Molecular dynamics investigation to identify Al atomic behavior

Molecular dynamics investigation to identify Al atomic behavior on various Cu surfaces 2007. 2. 6 김상필1, 2, 김영근 3, M. Sahashi 4, 이광렬 1, 정용재 2 1한국과학기술연구원 계산과학센터, 2한양대학교 신소재공학부 3고려대학교 신소재공학부, 4 Dept. of Electronic Eng. , Tohoku University
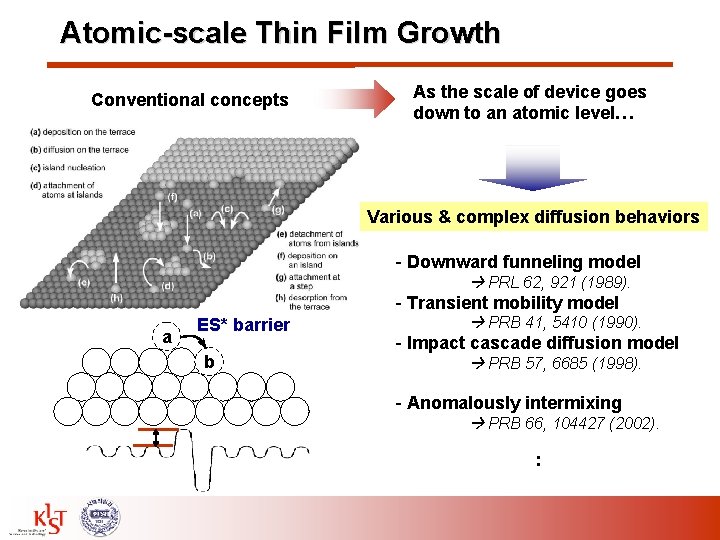
Atomic-scale Thin Film Growth Conventional concepts As the scale of device goes down to an atomic level… Various & complex diffusion behaviors - Downward funneling model PRL 62, 921 (1989). - Transient mobility model a ES* barrier b PRB 41, 5410 (1990). - Impact cascade diffusion model PRB 57, 6685 (1998). - Anomalously intermixing PRB 66, 104427 (2002). :

Research Strategy Atomic scale deposition behavior Morphology & Diffusion path Fundamental understanding Surface diffusion barrier calculation Using Molecular Dynamics & Statics method

EAM Potentials for Al-Cu System* “Simple analytic embedded-atom-potential model including a long-range force for fcc metals and their alloys”, J. Cai and Y. Y. Ye, Phys. Rev. B 54 8398 (1996). ao (Å) Ecoh (e. V) Bulk M. (GPa) 4. 05035 (~0. 01%) 3. 