Molecular characterisation of PantonValentine Leukocidin PVLpositive Staphylococcus aureus
- Slides: 1

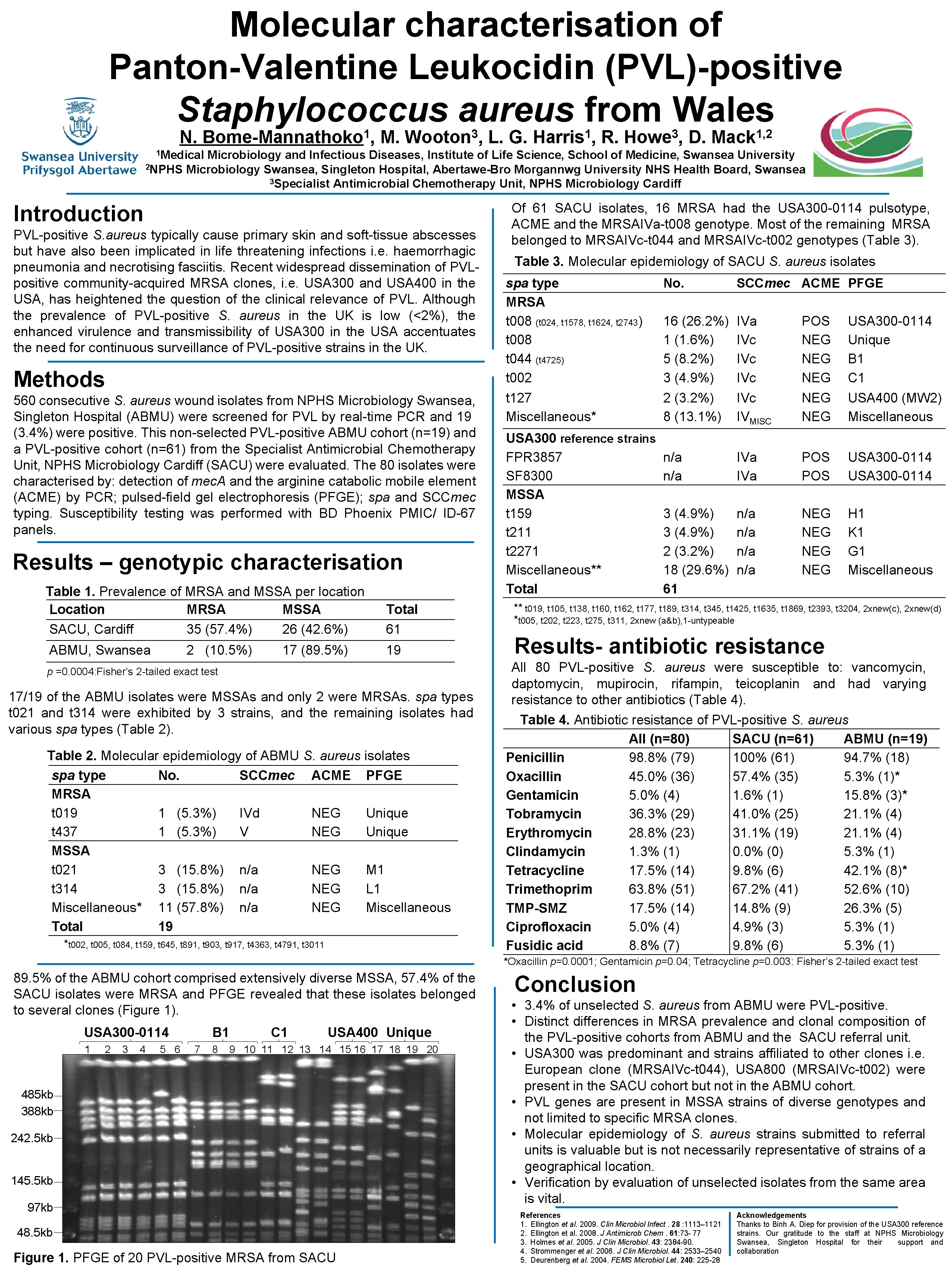
Molecular characterisation of Panton-Valentine Leukocidin (PVL)-positive Staphylococcus aureus from Wales N. 1 Bome-Mannathoko , M. 3 Wooton , L. G. 1 Harris , R. 3 Howe , D. 1, 2 Mack 1 Medical Microbiology and Infectious Diseases, Institute of Life Science, School of Medicine, Swansea University 2 NPHS Microbiology Swansea, Singleton Hospital, Abertawe-Bro Morgannwg University NHS Health Board, Swansea 3 Specialist Antimicrobial Chemotherapy Unit, NPHS Microbiology Cardiff Introduction PVL-positive S. aureus typically cause primary skin and soft-tissue abscesses but have also been implicated in life threatening infections i. e. haemorrhagic pneumonia and necrotising fasciitis. Recent widespread dissemination of PVLpositive community-acquired MRSA clones, i. e. USA 300 and USA 400 in the USA, has heightened the question of the clinical relevance of PVL. Although the prevalence of PVL-positive S. aureus in the UK is low (<2%), the enhanced virulence and transmissibility of USA 300 in the USA accentuates the need for continuous surveillance of PVL-positive strains in the UK. Methods 560 consecutive S. aureus wound isolates from NPHS Microbiology Swansea, Singleton Hospital (ABMU) were screened for PVL by real-time PCR and 19 (3. 4%) were positive. This non-selected PVL-positive ABMU cohort (n=19) and a PVL-positive cohort (n=61) from the Specialist Antimicrobial Chemotherapy Unit, NPHS Microbiology Cardiff (SACU) were evaluated. The 80 isolates were characterised by: detection of mec. A and the arginine catabolic mobile element (ACME) by PCR; pulsed-field gel electrophoresis (PFGE); spa and SCCmec typing. Susceptibility testing was performed with BD Phoenix PMIC/ ID-67 panels. Results – genotypic characterisation Table 1. Prevalence of MRSA and MSSA per location Location MRSA MSSA SACU, Cardiff 35 (57. 4%) 26 (42. 