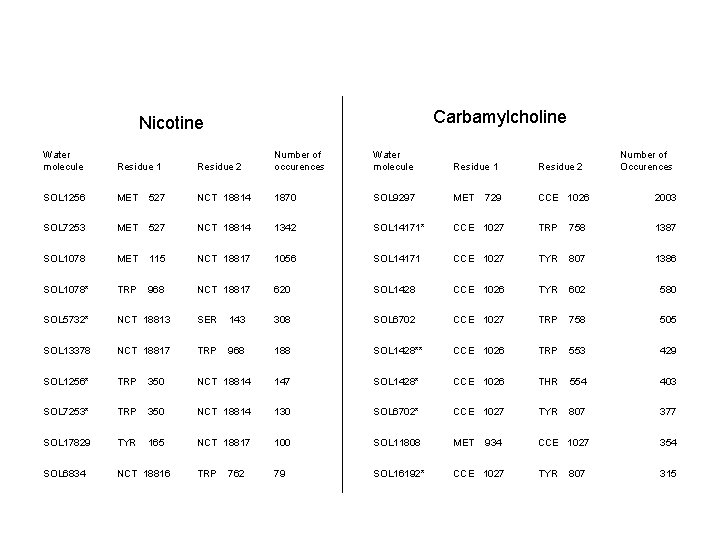
Carbamylcholine Nicotine Water molecule Residue 1 Residue 2 Number of occurences Water molecule Residue 1 Residue 2 SOL 1256 MET 527 NCT 18814 1870 SOL 9297 MET CCE 1026 2003 SOL 7253 MET 527 NCT 18814 1342 SOL 14171* CCE 1027 TRP 758 1387 SOL 1078 MET 115 NCT 18817 1056 SOL 14171 CCE 1027 TYR 807 1386 SOL 1078* TRP 968 NCT 18817 620 SOL 1428 CCE 1026 TYR 602 580 SOL 5732* NCT 18813 SER 143 308 SOL 6702 CCE 1027 TRP 758 505 SOL 13378 NCT 18817 TRP 968 188 SOL 1428** CCE 1026 TRP 553 429 SOL 1256* TRP 350 NCT 18814 147 SOL 1428* CCE 1026 THR 554 403 SOL 7253* TRP 350 NCT 18814 130 SOL 6702* CCE 1027 TYR 807 377 SOL 17829 TYR 165 NCT 18817 100 SOL 11808 MET CCE 1027 354 SOL 6834 NCT 18816 TRP 79 SOL 16192* CCE 1027 TYR 315 762 729 934 807 Number of Occurences
- Slides: 19