Molecular Cell Biology Kinetics Enzymology Cooper Kinetic analysis

Molecular Cell Biology Kinetics: Enzymology Cooper

Kinetic analysis • How cells change over long time periods (development, long term adaptive changes; hours - years) • Movement of proteins and membranes within cells dynamics of cellular events (sec - hrs) – Pulse chase analyses – Real time imaging: GFP and other fluorophores allow measurement of trafficking, diffusion, etc. (time-lapse, fluorescence recovery after photobleaching (FRAP), etc. ) • Kinetics of molecular interactions, enzyme reactions (msec - min)
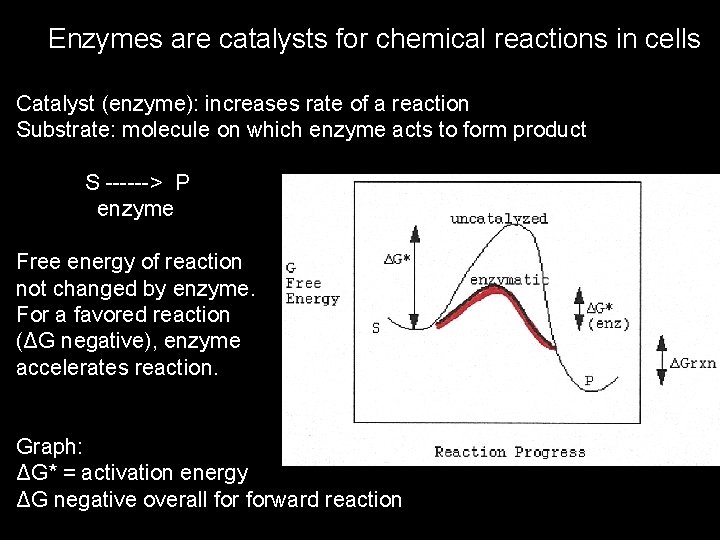
Enzymes are catalysts for chemical reactions in cells Catalyst (enzyme): increases rate of a reaction Substrate: molecule on which enzyme acts to form product S ------> P enzyme Free energy of reaction not changed by enzyme. For a favored reaction (ΔG negative), enzyme accelerates reaction. Graph: ΔG* = activation energy ΔG negative overall forward reaction

Enzymes as Catalysts Active Site: Region of the enzyme that does the work. Amino acid residues in this site assume certain 3 D conformation, which promotes the desired reaction. What does the Enzyme do to cause catalysis? • High affinity for substrate in its transition state, facilitating transition to product • Increased probability of proper orientation of substrates • Increased local concentration of substrates • Has atoms in places that push the reaction forward • Change hydration sphere of substrates
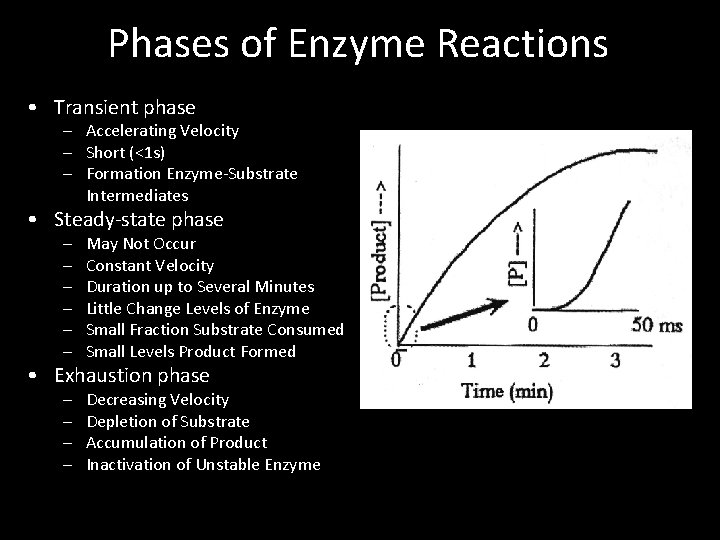
Phases of Enzyme Reactions • Transient phase – Accelerating Velocity – Short (<1 s) – Formation Enzyme-Substrate Intermediates • Steady-state phase – – – May Not Occur Constant Velocity Duration up to Several Minutes Little Change Levels of Enzyme Small Fraction Substrate Consumed Small Levels Product Formed – – Decreasing Velocity Depletion of Substrate Accumulation of Product Inactivation of Unstable Enzyme • Exhaustion phase

