Mole Calculations Mole Calculations Symbol n m M

Mole Calculations
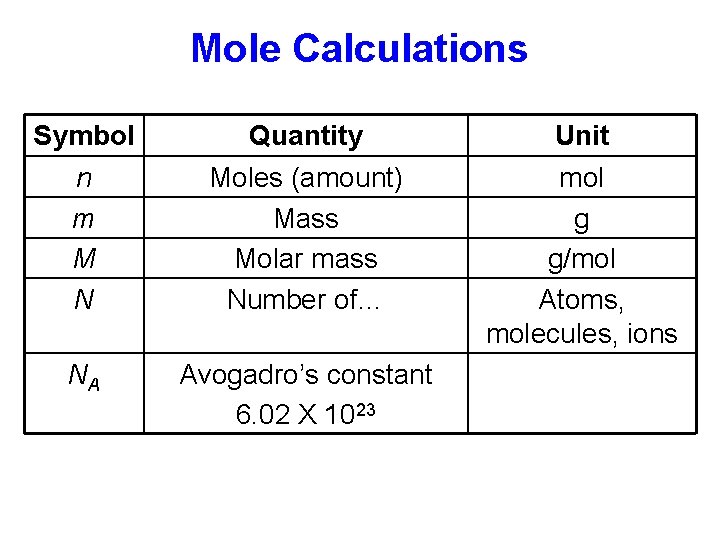
Mole Calculations Symbol n m M N Quantity Moles (amount) Mass Molar mass Number of… NA Avogadro’s constant 6. 02 X 1023 Unit mol g g/mol Atoms, molecules, ions
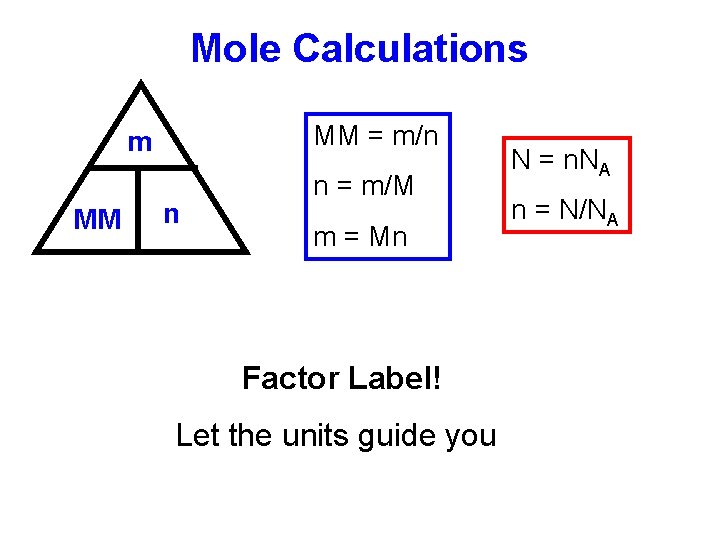
Mole Calculations MM = m/n m MM n n = m/M m = Mn Factor Label! Let the units guide you N = n. NA n = N/NA

Determining Molar Mass Recall: Add the individual molar masses MM(NH 4)3 PO 4 = = 3(N + 4 H) + P + 4 O 3 N + 12 H + P + 4 O 3(14. 01) + 12(1. 01) + 30. 97 + 4(16. 00) 149. 12 g/mol

MM = m/n n = m/M m = Mn Converting Mass to Moles Example: How many moles of calcium carbonate are there in 1. 5 kg? n = m/MM Given: = 1500 g 100. 09 g/mol m Ca. CO 3 = 1500 g = 15 mol MM = 40. 08 + 12. 01 + (3 X 16. 00) = 100. 09 g/mol n = 1500 g X 1 mol 100. 09 g = 15 mol

MM = m/n n = m/M m = Mn Converting Moles to Mass Example: What is the mass in grams of 0. 346 mol of sodium sulfate? m = MMn Given: n Na 2 SO 4 = 0. 346 mol MM = (2 X 22. 99) + 32. 06 + (4 X 16. 00) = 142. 04 g/mol = 142. 04 g X 0. 346 mol 1 mol = 49. 1 g