31876 (-1. 23%) 75. 565 (-0. 57%) Al 3 Cu 3. 9025 (-0. 97%) 3. 47614 84. 542 Al. Cu 3. 75912 (-1. 93%) 3. 59907 98. 7448 Al. Cu 3 3. 65454 (-1. 87%) 3. 64548 117. 274 Cu 3. 61503 (~0. 001%) 3. 5241 (-0. 45%) 139. 8872 (-0. 08%) Al (FCC) FCC (L 12) L 12 (L 10) (L 12) (FCC) L 10 *Al, Cu is the source material of GMR device
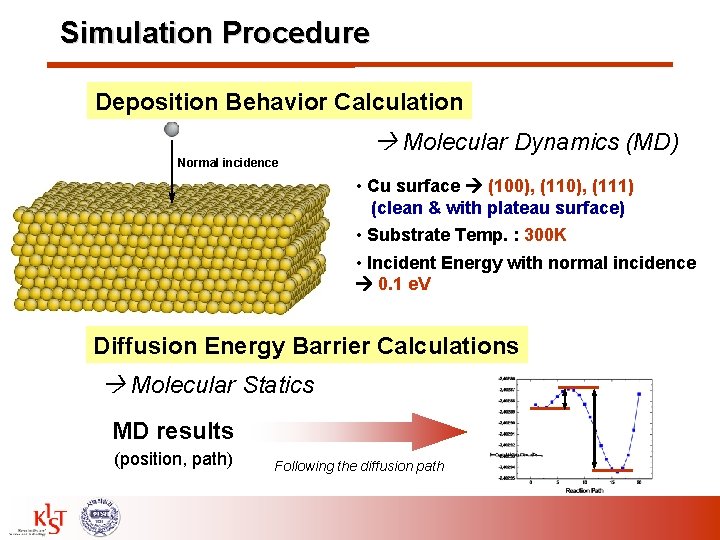
Simulation Procedure Deposition Behavior Calculation Molecular Dynamics (MD) Normal incidence • Cu surface (100), (111) (clean & with plateau surface) • Substrate Temp. : 300 K • Incident Energy with normal incidence 0. 1 e. V Diffusion Energy Barrier Calculations Molecular Statics MD results (position, path) Following the diffusion path

0. 5 ML Al on Cu(100) Clean surface Cu surface atoms With plateau Cu plateau atoms Al deposit atoms
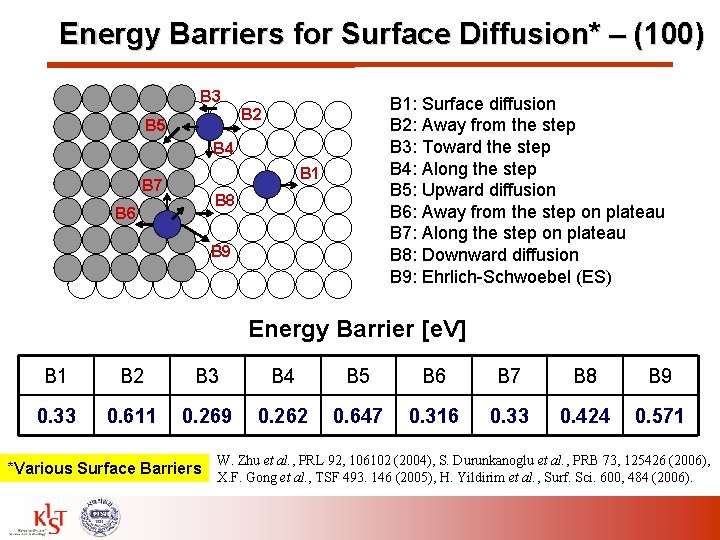
Energy Barriers for Surface Diffusion* – (100) B 3 B 1: Surface diffusion B 2: Away from the step B 3: Toward the step B 4: Along the step B 5: Upward diffusion B 6: Away from the step on plateau B 7: Along the step on plateau B 8: Downward diffusion B 9: Ehrlich-Schwoebel (ES) B 2 B 5 B 4 B 7 B 6 B 1 B 8 B 9 Energy Barrier [e. V] B 1 B 2 B 3 B 4 B 5 B 6 B 7 B 8 B 9 0. 