Molecular Diversity Arising from variation in carbon skeletons

Molecular Diversity Arising from variation in carbon skeletons Chapter 3

Carbon Chains • Carbon chains form the skeletons of most organic molecules. • Hydrocarbons—Organic molecules consisting of only carbon and hydrogen. • Hydrocarbons can undergo reactions that release a relatively large amount of energy.
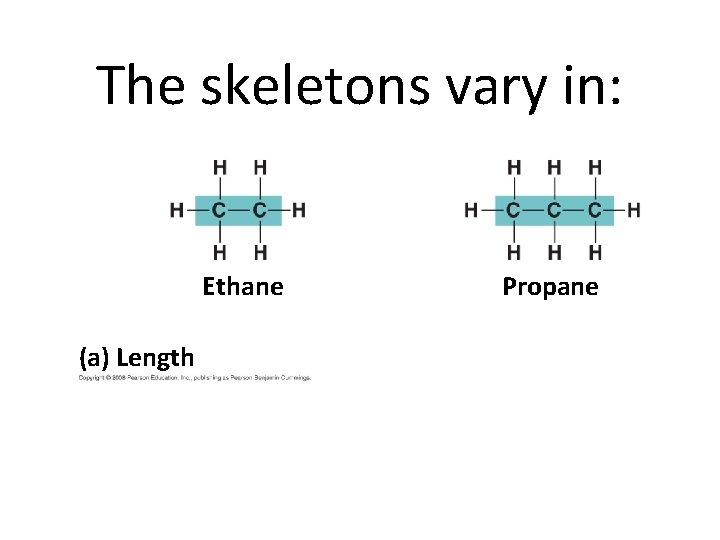
The skeletons vary in: Ethane (a) Length Propane
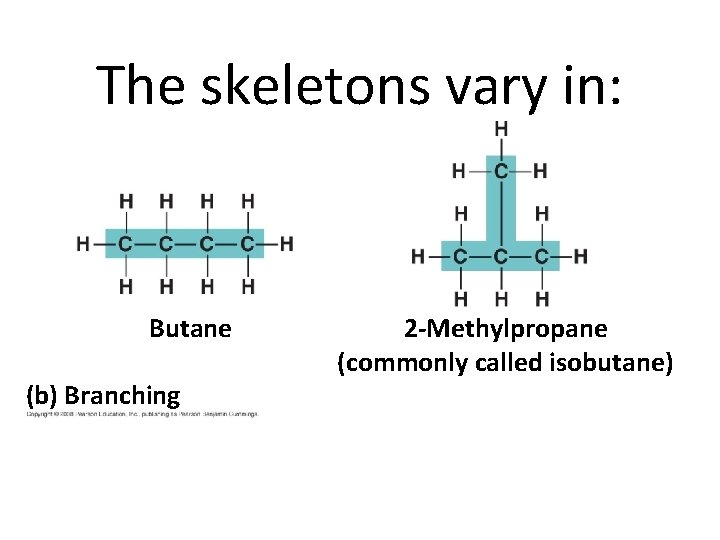
The skeletons vary in: Butane (b) Branching 2 -Methylpropane (commonly called isobutane)
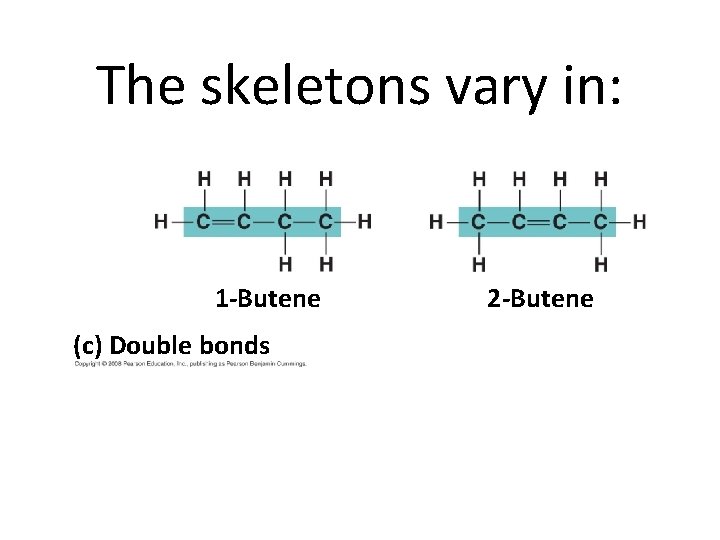
The skeletons vary in: 1 -Butene (c) Double bonds 2 -Butene
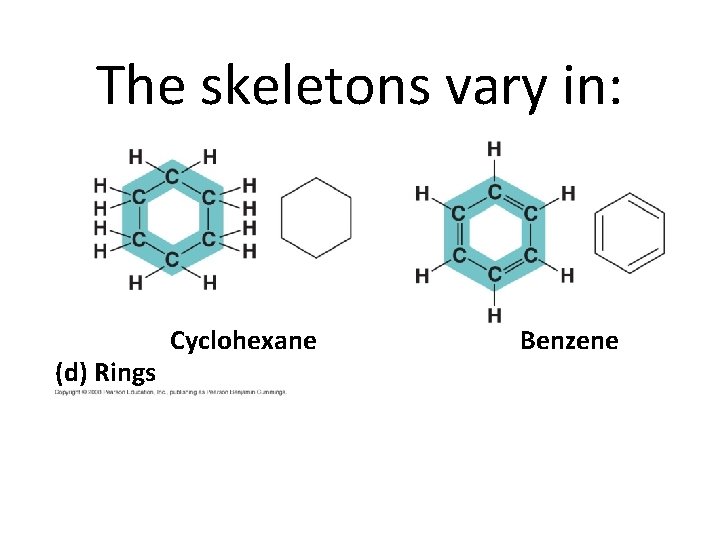
The skeletons vary in: (d) Rings Cyclohexane Benzene

