Mole Questions And Dimensional Analysis OUTCOME QUESTIONS C


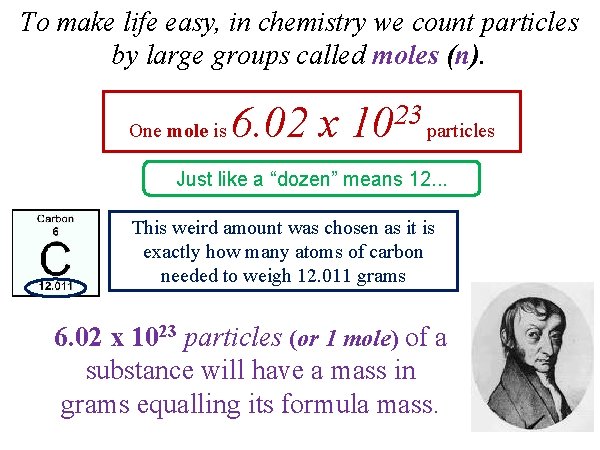


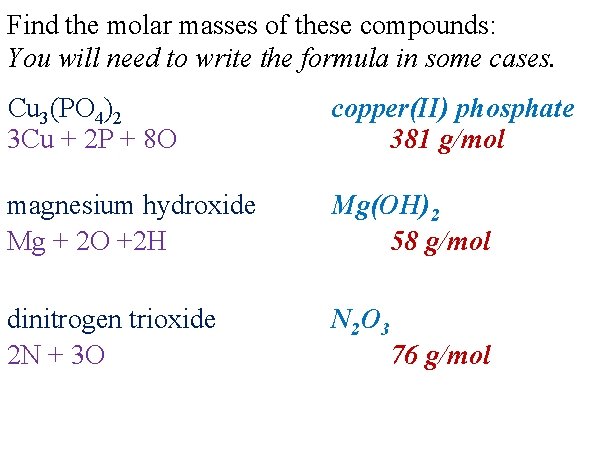






- Slides: 19

Mole Questions And Dimensional Analysis

OUTCOME QUESTION(S): C 11 -3 -12 MOLAR CALCULATIONS Understand the importance of the concept of the mole and calculate the molar mass of various substances. Interpret a balanced equation in terms of moles, mass, and volumes of gases. Include: molar volume Solve problems requiring interconversions between moles, mass, volume, and number of particles. Include: density Vocabulary & Concepts Avogadro’s number
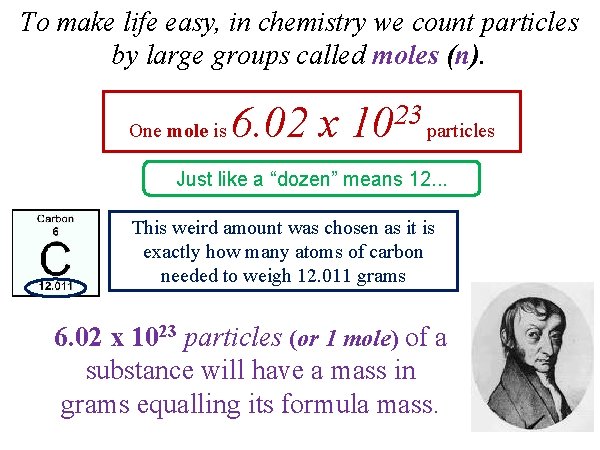
To make life easy, in chemistry we count particles by large groups called moles (n). One mole is 6. 02 x 23 10 particles Just like a “dozen” means 12. . . This weird amount was chosen as it is exactly how many atoms of carbon needed to weigh 12. 011 grams 6. 02 x 1023 particles (or 1 mole) of a substance will have a mass in grams equalling its formula mass.