33 0. 611 0. 269 0. 262 0. 647 0. 316 0. 33 0. 424 0. 571 W. Zhu et al. , PRL 92, 106102 (2004), S. Durunkanoglu et al. , PRB 73, 125426 (2006), *Various Surface Barriers X. F. Gong et al. , TSF 493. 146 (2005), H. Yildirim et al. , Surf. Sci. 600, 484 (2006).
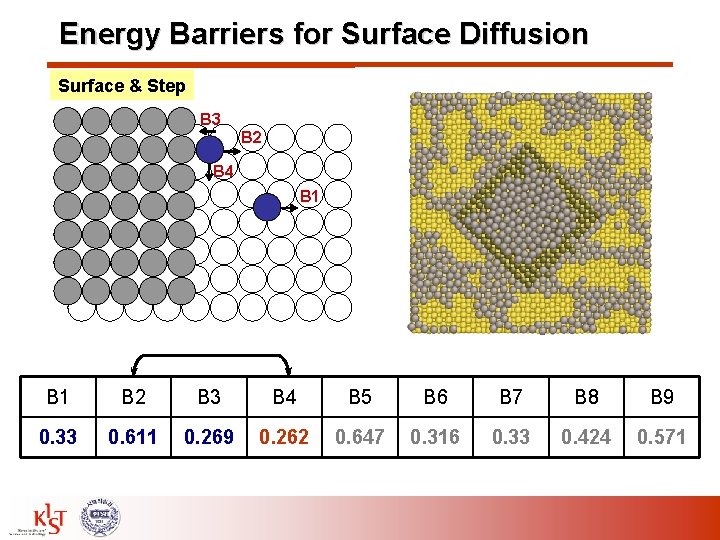
Energy Barriers for Surface Diffusion Surface & Step B 3 B 2 B 4 B 1 B 2 B 3 B 4 B 5 B 6 B 7 B 8 B 9 0. 33 0. 611 0. 269 0. 262 0. 647 0. 316 0. 33 0. 424 0. 571
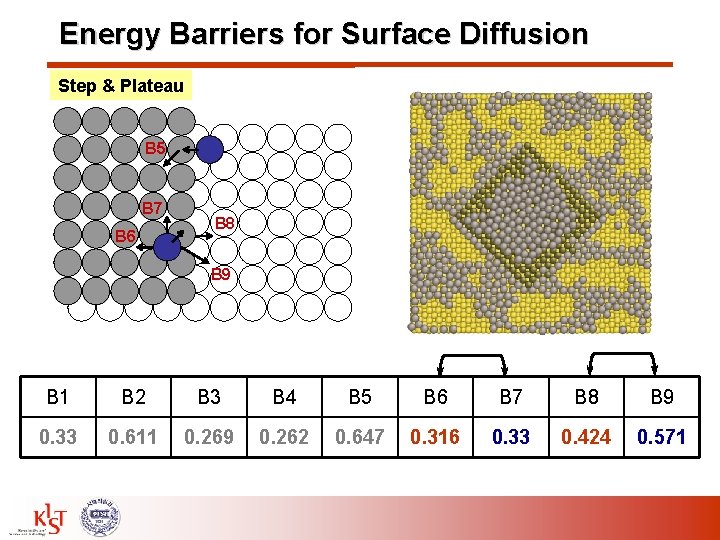
Energy Barriers for Surface Diffusion Step & Plateau B 5 B 7 B 6 B 8 B 9 B 1 B 2 B 3 B 4 B 5 B 6 B 7 B 8 B 9 0. 33 0. 611 0. 269 0. 262 0. 647 0. 316 0. 33 0. 424 0. 571

0. 5 ML Al on Cu(111) Clean surface Cu surface atoms With plateau Cu plateau atoms Al deposit atoms
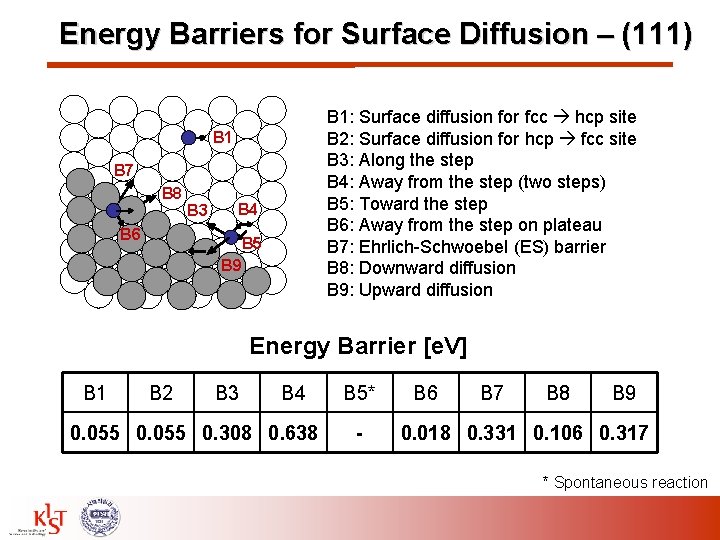
Energy Barriers for Surface Diffusion – (111) B 1: Surface diffusion for fcc hcp site B 2: Surface diffusion for hcp fcc site B 3: Along the step B 4: Away from the step (two steps) B 5: Toward the step B 6: Away from the step on plateau B 7: Ehrlich-Schwoebel (ES) barrier B 8: Downward diffusion B 9: Upward diffusion B 1 B 7 B 8 B 6 B 3 B 4 B 5 B 9 Energy Barrier [e. V] B 1 B 2 B 3 B 4 0. 