6%) Total 61 ABMU, Swansea 19 2 (10. 5%) 17 (89. 5%) p =0. 0004: Fisher’s 2 -tailed exact test 17/19 of the ABMU isolates were MSSAs and only 2 were MRSAs. spa types t 021 and t 314 were exhibited by 3 strains, and the remaining isolates had various spa types (Table 2). Table 2. Molecular epidemiology of ABMU S. aureus isolates spa type No. SCCmec ACME PFGE MRSA t 019 1 (5. 3%) IVd NEG Unique t 437 1 (5. 3%) V NEG Unique MSSA t 021 3 (15. 8%) n/a NEG M 1 t 314 3 (15. 8%) n/a NEG L 1 Miscellaneous* 11 (57. 8%) n/a NEG Miscellaneous Total 19 *t 002, t 005, t 084, t 159, t 645, t 891, t 903, t 917, t 4363, t 4791, t 3011 Of 61 SACU isolates, 16 MRSA had the USA 300 -0114 pulsotype, ACME and the MRSAIVa-t 008 genotype. Most of the remaining MRSA belonged to MRSAIVc-t 044 and MRSAIVc-t 002 genotypes (Table 3). Table 3. Molecular epidemiology of SACU S. aureus isolates spa type MRSA t 008 (t 024, t 1578, t 1624, t 2743) t 008 t 044 (t 4725) t 002 t 127 Miscellaneous* USA 300 reference strains FPR 3857 SF 8300 MSSA t 159 t 211 t 2271 Miscellaneous** Total No. SCCmec ACME PFGE 16 (26. 2%) 1 (1. 6%) 5 (8. 2%) 3 (4. 9%) 2 (3. 2%) 8 (13. 1%) IVa IVc IVc IVMISC POS NEG NEG NEG USA 300 -0114 Unique B 1 C 1 USA 400 (MW 2) Miscellaneous n/a IVa POS USA 300 -0114 3 (4. 9%) 2 (3. 2%) 18 (29. 6%) 61 n/a n/a NEG NEG H 1 K 1 G 1 Miscellaneous ** t 019, t 105, t 138, t 160, t 162, t 177, t 189, t 314, t 345, t 1425, t 1635, t 1869, t 2393, t 3204, 2 xnew(c), 2 xnew(d) *t 005, t 202, t 223, t 275, t 311, 2 xnew (a&b), 1 -untypeable Results- antibiotic resistance All 80 PVL-positive S. aureus were susceptible to: vancomycin, daptomycin, mupirocin, rifampin, teicoplanin and had varying resistance to other antibiotics (Table 4). Table 4. Antibiotic resistance of PVL-positive S. aureus All (n=80) SACU (n=61) ABMU (n=19) Penicillin 98. 8% (79) 100% (61) 94. 7% (18) Oxacillin 45. 0% (36) 57. 4% (35) 5. 3% (1)* Gentamicin 5. 0% (4) 1. 6% (1) 15. 8% (3)* Tobramycin 36. 3% (29) 41. 0% (25) 21. 1% (4) Erythromycin 28. 