Molecular Compounds Covalent Bonds The atoms held together

Molecular Compounds
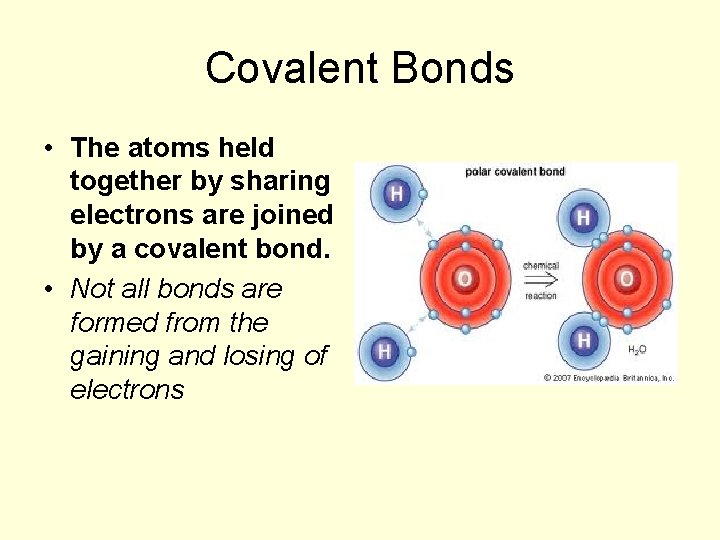
Covalent Bonds • The atoms held together by sharing electrons are joined by a covalent bond. • Not all bonds are formed from the gaining and losing of electrons
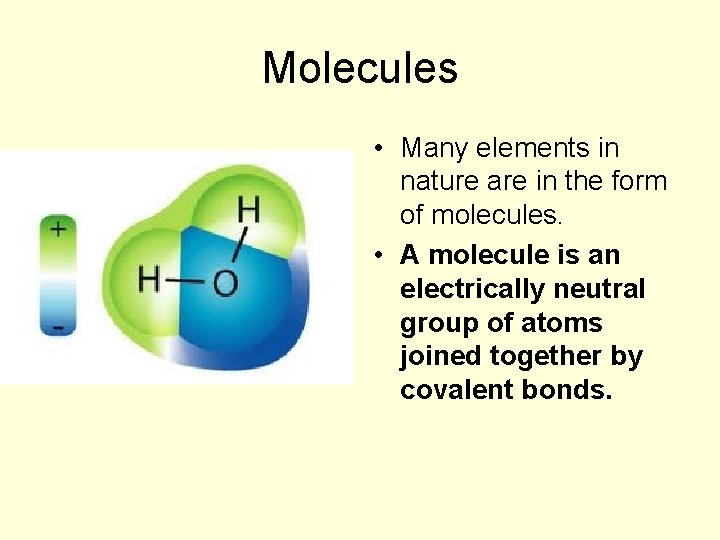
Molecules • Many elements in nature are in the form of molecules. • A molecule is an electrically neutral group of atoms joined together by covalent bonds.

Diatomic Molecules • A diatomic molecule consists of 2 atoms. • An example is the oxygen in this room = O 2
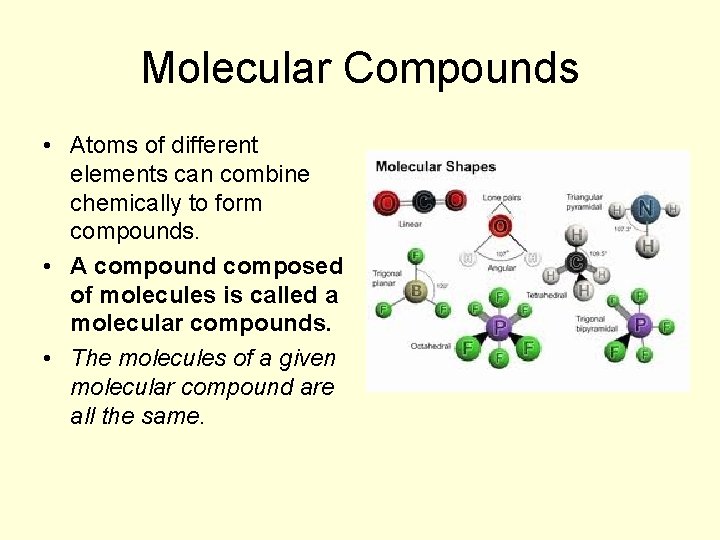
Molecular Compounds • Atoms of different elements can combine chemically to form compounds. • A compound composed of molecules is called a molecular compounds. • The molecules of a given molecular compound are all the same.

Properties of Molecular Compounds • Have relatively low melting and boiling points. • Many are gases or liquids at room temperature. • Most are formed from two or more nonmetals.

