Mole Review 1 Calculate the number of moles
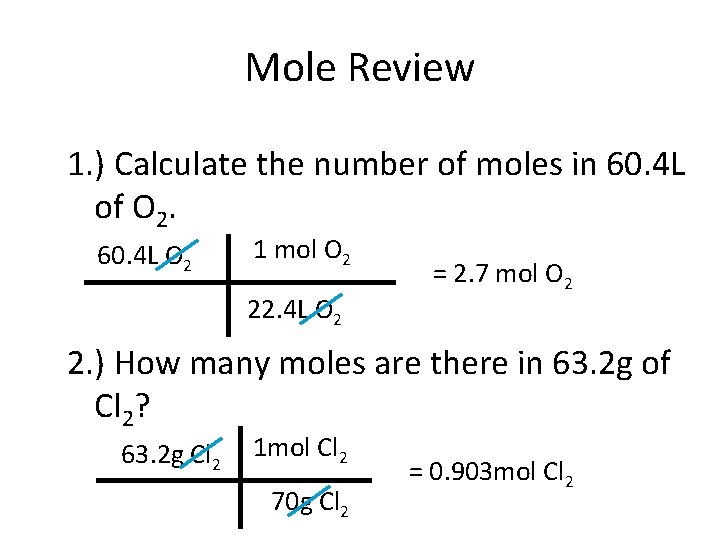
Mole Review 1. ) Calculate the number of moles in 60. 4 L of O 2. 60. 4 L O 2 1 mol O 2 22. 4 L O 2 = 2. 7 mol O 2 2. ) How many moles are there in 63. 2 g of Cl 2? 63. 2 g Cl 2 1 mol Cl 2 70 g Cl 2 = 0. 903 mol Cl 2

Ch. 9 Math In Chemistry Stoichiometry
Proportional Relationships Tiny Tyke Tricycle Company F + S + 3 W + H + 2 P → FSW 3 HP 2 Scheduled to make 640 tricycles. How many wheels should they order?

Proportional Relationships 2 1/4 c. flour 1 tsp. baking soda 1 tsp. salt 1 c. butter 3/4 c. sugar 3/4 c. brown sugar 1 tsp vanilla extract 2 eggs 2 c. chocolate chips Makes 5 dozen cookies. I have 5 eggs. How many cookies can I make? 5 eggs 5 doz. 2 eggs Ratio of eggs to cookies = 12. 5 dozen cookies

Proportional Relationships • Stoichiometry – mass relationships between substances in a chemical reaction • for example: you can determine the amount of a compound required to make another compound – based on the mole ratio • Mole Ratio – indicated by coefficients in a balanced equation 2 Mg + O 2 2 Mg. O 2 Moles of magnesium react with 1 mole of oxygen to form 2 moles of magnesium oxide.

2 Mg + O 2 2 Mg. O What would be the mole ratio of magnesium to magnesium oxide? 2: 2 Conversion factor = 2 mol Mg. O What would be the mole ratio of oxygen to magnesium? 1: 2 Conversion factor = 1 mol O 2 (Mole Ratio) 2 mol Mg