The chemical groups most important to life • The distinctive properties of an organic molecule depend not only on its carbon skeleton but also on the chemical groups attached to that skeleton. • These chemical groups are known as functional groups.

1. Hydroxyl Group (—OH) Compound Name: Alcohol -The specific name usually ends in –ol Examples: Ethanol, the alcohol present in alcoholic beverages.
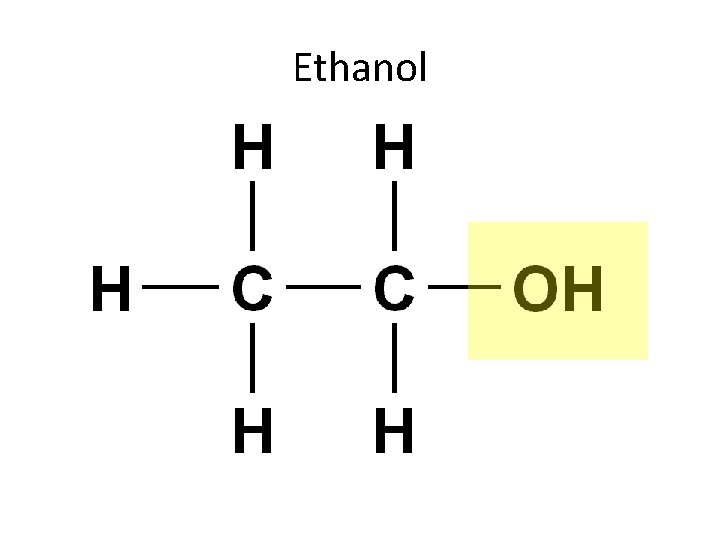
Ethanol
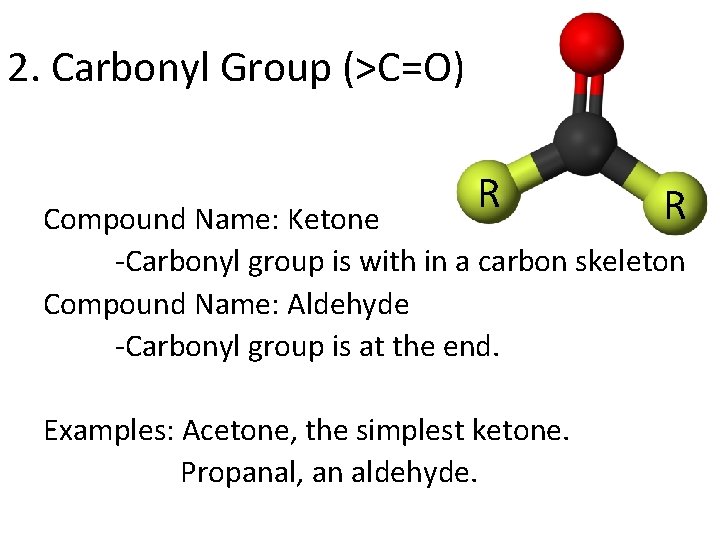