Molecular Formula • A molecular formula shows the kinds and numbers of atoms present in a molecule of a compound. • Example: H 2 O • A molecular formula represents the number of atoms in each mole. • Water has 2 Hydrogens and 1 Oxygen

Molecular Formulas • Describe molecules of elements • An oxygen molecule consists of 2 oxygen molecules bonded together = O 2 • A molecular formula shows how many atoms of each element a molecule contains. • It does NOT tell you about the molecules structure.

Different Forms of the Formula for Ammonia (NH 3)

Molecular Formula NH 3
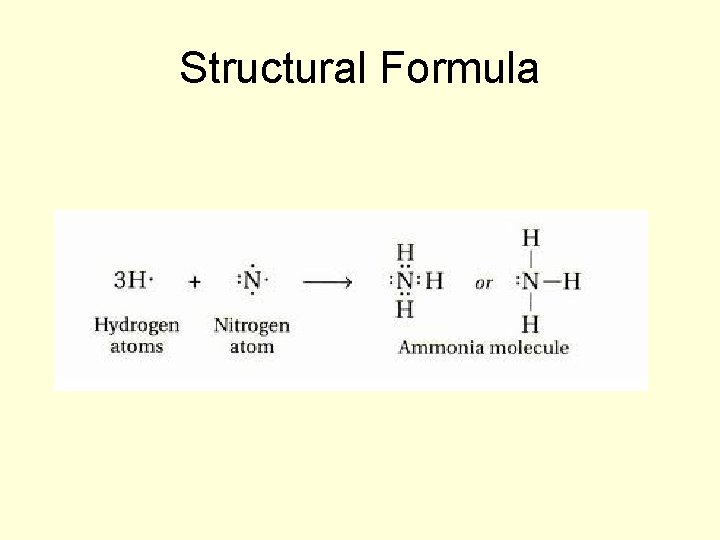
Structural Formula
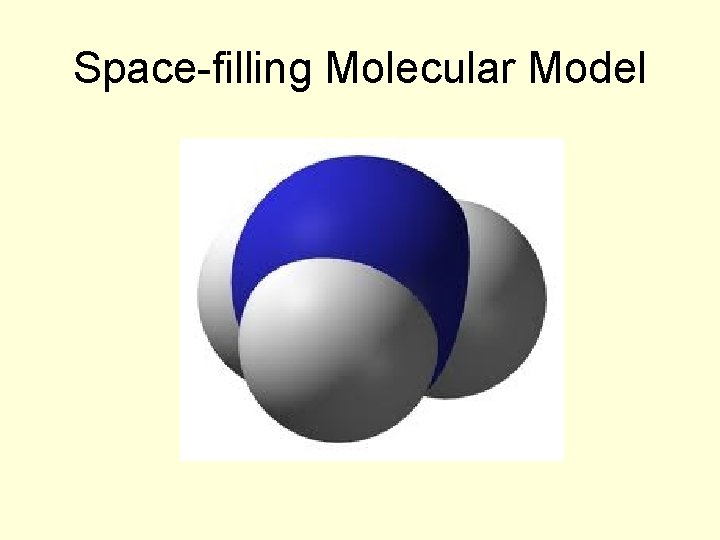
Space-filling Molecular Model
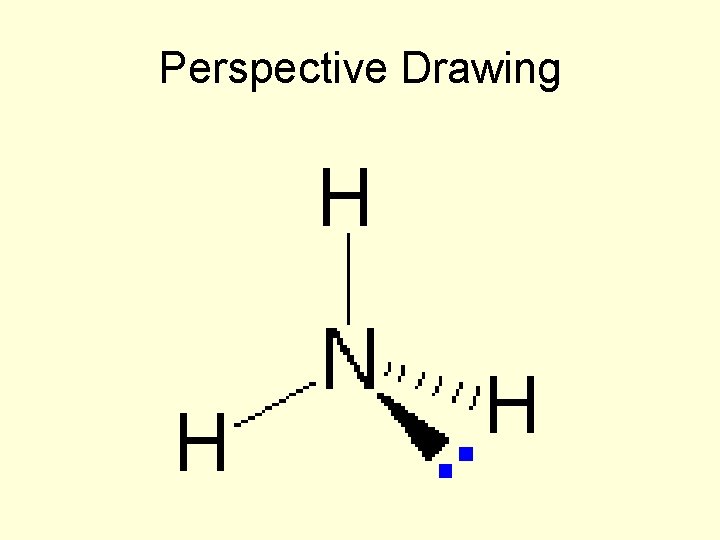
Perspective Drawing

Ball-and-stick Molecular Model
- Slides: 14