Practice 5 F 2 + 2 NH 3 N 2 F 4 + 6 HF 1. What is the mole ratio of NH 3 to F 2? 2: 5 Write the mole ratio as a conversion factor. 2 mol NH 3 5 mol F 2 2. What is the mole ratio of HF to N 2 F 4? 6: 1 Write the mole ratio as a conversion factor. 6 mol HF 1 mol N 2 F 4
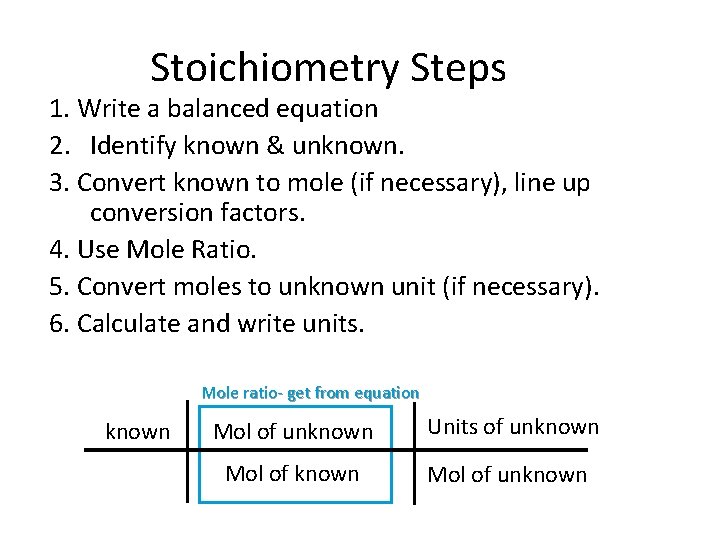
Stoichiometry Steps 1. Write a balanced equation 2. Identify known & unknown. 3. Convert known to mole (if necessary), line up conversion factors. 4. Use Mole Ratio. 5. Convert moles to unknown unit (if necessary). 6. Calculate and write units. Mole ratio- get from equation known Mol of unknown Units of unknown Mol of unknown

Mole - Mole Stoichiometry Formula: known mol unknown mol known 1. Write the known and unknown. 2. Use the balanced equation to find the mole ratio. 3. Calculate.

Mole-Mole Examples #1 __S + __O 2 → __SO 3 Ø Write the equation. Ø Balance the equation. Ø How many moles of SO 3 are produced when there are 4. 5 moles of S? Ø Known = Ø Unknown =
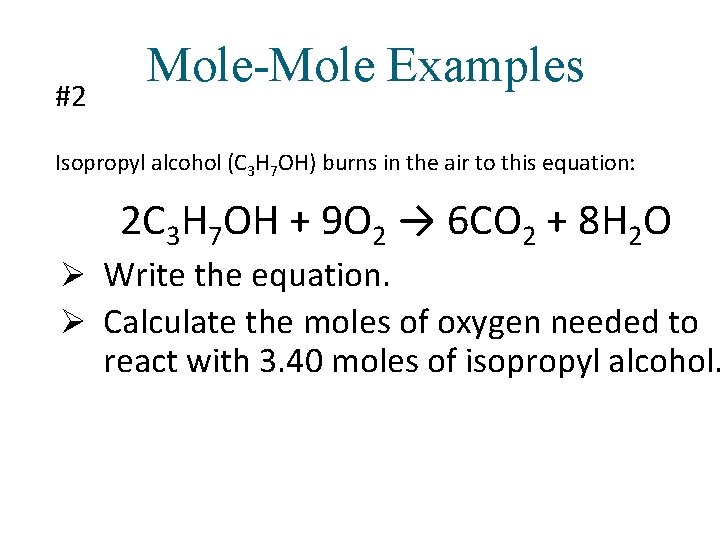
#2 Mole-Mole Examples Isopropyl alcohol (C 3 H 7 OH) burns in the air to this equation: 2 C 3 H 7 OH + 9 O 2 → 6 CO 2 + 8 H 2 O Ø Write the equation. Ø Calculate the moles of oxygen needed to react with 3. 40 moles of isopropyl alcohol.

Mole-Mole Examples #3 2 C 3 H 7 OH + 9 O 2 → 6 CO 2 + 8 H 2 O Ø Find the moles of water when 6. 20 mol O 2 reacts with C 3 H 7 OH.

Mass-Mass Stoichiometry • • Mass of reactants equals the mass of products, Law of Conservation of Mass ONLY mass and atoms are conserved in every chemical reaction 1. Write the known and unknown. 2. Find the molar mass of the known and unknown substances. 3. Use mole and molar mass conversion factors from Ch. 7 and mole ratios from the balanced equation to solve. Known g 1 mol Known mol unknown Molar mass Known mol known 1 mol unknown Mol ratioget from equation