055 0. 308 0. 638 B 5* - B 6 B 7 B 8 B 9 0. 018 0. 331 0. 106 0. 317 * Spontaneous reaction
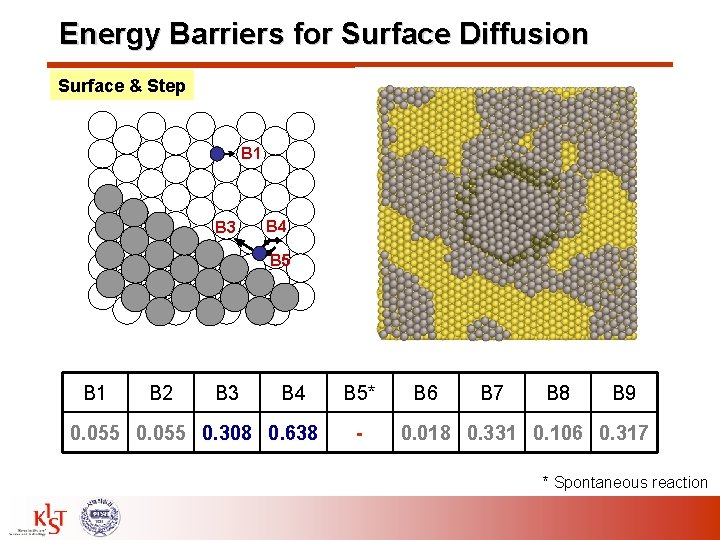
Energy Barriers for Surface Diffusion Surface & Step B 1 B 3 B 4 B 5 B 1 B 2 B 3 B 4 0. 055 0. 308 0. 638 B 5* - B 6 B 7 B 8 B 9 0. 018 0. 331 0. 106 0. 317 * Spontaneous reaction
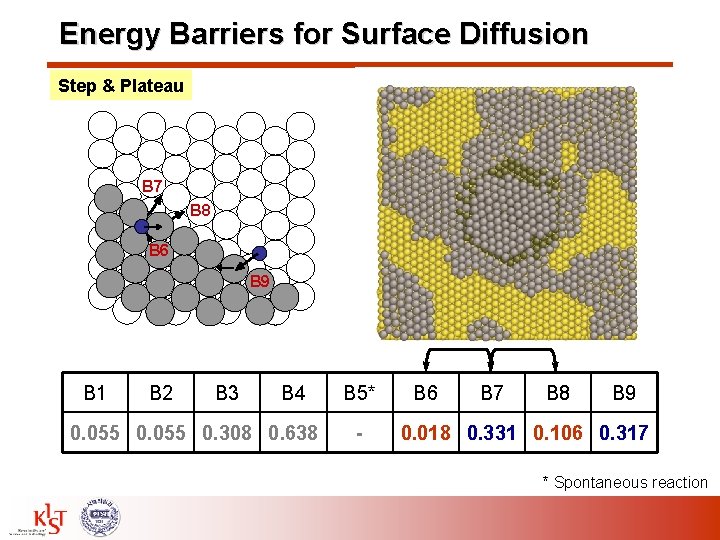
Energy Barriers for Surface Diffusion Step & Plateau B 7 B 8 B 6 B 9 B 1 B 2 B 3 B 4 0. 055 0. 308 0. 638 B 5* - B 6 B 7 B 8 B 9 0. 018 0. 331 0. 106 0. 317 * Spontaneous reaction

0. 5 ML Al on Cu (110) Clean surface Cu surface atoms With plateau Cu plateau atoms & same height with plateau atoms Al deposit atoms
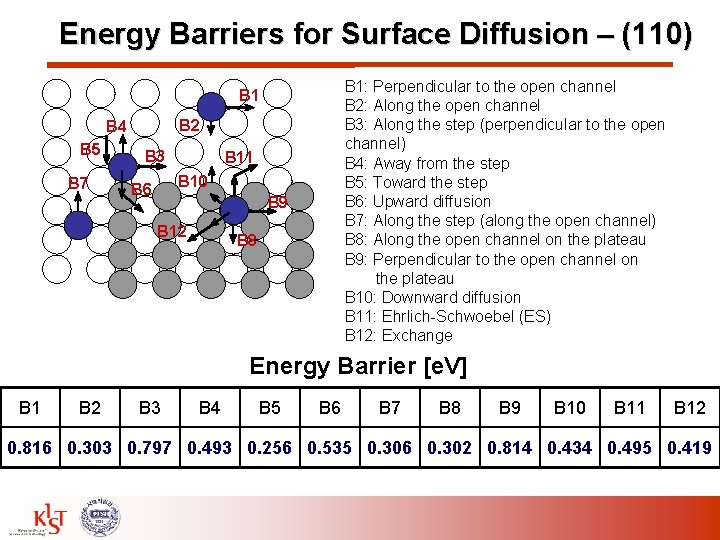
Energy Barriers for Surface Diffusion – (110) B 1: Perpendicular to the open channel B 2: Along the open channel B 3: Along the step (perpendicular to the open channel) B 4: Away from the step B 5: Toward the step B 6: Upward diffusion B 7: Along the step (along the open