What Can You Learn from What Happens at Steady State? • Turnover number => catalytic efficiency of enzyme • Affinity of enzyme for substrates • Lower bounds for rate constants • Inhibitors and p. H variations to probe active site • Details of mechanism require transient (presteady state) kinetic analysis

How to Measure Enzyme Activity at Steady State Need an assay that measures the product of the chemical reaction. For example. . . Enzyme β-galactosidase catalyzes this reaction: lactose ----------> glucose + galactose Measure the amount of glucose or galactose over time. Trick - use a substrate that produces a reaction product that absorbs light (creates color). Measure absorbance.

Color-Producing Substrates for β-galactosidase ONPG = ONP-galactose (ONP = o-nitro-phenol) ONPG -------> galactose + ONP (colorless) (yellow) X-gal = X-galactose (X = 4 -chloro-3 -bromo indole) X-gal --------> galactose + 4 -Cl-3 -Br-indigo (colorless) (deep blue) Measure absorbance with a spectrophotometer • Beer’s law - concentration proportional to absorbance • 96 -well format instruments

Optimizing assay • • No Enzyme -> No Product Optimize p. H, salt, other buffer conditions Optimize temperature Choose set of conditions to be kept constant • Amount of enzyme – Linear range of assay – More is better
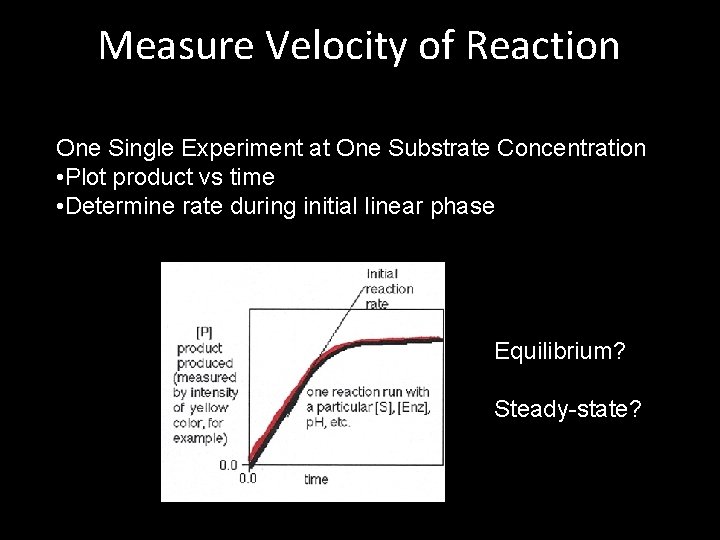
Measure Velocity of Reaction One Single Experiment at One Substrate Concentration • Plot product vs time • Determine rate during initial linear phase Equilibrium? Steady-state?
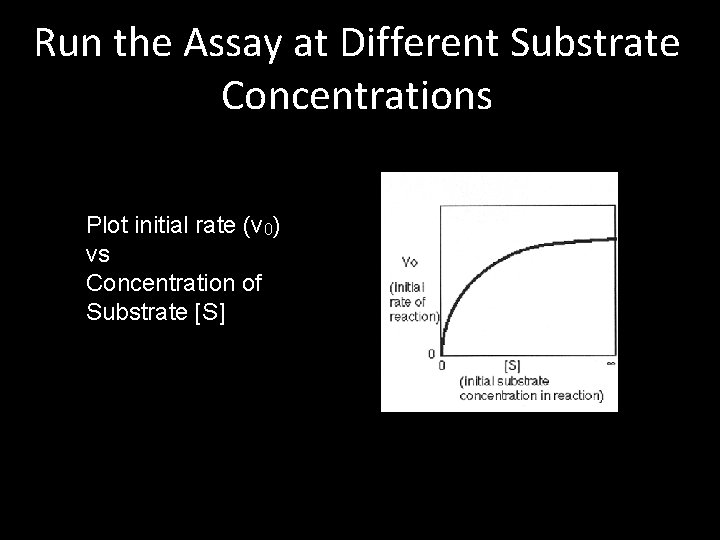
Run the Assay at Different Substrate Concentrations Plot initial rate (v 0) vs Concentration of Substrate [S]