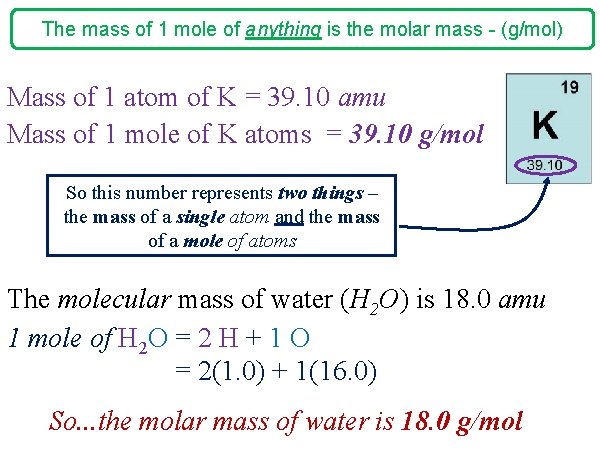
The mass of 1 mole of anything is the molar mass - (g/mol) Mass of 1 atom of K = 39. 10 amu Mass of 1 mole of K atoms = 39. 10 g/mol So this number represents two things – the mass of a single atom and the mass of a mole of atoms The molecular mass of water (H 2 O) is 18. 0 amu 1 mole of H 2 O = 2 H + 1 O = 2(1. 0) + 1(16. 0) So. . . the molar mass of water is 18. 0 g/mol

What is the molar mass of calcium nitrate? Ca(NO 3)2 Breakdown the compound: 1 Ca(NO 3)2 particle = 1 Ca atom + 2 N atoms + 6 O atoms 1 Ca(NO 3)2 mole = 1 Ca mole + 2 N moles + 6 O moles = (40. 0) + 2(14. 0) + 6(16. 0) So the molar mass of Ca(NO 3)2 is 164. 0 g/mol

What is the molar mass of ammonium dichromate? (NH 4)2 Cr 2 O 7 Breakdown the compound: 1 (NH 4)2 Cr 2 O 7 mole = 2 N mol+ 8 H mol + 2 Cr mol + 7 O mol Mole is abbreviated mol or n = 2(14. 0) + 8(1. 0) + 2(52. 0) + 7(16. 0) So the molar mass of (NH 4)2 Cr 2 O 7 is 252. 0 g/mol
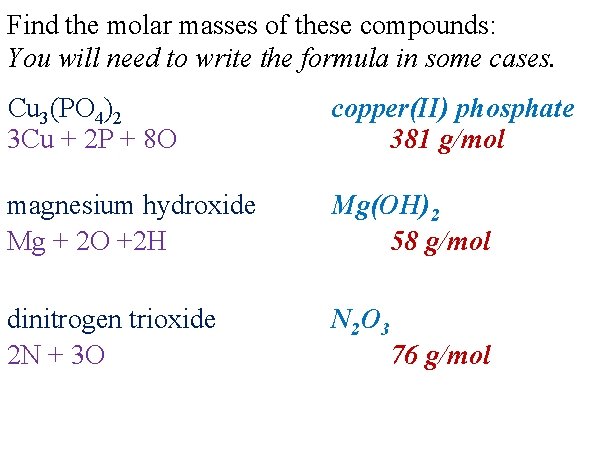
Find the molar masses of these compounds: You will need to write the formula in some cases. Cu 3(PO 4)2 3 Cu + 2 P + 8 O copper(II) phosphate 381 g/mol magnesium hydroxide Mg + 2 O +2 H Mg(OH)2 58 g/mol dinitrogen trioxide 2 N + 3 O N 2 O 3 76 g/mol

One mole is 6. 02 x 23 10 particles We call this Avogadro’s Number (NA) 6. 02 x 10 23 particles or 1 mole 6. 02 x 10 23 particles The term "particle" refers to the known value: atoms, molecules, ions, etc. 6. 02 x 1023 pencils is 1 mole of pencils 6. 02 x 1023 carbon atoms is 1 mole… 6. 02 x 1023 water molecules is 1 mole…

Rules for Dimensional Analysis 1. 2. 3. 4. 5. Start with what you know Identify (or create) the conversion factor Put units you want to cancel on bottom Put units you need on the top Multiply across top, divide across bottom Dimensional Analysis a wayoftopepperoni convert units: How many pizzas would 250 is pieces make? (make the value the same but in different units) 250 pepperoni 1 pizza 19 pepperoni = 13. 2 Pizzas Some people simplify this notation with added bars – called “factor label line”