Mass-Mass Stoichiometry #1 The reaction of fluorine with ammonia produces dinitrogen tetrafluoride and hydrogen fluoride. 5 F 2 + 2 NH 3 → N 2 F 4 + 6 HF How many grams of NH 3 are required to produce 7. 38 g HF?
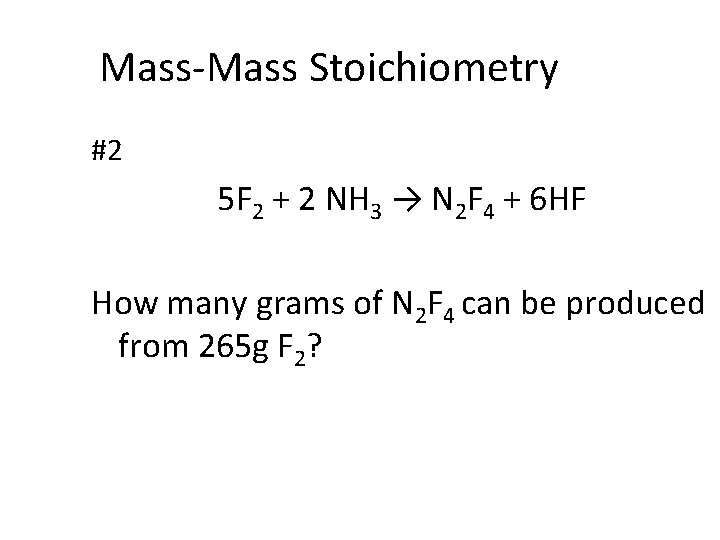
Mass-Mass Stoichiometry #2 5 F 2 + 2 NH 3 → N 2 F 4 + 6 HF How many grams of N 2 F 4 can be produced from 265 g F 2?
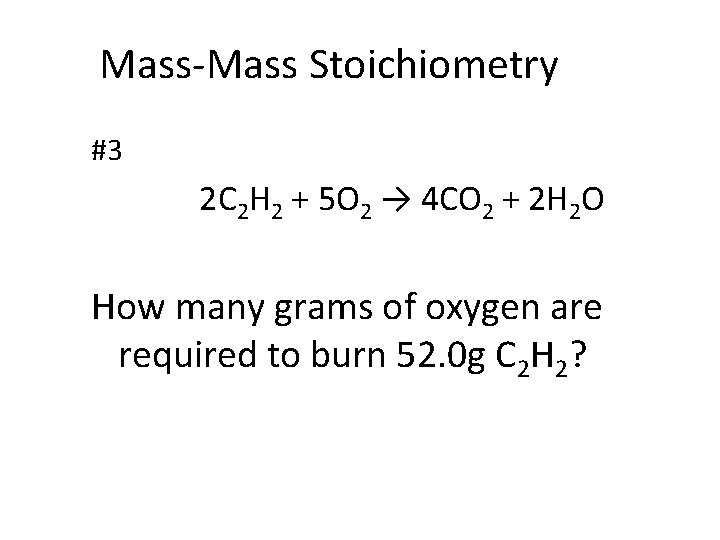
Mass-Mass Stoichiometry #3 2 C 2 H 2 + 5 O 2 → 4 CO 2 + 2 H 2 O How many grams of oxygen are required to burn 52. 0 g C 2 H 2?