Michaelis-Menten Plot • What’s interesting or useful about this plot? • Can we use this plot to compare results for different enzymes or conditions?

Deriving an Equation for the Curve Consider time zero • We measure the initial velocity of the reaction) • No product present: Back reaction is neglible, i. e. no k-2. The initial velocity, v 0, is therefore simply: v 0 = k 2[ES] (k 2 often called “kcat” - catalysis rate constant)
![Problem - [ES] cannot be measured • • However. . . [S 0] (the Problem - [ES] cannot be measured • • However. . . [S 0] (the](http://slidetodoc.com/presentation_image_h2/4ee8caa2546ae5a3e4d864027ed35e9a/image-14.jpg)
Problem - [ES] cannot be measured • • However. . . [S 0] (the initial concentration of substrate) is known [P] (product produced) can be measured [ETotal] (the amount of enzyme added to the reaction) is known • The individual rate equations allow us to solve, using algebra, for [ES] in terms of these known values
![At steady state, d[ES]/dt is zero. So. . . At steady state, d[ES]/dt is zero. So. . .](http://slidetodoc.com/presentation_image_h2/4ee8caa2546ae5a3e4d864027ed35e9a/image-15.jpg)
At steady state, d[ES]/dt is zero. So. . .
![Solving for [ES]. . . To simply, let’s define a constant, Km, the Michaelis Solving for [ES]. . . To simply, let’s define a constant, Km, the Michaelis](http://slidetodoc.com/presentation_image_h2/4ee8caa2546ae5a3e4d864027ed35e9a/image-16.jpg)
Solving for [ES]. . . To simply, let’s define a constant, Km, the Michaelis constant as. . . This simplifies the equation:
![But. . . we don’t know [E]. We do know that the total amount But. . . we don’t know [E]. We do know that the total amount](http://slidetodoc.com/presentation_image_h2/4ee8caa2546ae5a3e4d864027ed35e9a/image-17.jpg)
But. . . we don’t know [E]. We do know that the total amount of Enzyme is the sum of E and ES. . . [E 0] = [E] + [ES] thus. . [E] = [E 0] − [ES] Substituting for [E]. . .
![Rearrange to solve for [ES]. . . From before, the rate (or velocity) of Rearrange to solve for [ES]. . . From before, the rate (or velocity) of](http://slidetodoc.com/presentation_image_h2/4ee8caa2546ae5a3e4d864027ed35e9a/image-18.jpg)
Rearrange to solve for [ES]. . . From before, the rate (or velocity) of the reaction is. . . Substituting for [ES]. . .

V, the velocity (rate) of the reaction is. . . How does v depend on (vary with) S?

