Molar Mass Do Now You have a bag

Molar Mass

Do Now • You have a bag of blue and red marbles. You’re trying to figure out the mass of the bag, but you don’t have a balance/ scale. • What might you need to know to solve this problem? How might you be able to find the mass?

Do Now- con’t. • Your bag contains 10 blue marbles and 15 red marbles. • What else may we need to know?

Do Now- con’t. • Your bag contains 10 blue marbles and 15 red marbles. • Each blue marble weighs 2 grams, and each red marbles weighs 3 grams. • What is the mass of the bag?

Mass on the Molecular Level • Just like we weren’t able to directly weigh the bag of marbles, it’s impossible to “weigh” one molecule. – But, we can find the mass of a molecule by adding up the mass of its individual atoms • Molar Mass: the mass (in grams) of one mole of molecules – This is based on the atomic mass in the periodic table

Determining Molar Mass 1. From the formula, write down each of the different elements that are present. Mg. Cl 2: Mg Cl 2. Identify how many atoms of each element are present using the subscripts (no subscript = just one atom) Mg: 1 Cl: 2 3. Multiply the number of atoms of each element by that element’s atomic mass, then find the total mass by adding all the elements’ masses together Mg: 1 x 24. 3050 g = 24. 3050 g Cl: 2 x 35. 4527 g = + 70. 9054 g 95. 2104 g

Practice! • Determine the molar mass of H 2 SO 4 (sulfuric acid).

Practice! • Determine the molar mass of H 2 SO 4 (sulfuric acid). H 2 SO 4 H: 2 x 1. 0079 g = 2. 0158 g S: 1 x 32. 066 g = 32. 066 g O: 4 x 15. 999 = 63. 996 g 98. 0778 g

Practice! • What is the molar mass of (H 3 O)3 PO 4?

Practice! • What is the molar mass of (H 3 O)3 PO 4? • Note: when you have parentheses in the formula, the subscript outside them is distributed (multiplied) by all of the subscripts inside!

Practice! • What is the molar mass of (H 3 O)3 PO 4? H: (3 x 3) = 9 O: (1 x 3) + 4 = 7 P: 1 H: 9 x 1. 0079 = 9. 0711 O: 7 x 15. 999 = 111. 993 P: 1 x 30. 974 = 30. 974 152. 038 g

What exactly is a mole? • Avogadro’s Number: 6. 022 x 1023 • The number of carbon atoms in exactly 12 g of pure 12 C • 1 mole of ANYTHING = 6. 022 x 1023 units of that substance • How many teachers are in 1 mole of teachers? • How many pencils are in 1 mole of pencils? • How many candy bars are in 1 mole of candy bars?

What exactly is a mole? • Avogadro’s Number: 6. 022 x 1023 • The number of carbon atoms in exactly 12 g of pure 12 C • 1 mole of ANYTHING = 6. 022 x 1023 units of that substance • How many teachers are in 1 mole of teachers? • How many pencils are in 1 mole of pencils? • How many candy bars are in 1 mole of candy bars? 6. 022 x 1023!!!

If we know moles… • The molar mass is the grams of that substance that it takes to make up 1 mole • So, if we know how many moles we started with, we can determine the mass of our substance • OR, • We also know the number of particles in one mole (Avogadro’s number) • So, we can determine the number of molecules of our substance
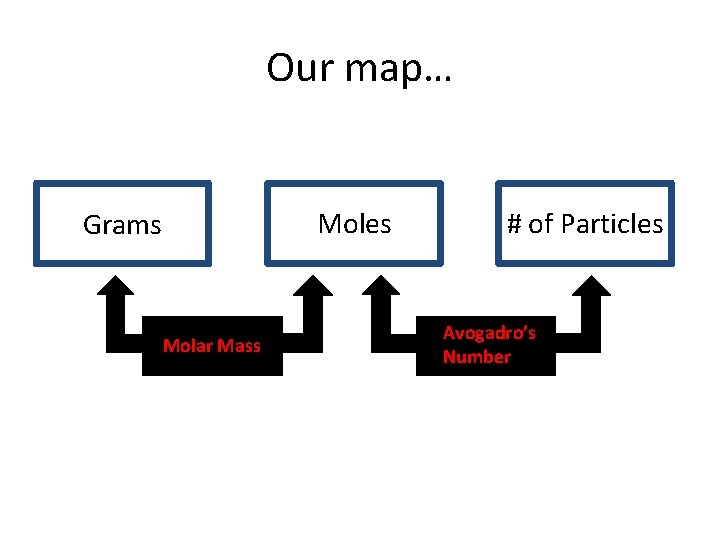
Our map… Moles Grams Molar Mass # of Particles Avogadro’s Number