Volume-Volume Stoichiometry • Formula to use: Known (L) 1 mol known mol unknown 22. 4 L unknown 1 22. 4 L known mol known 1 mol unknown 1. Write the known and unknown. 2. Use mole and volume conversion factors from Ch. 7 and the mole ratios from the balanced equation to solve.
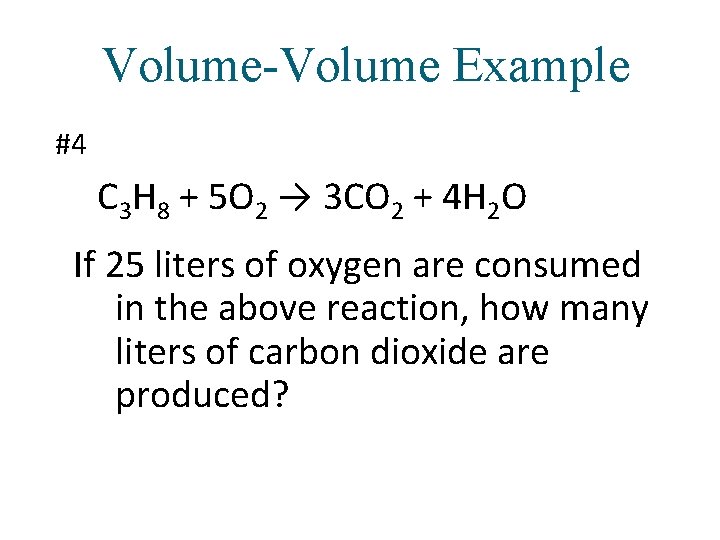
Volume-Volume Example #4 C 3 H 8 + 5 O 2 → 3 CO 2 + 4 H 2 O If 25 liters of oxygen are consumed in the above reaction, how many liters of carbon dioxide are produced?
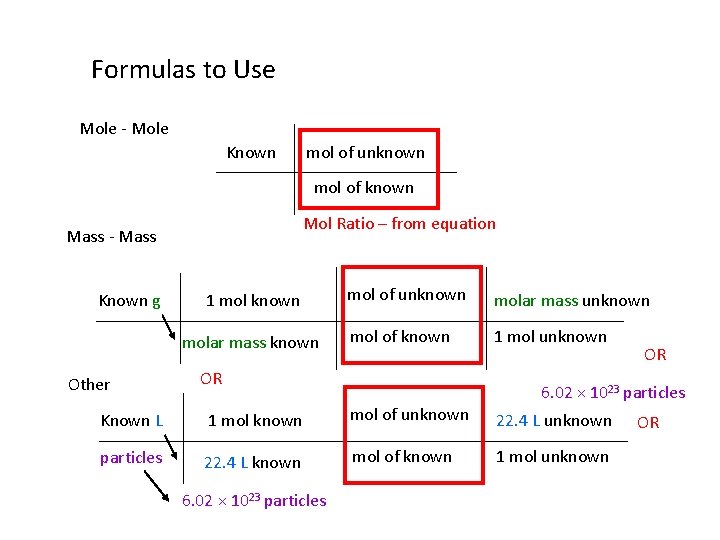
Formulas to Use Mole - Mole Known mol of unknown mol of known Mol Ratio – from equation Mass - Mass Known g 1 mol known molar mass known Other mol of unknown molar mass unknown mol of known 1 mol unknown OR 6. 02 × 1023 particles Known L 1 mol known mol of unknown 22. 4 L unknown particles 22. 4 L known mol of known 1 mol unknown 6. 02 × 1023 particles OR OR
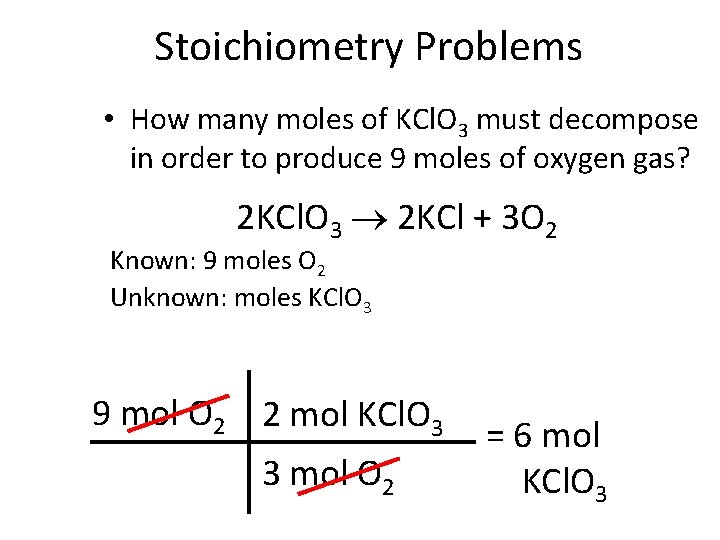
Stoichiometry Problems • How many moles of KCl. O 3 must decompose in order to produce 9 moles of oxygen gas? 2 KCl. O 3 2 KCl + 3 O 2 Known: 9 moles O 2 Unknown: moles KCl. O 3 9 mol O 2 2 mol KCl. O 3 3 mol O 2 = 6 mol KCl. O 3

