Mole a fixed amount of substance n SI

Mole • = a fixed amount of substance, n • = SI unit, mol • = 6. 02 x 1023 • How many particles are in 3 moles, 0. 5 moles and 0. 001 moles? • How many moles are 6. 02 x 1024 electrons and 3. 01 x 1021 ions?

one mole of a substance

Molar ratios • If there are 2 x 1022 C-atoms in a sample of ethane, C 2 H 6, how many hydrogen atoms are there in that same sample? • If you have 4. 5 x 1022 atoms of hydrogen in a sample of ammonia, how many molecules of ammonia do you have? • How many moles of oxygen are there in 2 moles of Al 2 O 3? • How many moles of NO 3 - are there in 1 mole of Ga(NO 3)3? • What is the total number of atoms in 0. 05 moles of NH 4 NO 3?

The mole concept • Find the mass of 1 molecule of ethanol, C 2 H 5 OH. • Find the number of carbon atoms in 0. 1 mole of C 2 H 5 OH. • Which sample contains the greatest number of particles? 5 g of NH 3 5 g of H 2 O 5 g of Ca. O?
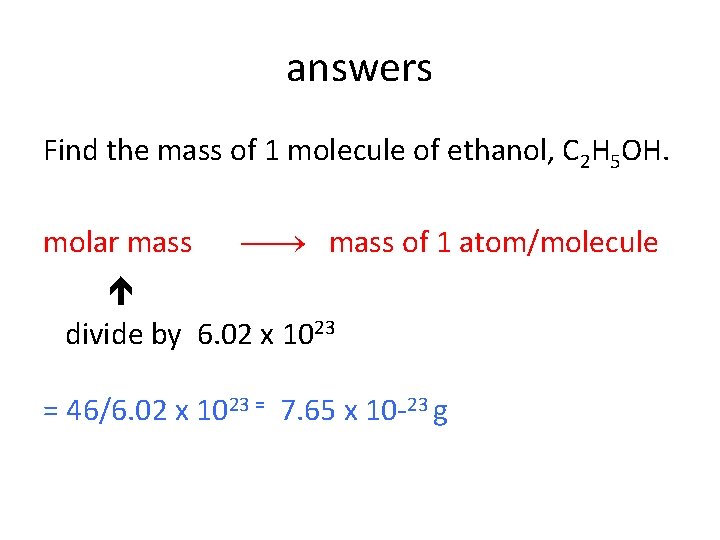
answers Find the mass of 1 molecule of ethanol, C 2 H 5 OH. molar mass of 1 atom/molecule divide by 6. 02 x 1023 = 46/6. 02 x 1023 = 7. 65 x 10 -23 g
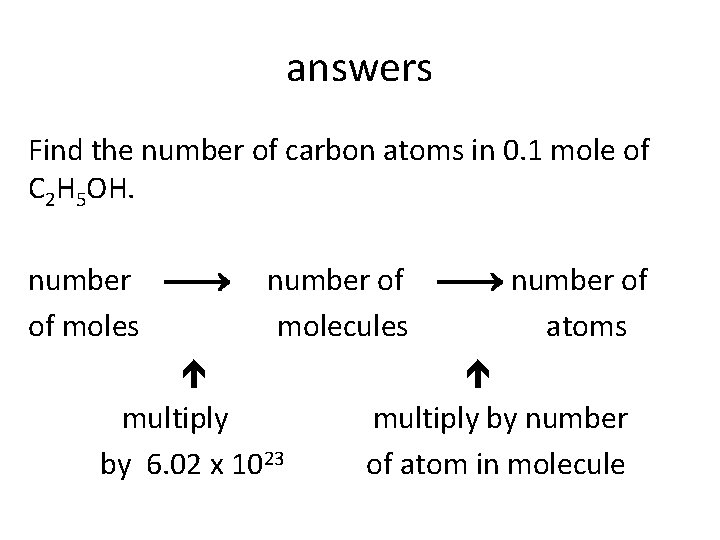
answers Find the number of carbon atoms in 0. 1 mole of C 2 H 5 OH. number of of moles molecules atoms multiply by number by 6. 02 x 1023 of atom in molecule

answer • Moles NH 3 = 5 g/ 17. 04 g = 0. 29 • Moles of H 2 O = 5 g /18. 01 g = 0. 28 • Moles Ca. O = 5 g / 56. 08 g = 0. 09
- Slides: 7