Molarity Problems Molarity molL Molarity moles of solute
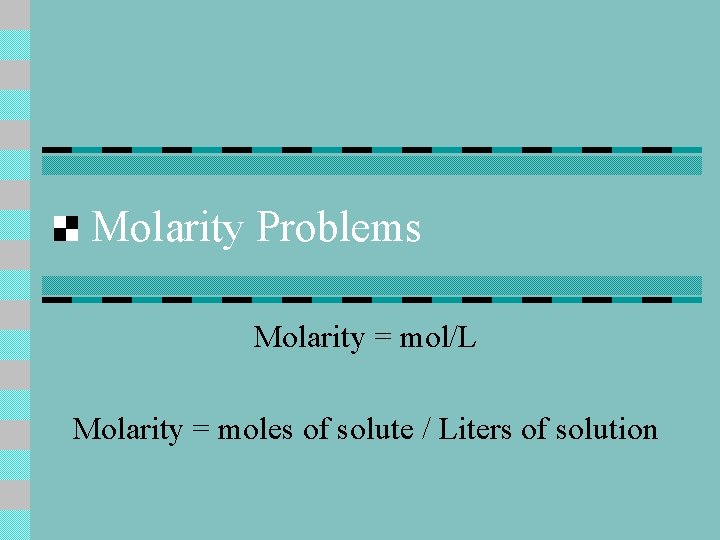
Molarity Problems Molarity = mol/L Molarity = moles of solute / Liters of solution
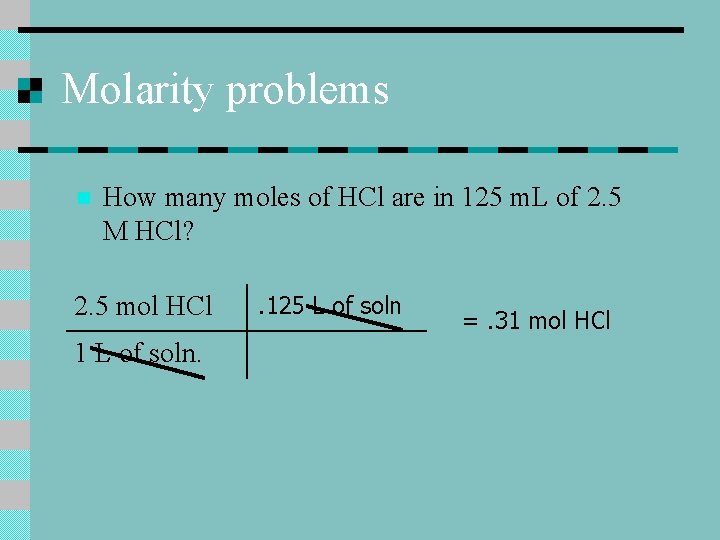
Molarity problems n How many moles of HCl are in 125 m. L of 2. 5 M HCl? 2. 5 mol HCl 1 L of soln. . 125 L of soln =. 31 mol HCl
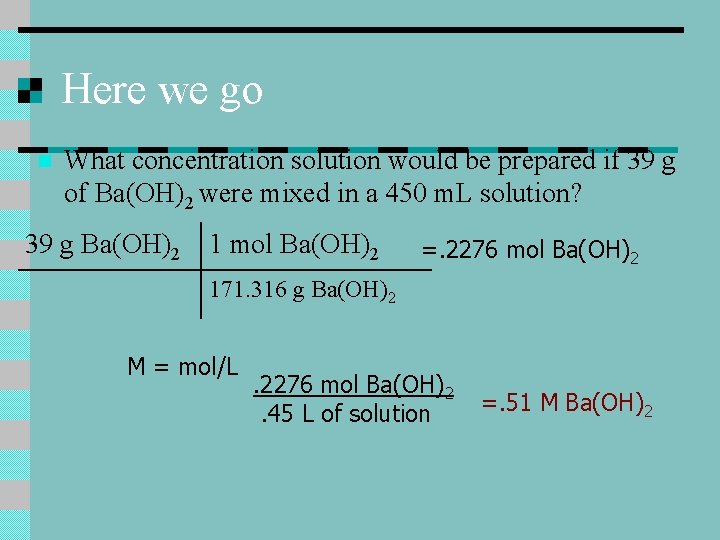
Here we go n What concentration solution would be prepared if 39 g of Ba(OH)2 were mixed in a 450 m. L solution? 39 g Ba(OH)2 1 mol Ba(OH)2 =. 2276 mol Ba(OH)2 171. 316 g Ba(OH)2 M = mol/L . 2276 mol Ba(OH)2. 45 L of solution =. 51 M Ba(OH)2