Going between Grams and Moles We have 24 g of carbon. We know that the molar mass is 12. 011 g/mol (there are 12. 011 grams of carbon in one mole) • SET UP THE CONVERSION! Start with what you know. The molar mass is our conversion factor.

Going between Grams and Moles We have 24 g of carbon. We know that the molar mass is 12. 011 g/mol (there are 12. 011 grams of carbon in one mole) • SET UP THE CONVERSION! Start with what you know. The molar mass is our conversion factor. 24 g C x 1 mole = 24 g C mol = 1. 998 mol C 1 12. 011 g C

Going between Grams and Moles • We have 3 moles of sulfur. How many grams do we have?

Going between Grams and Moles • We have 3 moles of sulfur. How many grams do we have? 3 mol S x 32. 066 g S = 96. 198 g S 1 1 mol S

Higher Order Questions • What is another real-life application of stoichiometry? • What is happening on a molecular level when two substances react to form new products? THINK-WRITE-PAIR-SHARE! Pick one topic to contemplate with your partner.
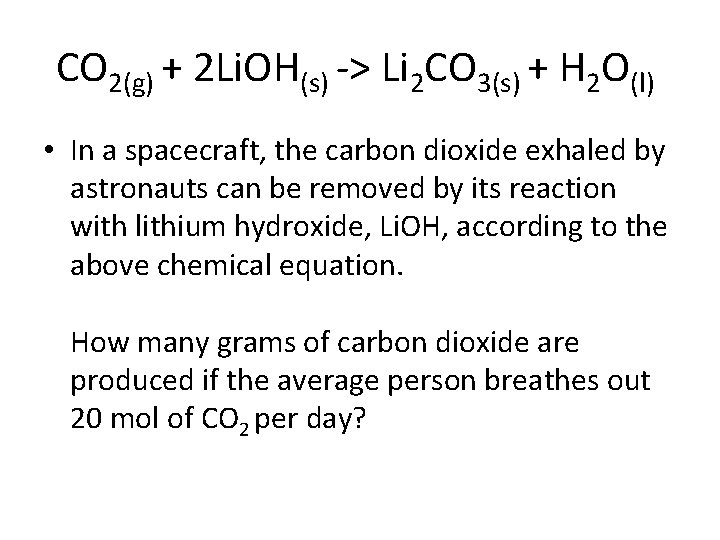
CO 2(g) + 2 Li. OH(s) -> Li 2 CO 3(s) + H 2 O(l) • In a spacecraft, the carbon dioxide exhaled by astronauts can be removed by its reaction with lithium hydroxide, Li. OH, according to the above chemical equation. How many grams of carbon dioxide are produced if the average person breathes out 20 mol of CO 2 per day?

You Practice! Convert from grams to moles: • 125 g phosphorus (P) • 75 g Na. Cl Convert from moles to grams: • 5 moles of carbon (C) • 7 moles of HBr

You Practice! Convert from grams to moles: • 125 g phosphorus (P) = 4. 04 mol P • 75 g Na. Cl = 1. 28 mol Na. Cl Convert from moles to grams: • 5 moles of carbon (C) = 60. 055 g C • 7 moles of HBr = 566. 4 g HBr

Going Between Moles and Particles • We have 15 x 1023 carbon atoms. How many moles is this? • SET UP THE CONVERSION! Start with what you know. Avogadro’s number is our conversion factor.