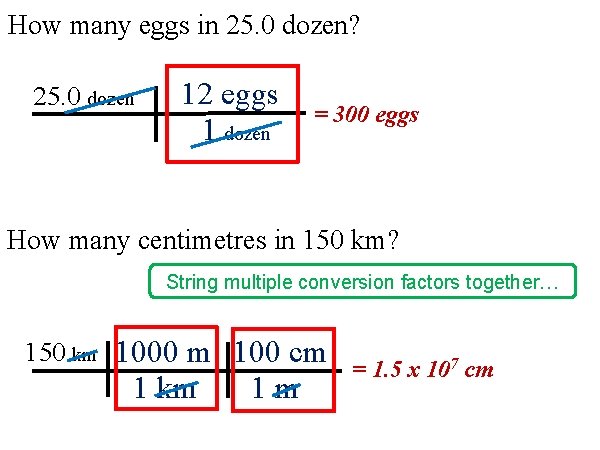
How many eggs in 25. 0 dozen? 25. 0 dozen 12 eggs 1 dozen = 300 eggs How many centimetres in 150 km? String multiple conversion factors together… 150 km 1000 m 100 cm 1 km 1 m = 1. 5 x 107 cm

How many atoms in 25. 0 moles of copper? atoms are the “particle” 25. 0 Cu mol 6. 02 x 1023 particles 1 mol = 1. 51 x 1025 Cu atoms How many molecules of water in 1. 50 x 10– 5 mol? molecules are the “particle” 1. 50 x 10 -5 H 2 O mol 6. 02 x 1023 particles 1 mol = 9. 03 x 1018 H 2 O molecules

How many moles is 5 atoms of zinc? atoms are the “particle” 5 Zn atoms 1 mol 6. 02 x 1023 particles = 8. 31 x 10 -24 Zn mol Note: all conversion factors can be flipped “upside down” depending on the need 6. 02 x 10 23 particles or 1 mole 6. 02 x 10 23 particles So 5 atoms of Zinc is the same as 8. 31 x 10 -24 moles

What is the mass of 1. 20 x 10– 5 moles of carbon tetrachloride, CCl 4? Now lets add the CCl 4 = = = 1 C + 4 Cl 1(12. 0) + 4(35. 5) 154. 0 g/mol 154. 0 g 1 mol or 1. 20 x 10 -5 mol CCl 4 1 mol 154. 0 g 1 molar mass to our dimensional analysis work… 1. 2. 3. 4. 5. What you know Add conversion factor Units to cancel on bottom Units in need on the top Multiply top, divide bottom = 1. 85 x 10 -3 g

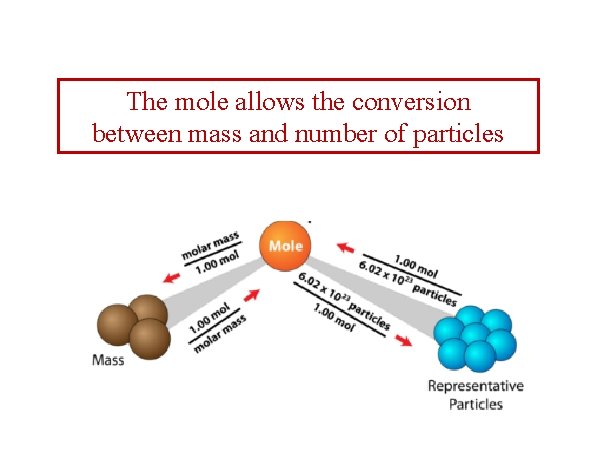
The mole allows the conversion between mass and number of particles

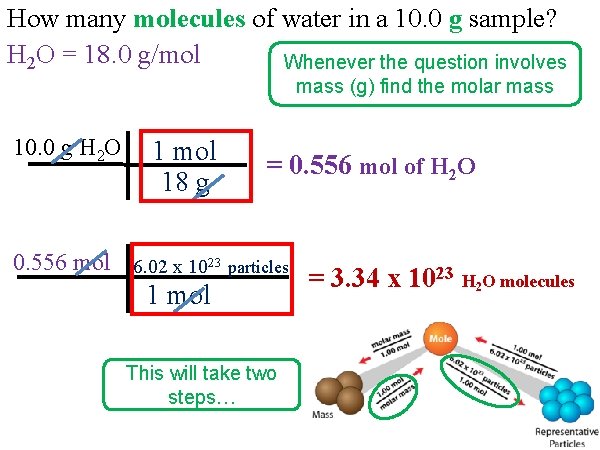
How many molecules of water in a 10. 0 g sample? H 2 O = 18. 0 g/mol Whenever the question involves mass (g) find the molar mass 10. 0 g H 2 O 1 mol 18 g = 0. 556 mol of H 2 O 0. 556 mol 6. 02 x 1023 particles 1 mol This will take two steps… = 3. 34 x 1023 H 2 O molecules