Stoichiometry Problems • How many grams of KCl. O 3 are required to produce 9. 00 L of O 2 at STP? 2 KCl. O 3 2 KCl + 3 O 2 Known: 9. 00 L O 2 Unknown: g KCl. O 3 9. 00 L O 2 1 mol 2 mol 122 g O 2 KCl. O 3 = 32. 68 22. 4 L 3 mol 1 mol g O 2 KCl. O 3
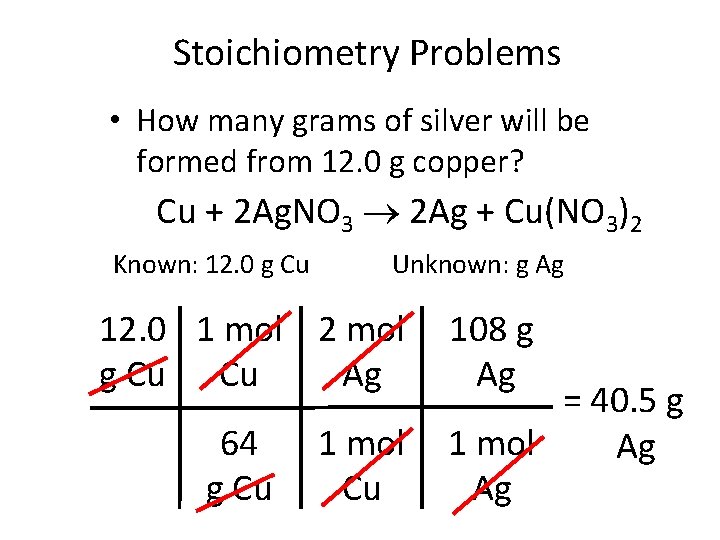
Stoichiometry Problems • How many grams of silver will be formed from 12. 0 g copper? Cu + 2 Ag. NO 3 2 Ag + Cu(NO 3)2 Known: 12. 0 g Cu Unknown: g Ag 12. 0 1 mol 2 mol g Cu Cu Ag 64 g Cu 1 mol Cu 108 g Ag = 40. 5 g 1 mol Ag Ag
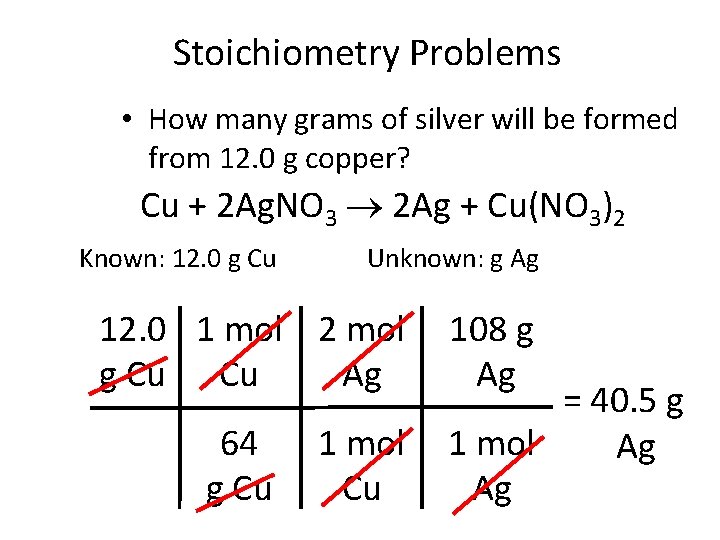
Stoichiometry Problems • How many grams of silver will be formed from 12. 0 g copper? Cu + 2 Ag. NO 3 2 Ag + Cu(NO 3)2 Known: 12. 0 g Cu Unknown: g Ag 12. 0 1 mol 2 mol g Cu Cu Ag 64 g Cu 1 mol Cu 108 g Ag = 40. 5 g 1 mol Ag Ag