Going Between Moles and Particles • We have 15 x 1023 carbon atoms. How many moles is this? • SET UP THE CONVERSION! Start with what you know. Avogadro’s number is our conversion factor. 15 x 1023 C atoms x = 2. 49 mol C 1 mol 6. 022 x 1023 atoms
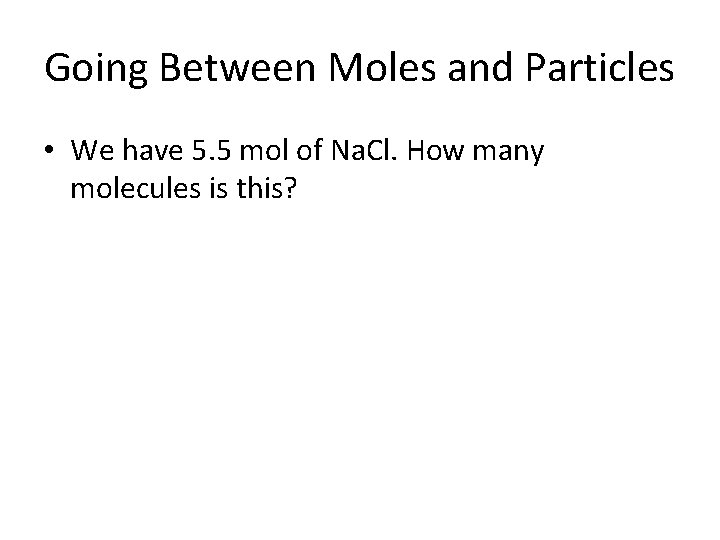
Going Between Moles and Particles • We have 5. 5 mol of Na. Cl. How many molecules is this?

Going Between Moles and Particles • We have 5. 5 mol of Na. Cl. How many molecules is this? 5. 5 mol Na. Cl x 6. 022 x 1023 molecules 1 mol = 33. 121 x 1023 molecules Na. Cl = 3. 3121 x 1024 molecules Na. Cl (correct scientific notation)

Going Between Grams and Particles • This is just another conversion! We don’t know the relationship between grams and particles, so what could we go to first?

Going Between Grams and Particles • This is just another conversion! We don’t know the relationship between grams and particles, so what could we go to first? • MOLES!!! Moles are like our base in metric conversions- when in doubt, convert to moles!

Going Between Grams and Particles • We have 600 x 1023 carbon atoms. How many grams is this?

Going Between Grams and Particles • We have 600 x 1023 carbon atoms. How many grams is this? 600 x 1023 atoms C x 1 mol C x 6. 022 x 1023 atoms C 12. 011 g C= 119. 7 g C 1 mol C

Going Between Grams and Particles • We have 30 g of pure copper. How many atoms of copper is this?

Going Between Grams and Particles • We have 30 g of pure copper. How many atoms of copper is this? 30 g Cu x 1 mol Cu x 6. 022 x 1023 atoms Cu = 63. 546 g Cu 1 mol Cu = 2. 8 x 1023 atoms Cu

Density • Density: a physical property of a substance, representing the mass per unit volume d = m/V m = mass (in grams) V = volume (in m. L) Note: 1 m. L = 1 cm 3 We can use the density to determine the mass if it isn’t given!

Using Density to Determine Mass • Determine the mass of mercury (Hg) in 30 m. L if the density of mercury is 13. 55 g/m. L. Set up the equation! Fill in what you know, then solve.

Using Density to Determine Mass • Determine the mass of mercury (Hg) in 30 m. L if the density of mercury is 13. 55 g/m. L. d = m/V 13. 55 g = m m. L 30 m. L 13. 55 g x 30 = m 406. 5 g = m

Using Density to Determine Mass • Turpentine has a density of 0. 85 g/m. L. How many grams are in 75 m. L?

Using Density to Determine Mass • Turpentine has a density of 0. 85 g/m. L. How many grams are in 75 m. L? d = m/V 0. 85 g = m m. L 75 m. L 0. 85 g x 75 = m 63. 75 g = m

Exit Ticket #1

Exit Ticket #1 Solve the following problems, showing all work for full credit. 1. What is the molar mass of acetic acid (HC 2 H 3 O 2)? (1 point) 2. How many pennies are in one mole of pennies? (1 point) 3. How many moles are in 50 g of magnesium? Show your work! (2 points) 4. How many grams are in 3. 5 mol of Ca. O? Show your work! (3 points) 5. How many molecules are in 15 g of HF? Show your work! (3 points)

Percent Composition • You always bring a lunch to school. You wanted to know heavy your lunch was, so you decided to weigh each of the foods you packed. Your sandwhich was 550 g. Your apple was 200 g. Your cookie was 50 g. Your yogurt was 200 g. What percent of the mass of your lunch was from the sandwich?

