Molarity Concentration Based on Mass Concentration amount of

Molarity

Concentration Based on Mass Concentration - amount of solute dissolved in a given amount of solution l Concentration of a solution has an effect on l l Physical properties l Melting l and boiling points Chemical properties l Solution reactivity

Moles and Equivalents Chemical equations represent the relative number of moles of reactants producing products l Many chemical reactions occur in solution where it is most useful to represent concentrations on a molar basis l

Molarity The most common mole-based concentration unit is molarity l Molarity l Symbolized M l Defined as the number of moles of solute per liter of solution l
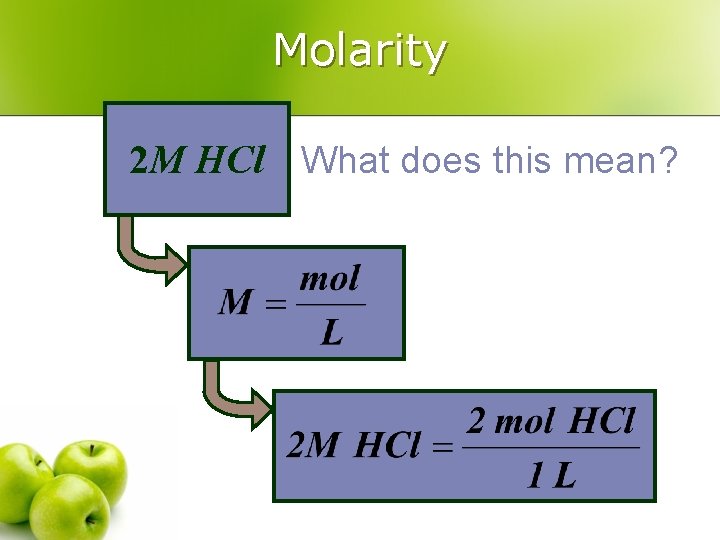
Molarity 2 M HCl What does this mean?
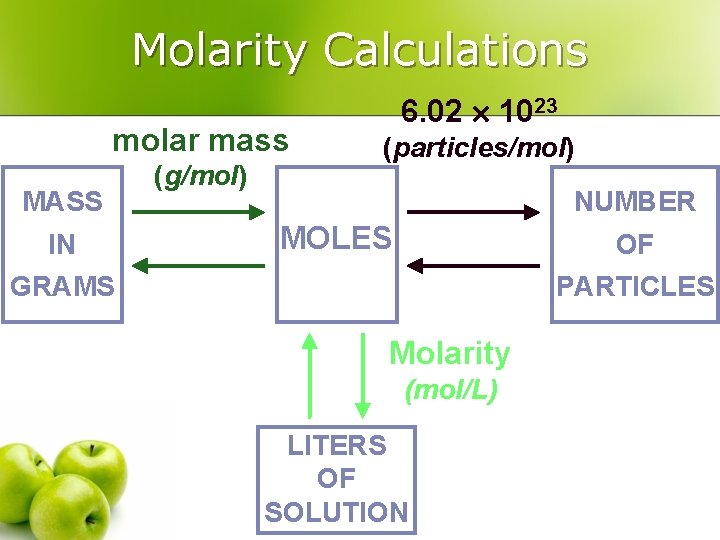
Molarity Calculations molar mass MASS IN (g/mol) 6. 02 1023 (particles/mol) NUMBER MOLES OF GRAMS PARTICLES Molarity (mol/L) LITERS OF SOLUTION

Calculating Molarity from Moles Calculate the molarity of 2. 0 L of solution containing 5. 0 mol Na. OH l Use the equation l l Substitute into the equation: MNa. OH = 5. 0 mol solute 2. 0 L solution = 2. 5 M

Molarity Calculations l How many grams of Na. Cl are required to make 0. 500 L of 0. 25 M Na. Cl? 0. 500 L 0. 25 mol 58. 44 g 1 L 1 mol = 7. 3 g Na. Cl

Molarity Calculations l Find the molarity of a 250 m. L solution containing 10. 0 g of Na. F. 10. 0 g 1 mol 41. 99 g M= 0. 238 mol 0. 25 L = 0. 238 mol Na. F = 0. 95 M Na. F

Molar Volume of a Gas Molar volume - the volume occupied by 1 mol of any gas l STP – Standard Temperature and Pressure l T = 273 K (or 0 o. C) l P = 1 atm l l At STP the molar volume of any gas is 22. 4 L l Vgas = molesgas x (22. 4 L/1 mol)

Molar Mass and Density l l A gas’s density will determine if it will sink or float in the air Molar mass = density x molar volume l At STP
- Slides: 11