2. Carbonyl Group (>C=O) R R Compound Name: Ketone -Carbonyl group is with in a carbon skeleton Compound Name: Aldehyde -Carbonyl group is at the end. Examples: Acetone, the simplest ketone. Propanal, an aldehyde.
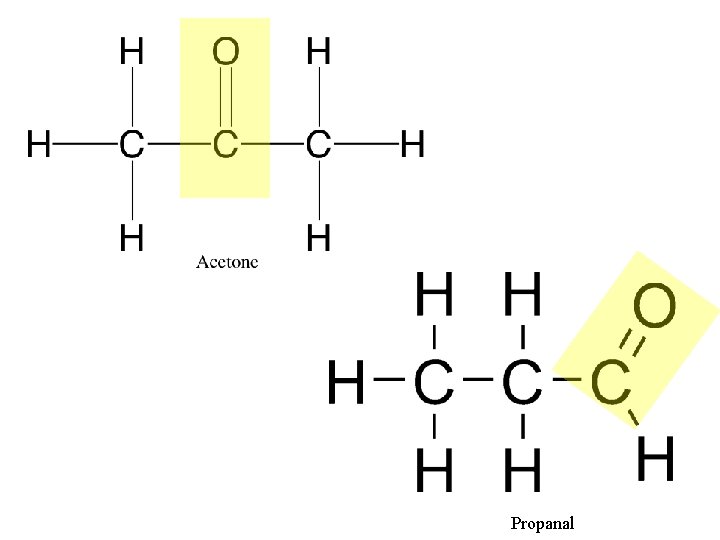
Propanal

3. Carboxyl Group (—COOH) Compound Name: Carboxylic acid or Organic acid R Examples: Acetic acid, which gives vinegar its sour taste.
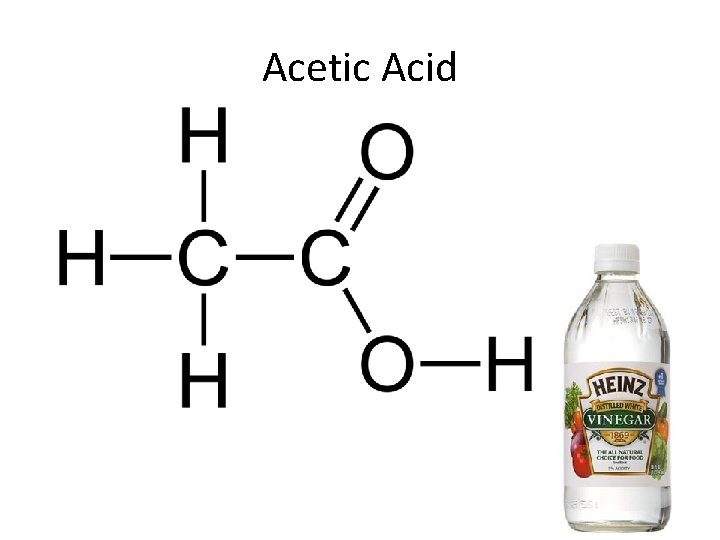
Acetic Acid
- Slides: 13