Percent Composition • Percent Composition: a given element’s portion of the molecule’s mass = molar mass of the element x 100% molar mass of the whole molecule 1. Find the mass of that element in the molecule 2. Find the total mass of the molecule 3. Enter values into the above equation **If you add up the percentages for all elements in the compound, you should get 100%
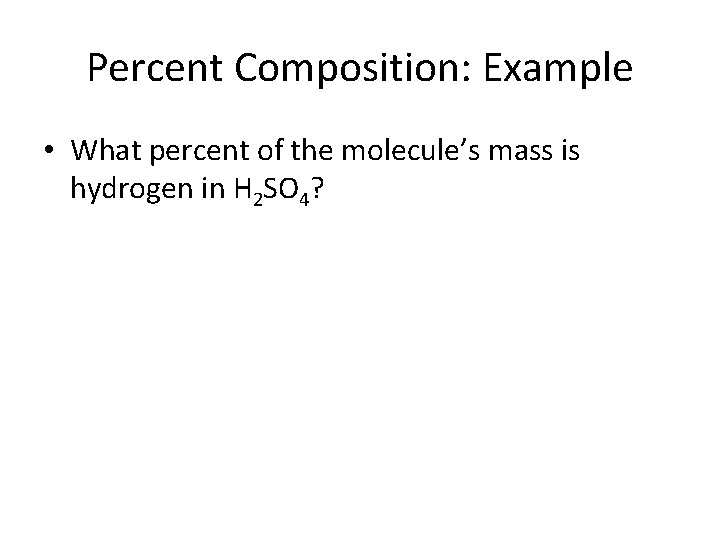
Percent Composition: Example • What percent of the molecule’s mass is hydrogen in H 2 SO 4?
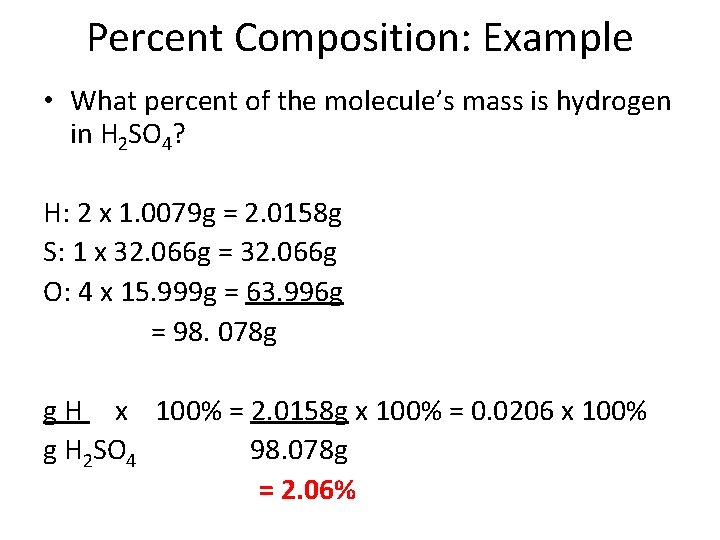
Percent Composition: Example • What percent of the molecule’s mass is hydrogen in H 2 SO 4? H: 2 x 1. 0079 g = 2. 0158 g S: 1 x 32. 066 g = 32. 066 g O: 4 x 15. 999 g = 63. 996 g = 98. 078 g g H x 100% = 2. 0158 g x 100% = 0. 0206 x 100% g H 2 SO 4 98. 078 g = 2. 06%

Percent Composition: Example 2 • Still using H 2 SO 4, determine the percentage of the mass for sulfur and oxygen.