channel) B 8: Along the open channel on the plateau B 9: Perpendicular to the open channel on the plateau B 10: Downward diffusion B 11: Ehrlich-Schwoebel (ES) B 12: Exchange B 1 B 2 B 4 B 5 B 7 B 3 B 11 B 10 B 6 B 9 B 12 B 8 Energy Barrier [e. V] B 1 B 2 B 3 B 4 B 5 B 6 B 7 B 8 B 9 B 10 B 11 B 12 0. 816 0. 303 0. 797 0. 493 0. 256 0. 535 0. 306 0. 302 0. 814 0. 434 0. 495 0. 419
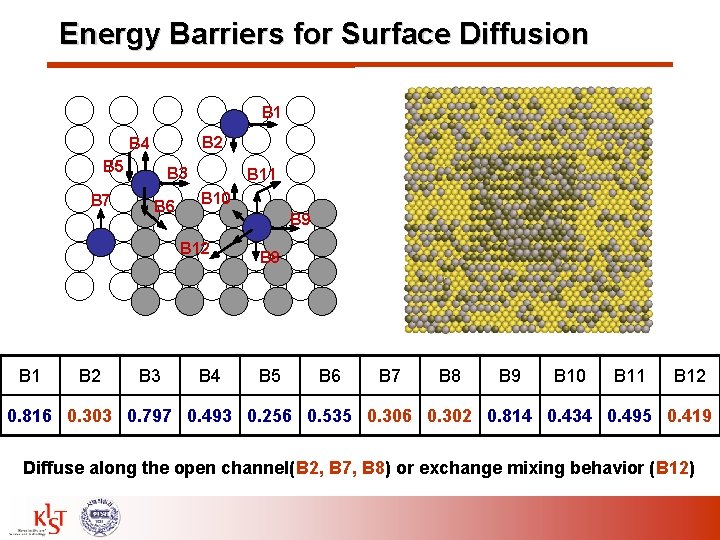
Energy Barriers for Surface Diffusion B 1 B 2 B 4 B 5 B 7 B 3 B 6 B 11 B 10 B 9 B 12 B 1 B 2 B 3 B 4 B 8 B 5 B 6 B 7 B 8 B 9 B 10 B 11 B 12 0. 816 0. 303 0. 797 0. 493 0. 256 0. 535 0. 306 0. 302 0. 814 0. 434 0. 495 0. 419 Diffuse along the open channel(B 2, B 7, B 8) or exchange mixing behavior (B 12)
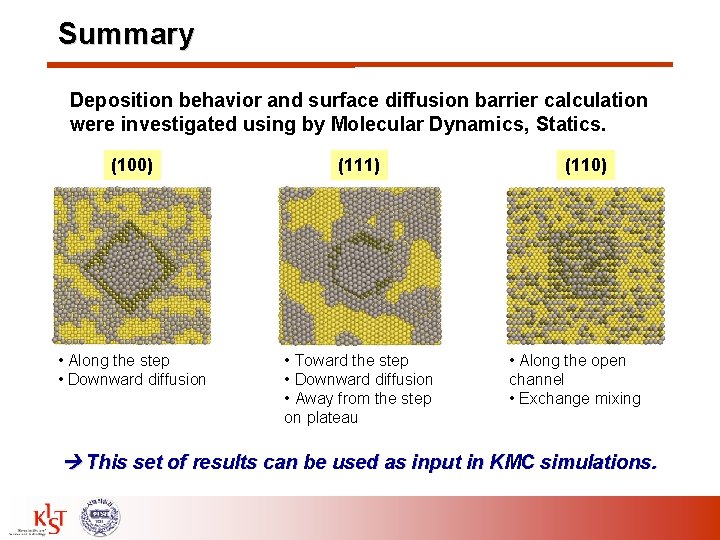
Summary Deposition behavior and surface diffusion barrier calculation were investigated using by Molecular Dynamics, Statics. (100) (111) • Along the step • Downward diffusion • Toward the step • Downward diffusion • Away from the step on plateau (110) • Along the open channel • Exchange mixing This set of results can be used as input in KMC simulations.
- Slides: 17