Limiting Reactants/Reagents • Available Ingredients – 4 slices of bread – 1 jar of peanut butter – 1/2 jar of jelly n Limiting Reactant/Reagents n bread n Excess Reactants/Reagents n peanut butter and jelly

Limiting Reactants/Reagents • Limiting Reactant/Reagent – used up in a reaction – determines the amount of product • Excess Reactant/Reagent – added to ensure that the other reactant is completely used up – cheaper & easier to recycle

To Determine Limiting Reagents 1. Write a balanced equation. 2. For each reactant, calculate the amount of product formed. 3. Smaller answer indicates: – limiting reactant – actual amount of product

Limiting Reagents 79. 1 g of zinc react with 0. 90 L of HCl. Identify the limiting and excess reactants. How many liters of hydrogen are formed at STP? Zn + 2 HCl 79. 1 g 0. 90 L Zn. Cl 2 + H 2 ? L
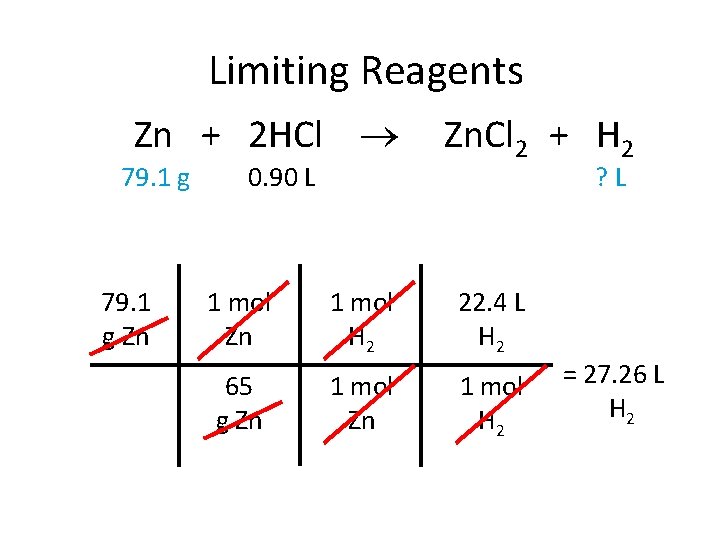
Limiting Reagents Zn + 2 HCl 79. 1 g Zn 0. 90 L Zn. Cl 2 + H 2 ? L 1 mol Zn 1 mol H 2 22. 4 L H 2 65 g Zn 1 mol H 2 = 27. 26 L H 2
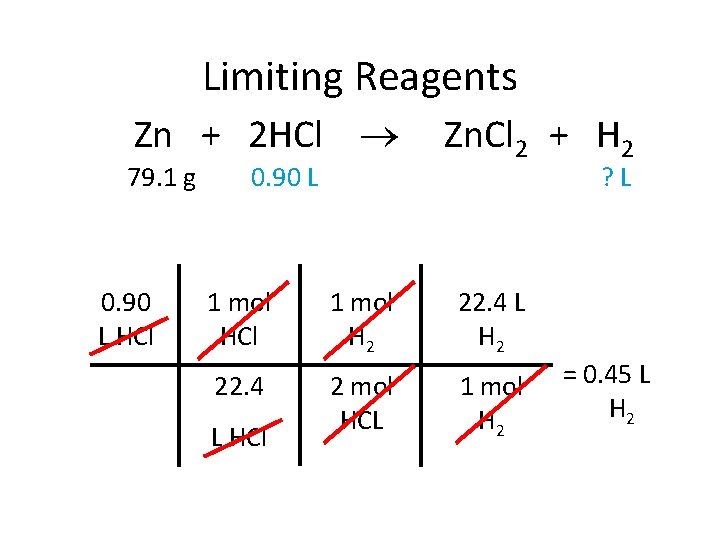
Limiting Reagents Zn + 2 HCl 79. 1 g 0. 90 L HCl 0. 90 L Zn. Cl 2 + H 2 ? L 1 mol HCl 1 mol H 2 22. 4 L H 2 22. 4 2 mol HCL 1 mol H 2 L HCl = 0. 45 L H 2
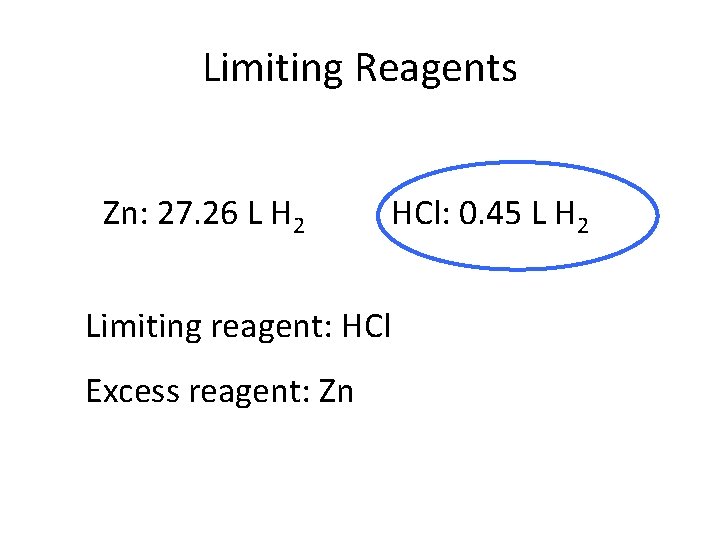
Limiting Reagents Zn: 27. 26 L H 2 Limiting reagent: HCl Excess reagent: Zn HCl: 0. 45 L H 2