8% (23) 31. 1% (19) 21. 1% (4) Clindamycin 1. 3% (1) 0. 0% (0) 5. 3% (1) Tetracycline 17. 5% (14) 9. 8% (6) 42. 1% (8)* Trimethoprim 63. 8% (51) 67. 2% (41) 52. 6% (10) TMP-SMZ 17. 5% (14) 14. 8% (9) 26. 3% (5) Ciprofloxacin 5. 0% (4) 4. 9% (3) 5. 3% (1) Fusidic acid 8. 8% (7) 9. 8% (6) 5. 3% (1) *Oxacillin p=0. 0001; Gentamicin p=0. 04; Tetracycline p=0. 003: Fisher’s 2 -tailed exact test 89. 5% of the ABMU cohort comprised extensively diverse MSSA, 57. 4% of the SACU isolates were MRSA and PFGE revealed that these isolates belonged to several clones (Figure 1). USA 300 -0114 1 2 3 4 5 6 B 1 7 8 C 1 USA 400 Unique 9 10 11 12 13 14 15 16 17 18 19 20 485 kb 388 kb 242. 5 kb 145. 5 kb 97 kb 48. 5 kb Figure 1. PFGE of 20 PVL-positive MRSA from SACU Conclusion • 3. 4% of unselected S. aureus from ABMU were PVL-positive. • Distinct differences in MRSA prevalence and clonal composition of the PVL-positive cohorts from ABMU and the SACU referral unit. • USA 300 was predominant and strains affiliated to other clones i. e. European clone (MRSAIVc-t 044), USA 800 (MRSAIVc-t 002) were present in the SACU cohort but not in the ABMU cohort. • PVL genes are present in MSSA strains of diverse genotypes and not limited to specific MRSA clones. • Molecular epidemiology of S. aureus strains submitted to referral units is valuable but is not necessarily representative of strains of a geographical location. • Verification by evaluation of unselected isolates from the same area is vital. References 1. Ellington et al. 2009. Clin Microbiol Infect. 28 : 1113– 1121 2. Ellington et al. 2008. J Antimicrob Chem. 61: 73 - 77 3. Holmes et al. 2005. J Clin Microbiol. 43: 2384 -90. 4. Strommenger et al. 2006. J Clin Microbiol. 44: 2533– 2540 5. Deurenberg et al. 2004. FEMS Microbiol Let. 240: 225 -28 Acknowledgements Thanks to Binh A. Diep for provision of the USA 300 reference strains. Our gratitude to the staff at NPHS Microbiology Swansea, Singleton Hospital for their support and collaboration