Percent Composition: Example 2 • Still using H 2 SO 4, determine the percentage of the mass for sulfur and oxygen. • S: 32. 066 g/ 98. 078 g x 100% = 32. 69% S • O: 63. 996 g/ 98. 078 g x 100% = 65. 25% O

Empirical Formulas • Empirical Formula: the simplest whole-number ratio of atoms in a compound 1. Assume that you have 100 g of substance 2. Determine the number of grams of each element from the percent composition (ex. 50% carbon = 50 g carbon) 3. Convert each element from grams to moles 4. Divide the moles of each element by the smallest value to get the simplest whole-number ratio • • If you have a decimal of 0. 5, multiply EVERYTHING by 2 If you have a decimal of 0. 333, multiply EVERYTHING by 3 If you have a decimal of 0. 25, multiply EVERYTHING by 4 If the number is VERY close to a whole number (ex. 0. 999), it is acceptable to round

Empirical Formula Example • Determine the empirical formula for a compound consisting of the following: 71. 65% Cl 24. 27% C 4. 07% H

Empirical Formula Example • Determine the empirical formula for a compound consisting of the following: 71. 65% Cl 24. 27% C 4. 07% H Cl: 71. 65 g x 1 mol/35. 45 g = 2. 021 mol/ 2. 021 = 1 C: 24. 27 g x 1 mol/ 12. 01 g = 2. 021 mol/ 2. 021 = 1 H: 4. 07 g x 1 mol/1. 008 g = 4. 04 mol/ 2. 021 = 2 Cl. CH 2

Molecular Formula • Molecular Formula: the exact formula of a molecule – May or may not be the same as the empirical formula – You need to know the molar mass of the compound! 1. Determine the empirical formula 2. Find the molar mass of the empirical formula 3. Divide the molar mass of the compound by the molar mass from the empirical formula 4. Multiply the subscripts in the empirical formula by the above ratio

Molecular Formula Example • Earlier, we determined the empirical formula was Cl. CH 2. Given that the molar mass of the compound is 98. 96 g/mol, determine the molecular formula.

Molecular Formula Example • Earlier, we determined the empirical formula was Cl. CH 2. Given that the molar mass of the compound is 98. 96 g/mol, determine the molecular formula. • Cl. CH 2: 35. 45 g + 12. 01 g + 2. 02 g = 49. 48 g/mol • 98. 96 g/mol = 2 49. 48 g/mol • (Cl. CH 2)2 = Cl 2 C 2 H 4

Practice • Determine the empirical and molecular formulas for a compound containing 40. 0% carbon, 6. 7% hydrogen, and 53. 5% oxygen. The compound has a molar mass of 180. 156 g/mol.

Practice Determine the empirical and molecular formulas for a compound containing 40. 0% carbon, 6. 7% hydrogen, and 53. 5% oxygen. The compound has a molar mass of 180. 156 g/mol. C: 40. 0 g x 1 mol/12. 0107 g = 3. 33 mol/ 3. 33 = 1 H: 6. 7 g x 1 mol/ 1. 00794 g = 6. 65 mol/ 3. 33 = 2 O: 53. 3 g x 1 mol/15. 9994 g = 3. 33 mol/ 3. 33 = 1 Empirical: CH 2 O = 30. 026 g/mol 180. 156 = 6 : CH 2 O Molecular: C 6 H 12 O 6 30. 026

Exit Ticket #2

Exit Ticket #2 - Show ALL Work! 1. What is the percent composition of oxygen in H 2 O? (2 points) 2. Which of the following is an empirical formula? (1 point) a. C 6 H 12 O 6 b. C 2 H 4 NO c. C 2 H 6 d. All of the above. 3. A compound’s empirical formula is C 3 H 7 and its molecular weight is 86 g/mole. Find its molecular formula. (2 points) 4. A compound consists of 72. 2% magnesium and 27. 8% nitrogen by mass. What is its empirical formula? (2 points) 5. A compound is 58. 8 % C, 9. 9 % H, and 31. 3 % O. If its molecular mass is 306 g/mole, what is the molecular formula? (3 points)

Exit Ticket #3

Exit Ticket #3 • Balance the following reactions. Write a coefficient 1 wherever needed so I can tell you didn’t just leave it blank. (2 points each) 1. S 8 + NO 2 SO 2 + N 2 2. C 3 H 8 + O 2 CO 2 + H 2 O 3. C 6 H 6 + HNO 3 C 6 H 5 NO 2 + H 2 O 4. C 5 H 10 + O 2 CH 2 O 5. N 2 + C 2 H 6 N 2 H 4 + C 2 H 2

Stoichiometry • Stoichiometry: the area of chemistry that considers the quantities of materials consumed and produced in chemical reactions • The coefficients in a balanced reaction tell you the RATIO of MOLES for each compound that are needed for the reaction

Reading the Reaction Equation • Cl 2 + 2 KBr 2 KCl + Br 2 – How many moles of Cl 2 will react with 2 moles of KBr? • P 4 + 5 O 2 + 6 H 2 O 4 H 3 PO 4 – How many moles of O 2 are needed to produce 4 moles of H 3 PO 4? • 2 Fe + 3 Cl 2 2 Fe. Cl 3 – How many moles of Fe will produce 2 moles of Fe. Cl 3?