N = n. NA Finding Number of Atoms or Molecules How many atoms of copper are there in 2. 0 moles? Given: n Cu = 2. 0 mol NA = 6. 02 X 1023 N = n. NA = 2. 0 mol X 6. 02 X 1023 atoms 1 mol = 1. 2 X 1024 atoms of copper
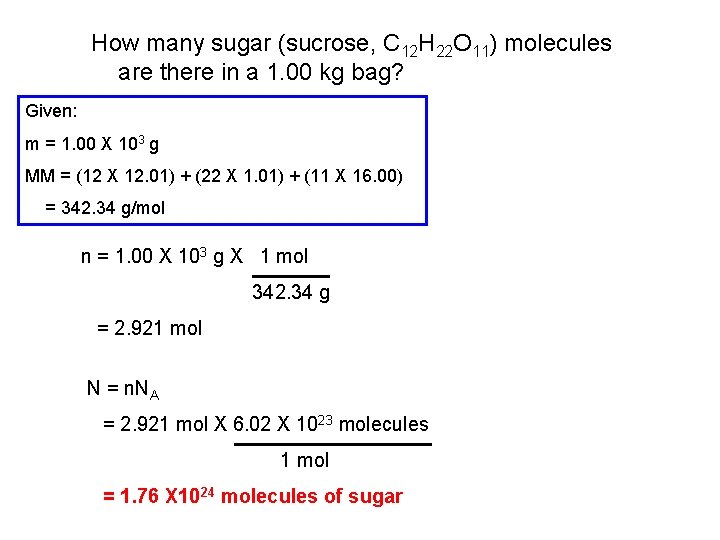
How many sugar (sucrose, C 12 H 22 O 11) molecules are there in a 1. 00 kg bag? Given: m = 1. 00 X 103 g MM = (12 X 12. 01) + (22 X 1. 01) + (11 X 16. 00) = 342. 34 g/mol n = 1. 00 X 103 g X 1 mol 342. 34 g = 2. 921 mol N = n. NA = 2. 921 mol X 6. 02 X 1023 molecules 1 mol = 1. 76 X 1024 molecules of sugar

How many carbon atoms are there in a 1. 00 kg bag of sugar (sucrose, C 12 H 22 O 11) ? N = 1. 76 X 1024 molecules of sugar X 12 carbon atoms 1 molecule sugar = 2. 11 X 1025 carbon atoms
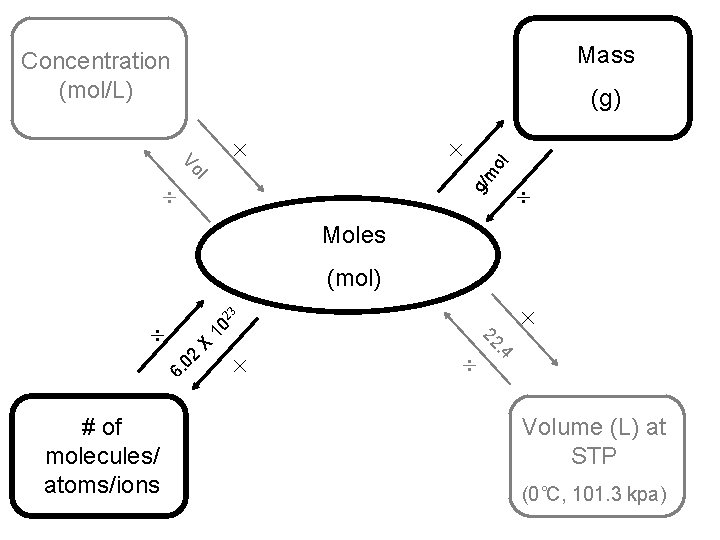
Mass Concentration (mol/L) l g/ m Vo ol (g) Moles 23 (mol) 10 X 02 6. . 4 # of molecules/ atoms/ions 22 Volume (L) at STP (0˚C, 101. 3 kpa)

Homework • Finish Mole Worksheet – Change “volume” to “mass” in Q 3 • P. 170 #7 -13 • P. 171 #1 -5 • P. 172 -173#1 -8
- Slides: 11