More For a lab in this chapter, I need to make. 60 L of 3. 0 M Na. OH, what mass of Na. OH did I need? n. 6 L x 3. 0 M Na. OH = 1. 8 mol Na. OH n 1. 8 mol Na. OH x 39. 998 g/mol n = 72 g Na. OH n

Molarity Problems n A 0. 24 M solution of Na 2 SO 4 contains 0. 36 moles of Na 2 SO 4. How many liters were required to make this solution? 0. 36 mol Na 2 SO 4 = 1. 5 L Na 2 SO 4 1 L soln 0. 24 mol

Getting tougher + Ba. Cl 2 2 Ag. Cl + Ba(NO 3)2 n Balance the equation. If 1. 2 L of. 50 M Ag. NO 3 is reacted completely, what molarity solution of Ba(NO 3)2 will be created if the volume increased to 1. 5 L? n 2 Ag. NO 3 1. 2 L x. 5 M Ag. NO 3 =. 6 mol Ag. NO 3 1 mol Ba(NO 3)2 =. 3 mol Ba(NO 3)2 2 mol Ag NO 3 1. 5 L =. 20 M Ba(NO 3)2
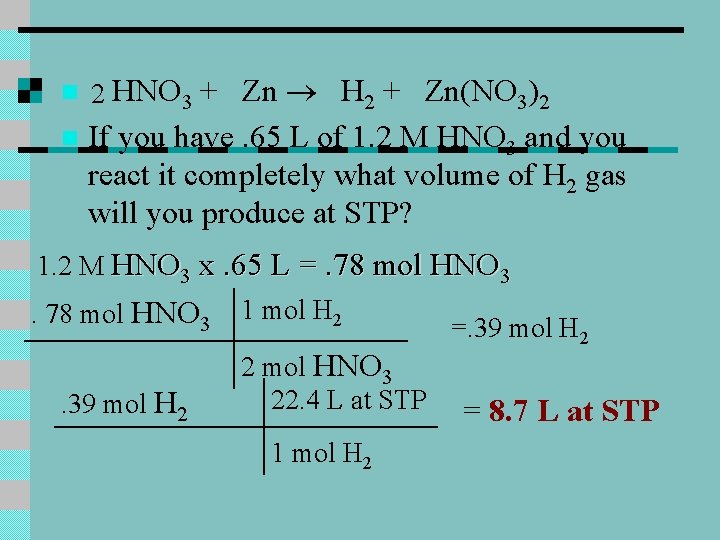
+ Zn H 2 + Zn(NO 3)2 n If you have. 65 L of 1. 2 M HNO 3 and you react it completely what volume of H 2 gas will you produce at STP? n 2 HNO 3 1. 2 M HNO 3 x. 65 L =. 78 mol HNO 3. 39 mol H 2 1 mol H 2 2 mol HNO 3 22. 4 L at STP 1 mol H 2 =. 39 mol H 2 = 8. 7 L at STP

+ Zn H 2 + Zn(NO 3)2 n If you have. 65 L of 1. 2 M HNO 3 and you react it completely, what conc. of Zn(NO 3)2 will be left if the volume increases to. 75 L? n 2 HNO 3 1. 2 M HNO 3 x. 65 L =. 78 mol HNO 3. 78 mol HNO 3 1 mol Zn(NO 3)2 =. 39 mol Zn(NO 3)2 2 mol HNO 3 =. 52 M Zn(NO 3)2 . 75 L

Fe + 3 H 2 SO 4 Fe 2(SO 4)3 + 3 H 2 n If 350 m. L of 2. 3 M H 2 SO 4 is completely reacted, what is the volume of hydrogen gas produced at 24 o C and 114 k. Pa? . 35 L x 2. 3 M =. 805 mol H 2 SO 4 n 2 . 805 mol H 2 SO 4 1 mol H 2 SO 4 =. 805 mol H 2 PV = n. RT 114 k. Pa V =. 805 mol (8. 31) 297 K =17 L H 2
- Slides: 9