Reading the Reaction Equation • Cl 2 + 2 KBr 2 KCl + Br 2 – How many moles of Cl 2 will react with 2 moles of KBr? 1 mole • P 4 + 5 O 2 + 6 H 2 O 4 H 3 PO 4 – How many moles of O 2 are needed to produce 4 moles of H 3 PO 4? 5 moles • 2 Fe + 3 Cl 2 2 Fe. Cl 3 – How many moles of Fe will produce 2 moles of Fe. Cl 3? 2 moles

Stoichiometry P 4 + 5 O 2 + 6 H 2 O 4 H 3 PO 4 What is the mole ratio between O 2 and H 3 PO 4? (How many moles of O 2 per how many moles of H 3 PO 4? )

Stoichiometry P 4 + 5 O 2 + 6 H 2 O 4 H 3 PO 4 What is the mole ratio between O 2 and H 3 PO 4? (How many moles of O 2 per how many moles of H 3 PO 4? ) There are 5 mol of O 2 per 4 mol of H 3 PO 4 5 mol O 2 or 4 mol H 3 PO 4 5 mol O 2

Stoichiometry P 4 + 5 O 2 + 6 H 2 O 4 H 3 PO 4 What is the mole ratio between P 4 and O 2? What is the mole ratio between P 4 and H 2 O? What is the mole ratio between P 4 and H 3 PO 4?

Stoichiometry P 4 + 5 O 2 + 6 H 2 O 4 H 3 PO 4 What is the mole ratio between P 4 and O 2? 1 mol P 4/ 5 mol O 2 What is the mole ratio between P 4 and H 2 O? 1 mol P 4/ 6 mol H 2 O What is the mole ratio between P 4 and H 3 PO 4? 1 mol P 4/4 mol H 3 PO 4

Stoichiometry P 4 + 5 O 2 + 6 H 2 O 4 H 3 PO 4 What is the mole ratio between O 2 and H 2 O? What is the mole ratio between H 2 O and H 3 PO 4?

Stoichiometry P 4 + 5 O 2 + 6 H 2 O 4 H 3 PO 4 What is the mole ratio between O 2 and H 2 O? 5 mol O 2/ 6 mol H 2 O What is the mole ratio between H 2 O and H 3 PO 4? 6 mol H 2 O/ 4 mol H 3 PO 4

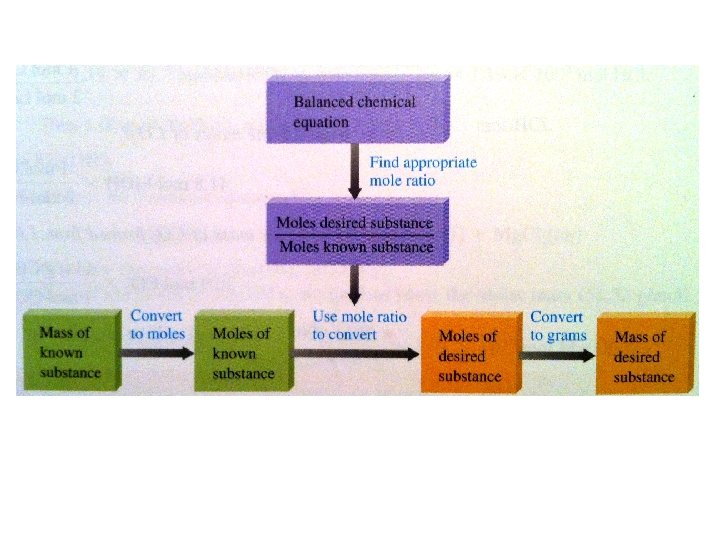

Moles to Moles • We use the mole ratio (from the balanced reaction) as a conversion factor P 4 + 5 O 2 + 6 H 2 O 4 H 3 PO 4 • If we started with 6 mol of O 2, how many moles of H 3 PO 4 would we produce? (Set up the conversion!)