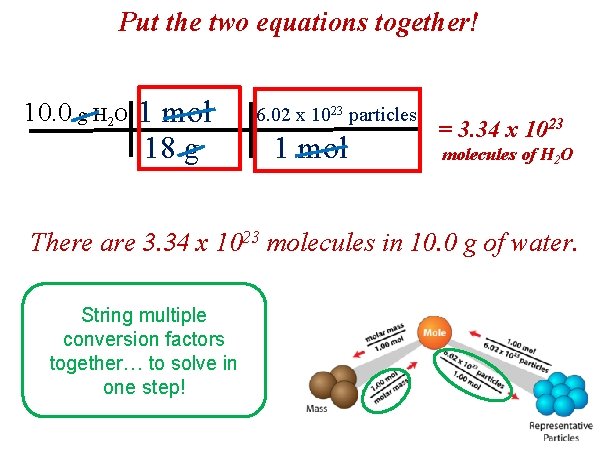
Put the two equations together! 10. 0 g H 2 O 1 mol 18 g 6. 02 x 1023 particles 1 mol = 3. 34 x 1023 molecules of H 2 O There are 3. 34 x 1023 molecules in 10. 0 g of water. String multiple conversion factors together… to solve in one step!

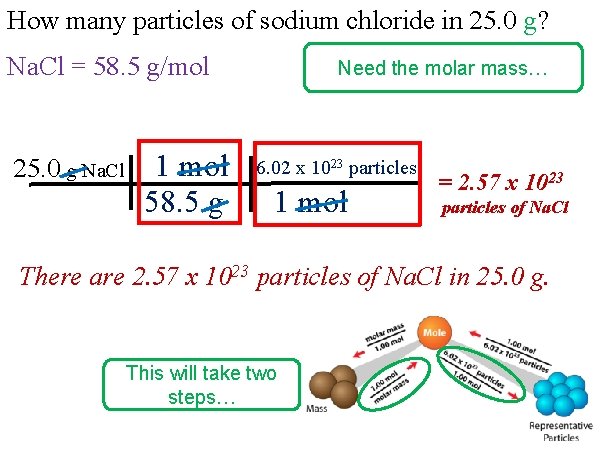
How many particles of sodium chloride in 25. 0 g? Na. Cl = 58. 5 g/mol 25. 0 g Na. Cl 1 mol 58. 5 g Need the molar mass… 6. 02 x 1023 particles 1 mol = 2. 57 x 1023 particles of Na. Cl There are 2. 57 x 1023 particles of Na. Cl in 25. 0 g. This will take two steps…

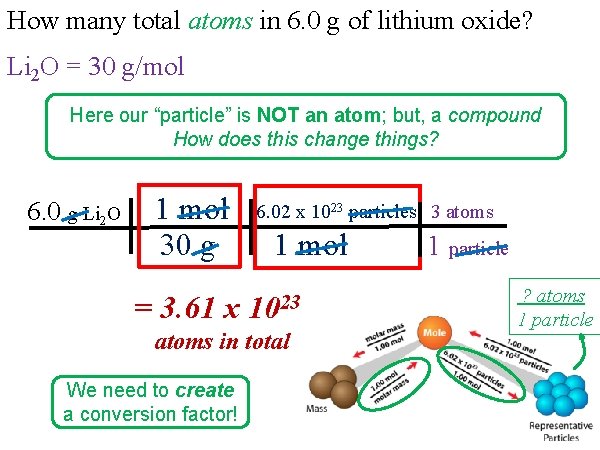
How many total atoms in 6. 0 g of lithium oxide? Li 2 O = 30 g/mol Here our “particle” is NOT an atom; but, a compound How does this change things? 6. 0 g Li 2 O 1 mol 30 g = 3. 61 x 6. 02 x 1023 particles 3 atoms 1 mol 1023 atoms in total We need to create a conversion factor! 1 particle ? atoms 1 particle

CAN YOU / HAVE YOU? C 11 -3 -12 MOLAR CALCULATIONS Understand the importance of the concept of the mole and calculate the molar mass of various substances. Interpret a balanced equation in terms of moles, mass, and volumes of gases. Include: molar volume Solve problems requiring interconversions between moles, mass, volume, and number of particles. Include: density Vocabulary & Concepts Avogadro’s number