V 0 Km is the “Michaelis-Menten constant” - the substrate concentration at which reaction velocity is half-maximal. Km = (k-1 + k 2)/k 1 Typical values? n. M to m. M Vmax = k 2 [E]total = kcat [E]total Typical kcat values? 1 -1000 per second
![Consider three situations. . . V 0 1. [S] very large, much greater than Consider three situations. . . V 0 1. [S] very large, much greater than](http://slidetodoc.com/presentation_image_h2/4ee8caa2546ae5a3e4d864027ed35e9a/image-21.jpg)
Consider three situations. . . V 0 1. [S] very large, much greater than Km The enzyme will be saturated with substrate. [S] + Km = ~ [S], so the rate equation simplifies to. . . v 0 = Vmax 2. [S] very small, much less than Km [S] + Km = ~ Km , so the equation simplifies to. . . v 0 linearly proportional to [S] 3. [S]=Km v 0 = 50% of Vmax

How Km values affect metabolism • Glucose + ATP --> glucose-6 -P + ADP + H+ • Typical cell [glucose] = 5 m. M • Two enzymes catalyze above reaction – Hexokinase • Km (glucose) = 0. 1 m. M • Km << [S], so velocity independent of [glucose] • Reaction is inhibited by product--regulated by product utilization – Glucokinase • Km (glucose) = 10 m. M • Km > [S], promotes glucose utilization only when [glucose] is high • Reaction not inhibited by product--regulated by substrate availability
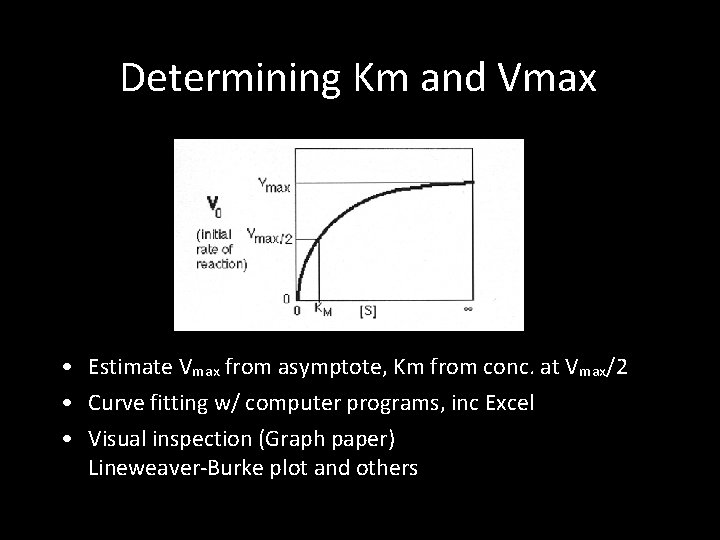
Determining Km and Vmax • Estimate Vmax from asymptote, Km from conc. at Vmax/2 • Curve fitting w/ computer programs, inc Excel • Visual inspection (Graph paper) Lineweaver-Burke plot and others
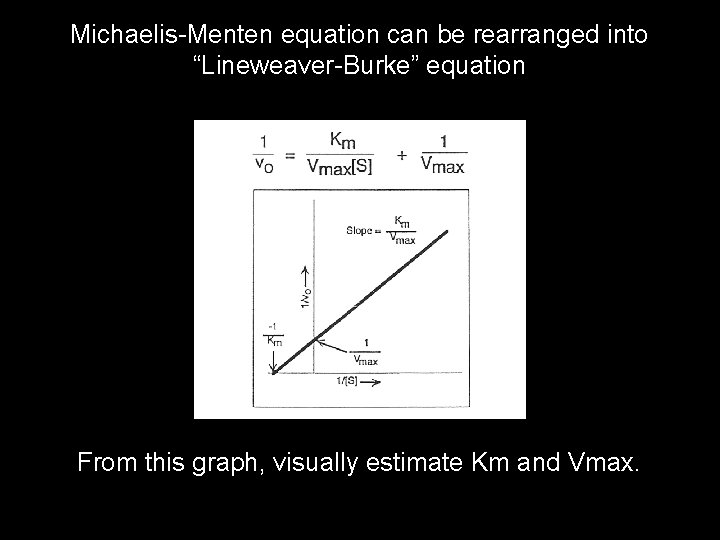
Michaelis-Menten equation can be rearranged into “Lineweaver-Burke” equation From this graph, visually estimate Km and Vmax.