Moles to Moles P 4 + 5 O 2 + 6 H 2 O 4 H 3 PO 4 • If we started with 6 mol of O 2, how many moles of H 3 PO 4 would we produce? (Set up the conversion!) 6 mol O 2 x 4 mol H 3 PO 4 = 24 mol H 3 PO 4 = 4. 8 mol 1 5 mol O 2 5 mol 4. 8 mol of H 3 PO 4

Moles to Grams P 4 + 5 O 2 + 6 H 2 O 4 H 3 PO 4 • We determined that if we start with 6 mol O 2, we produce 4. 8 mol H 3 PO 4. How many grams is this? (Continue with the same conversion setup! Round molar mass to the nearest whole number. )

Moles to Grams P 4 + 5 O 2 + 6 H 2 O 4 H 3 PO 4 • We determined that if we start with 6 mol O 2, we produce 4. 8 mol H 3 PO 4. How many grams is this? 6 mol O 2 x 4 mol H 3 PO 4 x 98 g H 3 PO 4 = 470. 4 g 1 5 mol O 2 1 mol H 3 PO 4

Grams to Moles to Grams P 4 + 5 O 2 + 6 H 2 O 4 H 3 PO 4 • If we start with 25 g of O 2, how many grams of H 3 PO 4 will we produce? (Round molar masses to the nearest whole number)

Grams to Moles to Grams P 4 + 5 O 2 + 6 H 2 O 4 H 3 PO 4 • If we start with 25 g of O 2, how many grams of H 3 PO 4 will we produce? 25 g O 2 x 1 mol O 2 x 4 mol H 3 PO 4 x 98 g H 3 PO 4 = 1 32 g 5 mol O 2 1 mol H 3 PO 4 =61. 25 g H 3 PO 4

Exit Ticket #4

Limiting Reactant • Limiting Reactant: the reactant that limits the amount of product that can be formed in a reaction • Hint: when the word “excess” is used in a problem, it means there is MORE than enough of that reactant, so that reactant will not be limiting

Finding the Limiting Reactant • For each of your reactants, multiply the number of MOLES you start with by the coefficient in front of that compound in the balanced reaction • Whichever compound has the smaller number is the limiting reactant

Finding the Limiting Reactant 1 C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O You have 3 mol C 3 H 8 of and 1. 8 mol of O 2. What is the limiting reactant?

Finding the Limiting Reactant 1 C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O You have 3 mol C 3 H 8 of and 1. 8 mol of O 2. What is the limiting reactant? C 3 H 8: 3 mol x 1 = 3 mol Limiting O 2: 1. 8 mol x 5 = 9 mol

Finding the Limiting Reactant 1 C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O You have 7. 6 mol C 3 H 8 of and 2. 3 mol of O 2. What is the limiting reactant?

Finding the Limiting Reactant 1 C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O You have 7. 6 mol C 3 H 8 of and 2. 3 mol of O 2. What is the limiting reactant? C 3 H 8: 7. 6 mol x 1 = 7. 6 mol Limiting O 2: 2. 3 mol x 5 = 11. 5 mol

Finding the Limiting Reactant 1 C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O You have 15. 9 mol C 3 H 8 of and 3. 3 mol of O 2. What is the limiting reactant? C 3 H 8: 15. 9 mol x 1 = 15. 9 mol O 2: 3. 1 mol x 5 = 15. 5 mol Limiting

Finding the Limiting Reactant 1 C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O You have 7. 5 mol C 3 H 8 of and 1. 5 mol of O 2. What is the limiting reactant? C 3 H 8: 7. 5 mol x 1 = 7. 5 mol O 2: 1. 5 mol x 5 = 7. 5 mol No limiting reactant

Finding the Limiting Reactant 1 C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O You have 45 g C 3 H 8 of and 40 g of O 2. What is the limiting reactant?

Finding the Limiting Reactant 1 C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O You have 45 g C 3 H 8 of and 40 g of O 2. What is the limiting reactant? *Since we’re given grams, we must convert to moles!