Percent Yield • Percent yield- the ratio of the actual yield to theoretical yield • Actual yield- the amount of product formed when a reaction is carried out in the laboratory • Theoretical yield- the calculated amount of product formed during a reaction (mathematical calculation used to make answer keys)
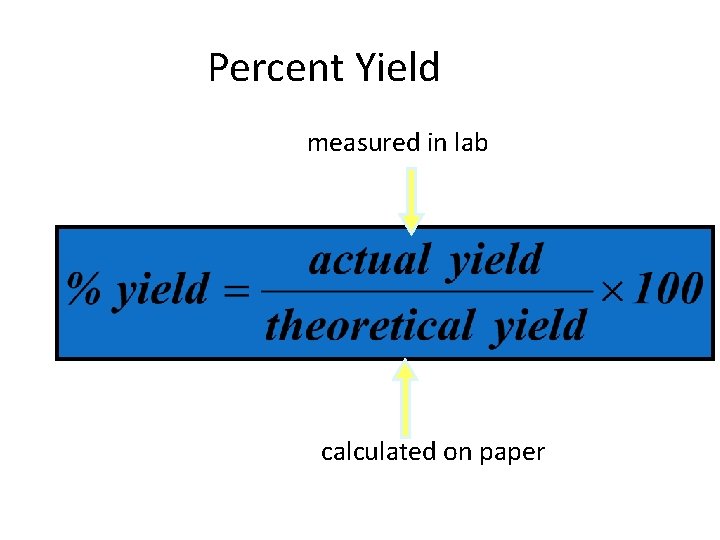
Percent Yield measured in lab calculated on paper

Percent Yield • When 45. 8 g of K 2 CO 3 react with excess HCl, 46. 3 g of KCl are formed. Calculate theoretical and % yields of KCl. K 2 CO 3 + 2 HCl 2 KCl + H 2 O + CO 2 45. 8 g ? g actual: 46. 3 g

B. Percent Yield K 2 CO 3 + 2 HCl 2 KCl + H 2 O + CO 2 45. 8 g ? g actual: 46. 3 g Theoretical Yield: 45. 8 g K 2 CO 3 1 mol K 2 CO 3 138 g K 2 CO 3 2 mol KCl 74 g KCl = 49. 12 1 mol g KCl K 2 CO 3 KCl

B. Percent Yield K 2 CO 3 + 2 HCl 2 KCl + H 2 O + CO 2 45. 8 g 49. 12 g actual: 46. 3 g Theoretical Yield = 49. 12 g KCl % Yield = 46. 3 g 49. 12 g 100 = 94. 3%
- Slides: 35