Regulating enzyme activity • • • Allosteric regulation Reversible covalent modifications Enzyme availability (synthesis, degradation, localization) Substrate availability (synthesis, degradation, localization) Inhibition – By specific metabolites within the cell – By drugs, toxins, etc. – By specific analogues in study of reaction mechanism

Competitive Inhibition Competitive inhibitor. . . • binds to free enzyme • prevents simultaneous binding of substrate - i. e. competes with substrate • Apparent Km of the substrate is therefore increased • High substrate concentration: - substrate overcomes inhibition by mass action - v 0 approaches Vmax (which does not change)

Example of Competitive Inhibition • Et. OH Rx for Me. OH poisoning • Methanol (ingested from solid alcohol, paint strippers, windshield washer fluid, etc. ) is metabolized by alcohol dehydrogenase to formaldehyde and formic acid. Leads to metabolic acidosis and optic neuritis (from formate) that can cause blindness. • Treatment: Infuse Et. OH to keep blood concentration at 100 -200 mg/d. L (legally intoxicated) for long enough to excrete the Me. OH. • Et. OH serves as a competitive inhibitor. Ethylene glycol poisoning is treated in the same way.

Noncompetitive Inhibition • Noncompetitive inhibitor. . . • Binds to a site on the enzyme (E or ES) that inactivates the enzyme • Decreases total amount of enzyme available for catalysis, decreasing Vmax • Remaining active enzyme molecules are unaffected, so Km is unchanged
![Uncompetitive Inhibition • Uncompetitive inhibitor. . . • Binds specifically to the [ES] complex Uncompetitive Inhibition • Uncompetitive inhibitor. . . • Binds specifically to the [ES] complex](http://slidetodoc.com/presentation_image_h2/4ee8caa2546ae5a3e4d864027ed35e9a/image-29.jpg)
Uncompetitive Inhibition • Uncompetitive inhibitor. . . • Binds specifically to the [ES] complex (and inactivates it • Fraction of enzyme inhibited increases as [S] increases • So both Km and Vmax are affected

Summary: Types of Inhibitors • Competitive – Binds Free Enzyme Only – Km Increased • Noncompetitive – Binds E and ES – Vmax Decreased • Uncompetitive – Binds ES only – Vmax Decreased – Km Decreased
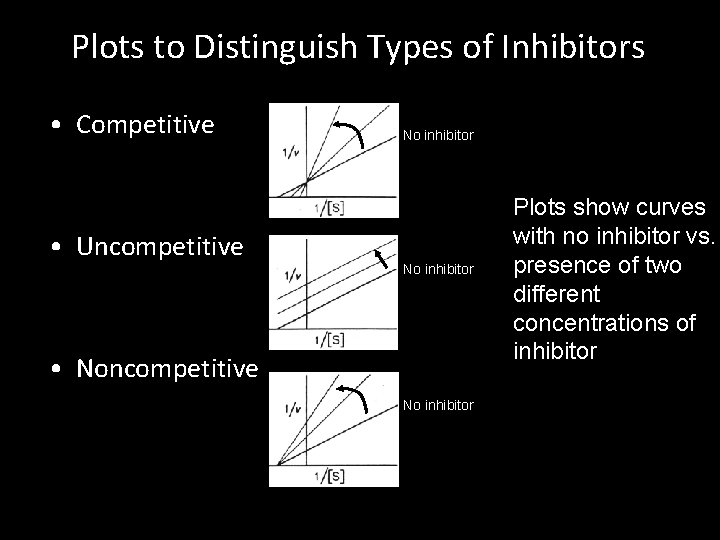
Plots to Distinguish Types of Inhibitors • Competitive • Uncompetitive No inhibitor • Noncompetitive No inhibitor Plots show curves with no inhibitor vs. presence of two different concentrations of inhibitor

Reading and Homework for Kinetics • Alberts (5 th edition) pp. 159 -166 • Lodish (6 th edition) pp. 79 -85 • See handout or website for homework
- Slides: 32