Finding the Limiting Reactant 1 C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O You have 45 g C 3 H 8 of and 40 g of O 2. What is the limiting reactant? C 3 H 8: 45 g x 1 mol = 1. 02 mol x 1 = 1. 02 mol (limiting) 44. 097 g O 2: 40 g x 1 mol = 1. 25 mol x 5 = 6. 25 mol 31. 998 g

Determining Yield • Theoretical Yield: the amount of product that a reaction produces if it reaches completion under perfect conditions – Once you know the limiting reactant, you can calculate how much of your product the reaction should produce – To set up the conversion, use whatever amount of the limiting reactant was given to you, then set up the conversion from there

Determining Yield 1 C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O You have 3 mol C 3 H 8 of and 1. 8 mol of O 2. How many grams of H 2 O will be produced? C 3 H 8: 3 mol x 1 = 3 mol Limiting O 2: 1. 8 mol x 5 = 9 mol

Determining Yield 1 C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O You have 3 mol C 3 H 8 of and 1. 8 mol of O 2. How many grams of H 2 O will be produced? C 3 H 8: 3 mol x 1 = 3 mol Limiting O 2: 1. 8 mol x 5 = 9 mol 3 mol C 3 H 8 x 4 mol H 2 O x 18 g H 2 O = 216 g H 2 O 1 1 mol C 3 H 8 1 mol H 2 O

Determining Yield 1 C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O You have 7. 6 mol C 3 H 8 of and 2. 3 mol of O 2. How many grams of CO 2 are produced? C 3 H 8: 7. 6 mol x 1 = 7. 6 mol Limiting =11. 5 mol O 2: 2. 3 mol x 5

Determining Yield 1 C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O You have 7. 6 mol C 3 H 8 of and 2. 3 mol of O 2. How many grams of CO 2 are produced? C 3 H 8: 7. 6 mol x 1 = 7. 6 mol Limiting =11. 5 mol O 2: 2. 3 mol x 5 7. 6 mol C 3 H 8 x 3 mol CO 2 x 44 g CO 2 = 1, 003 g H 2 O 1 1 mol C 3 H 8 1 mol CO 2

Determining Yield 1 C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O You have 200 g C 3 H 8 of and 200 g of O 2. How many grams of H 2 O are produced?

Determining Yield 1 C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O You have 200 g C 3 H 8 of and 200 g of O 2. How many grams of H 2 O are produced? C 3 H 8: 200 g x 1 mol = 4. 55 mol x 1 = 4. 55 mol: Limiting 44 g O 2: 200 g x 1 mol = 6. 25 mol x 5 = 31. 25 mol 32 g 200 g C 3 H 8 x 1 mol C 3 H 8 x 4 mol H 2 O x 18 g H 2 O = 327. 3 g 1 44 g C 3 H 8 1 mol H 2 O

Percent Yield (Recovery) • Percent Yield (Percent Recovery): the percent of theoretical yield that was obtained during an experiment – Will always be less than 100% Percent Yield = Actual Yield x 100% Theoretical Yield

Percent Yield (Recovery) Previously, we calculated that 216 g H 2 O would be produced if we started with 3 mol C 3 H 8 and 1. 8 mol of O 2. If we recovered 200 g, what was our percent yield (percent recovery)?

Percent Yield (Recovery) Previously, we calculated that 216 g H 2 O would be produced if we started with 3 mol C 3 H 8 and 1. 8 mol of O 2. If we recovered 200 g, what was our percent yield (percent recovery)? % Yield = 200 g x 100 = 92. 6% 216 g

Percent Yield (Recovery) Previously, we calculated that 1, 003 g H 2 O would be produced if we started with 7. 6 mol C 3 H 8 and 2. 3 mol of O 2. If we recovered 578 g, what was our percent yield (percent recovery)? % Yield = 578 g x 100 = 57. 6% 1, 003 g

Summary of Stoichiometry 1. Balance the reaction. 2. Find the limiting reactant by multiplying starting moles by the coefficients in the reaction. 3. Determine theoretical yield (conversions). 4. If given actual yield, determine percent yield.

Making Solutions: Molarity • Molarity: the concentration of a solution, measured in moles of solute per liter of solvent M = mol solute / L solvent Solute: what you’re dissolving Solvent: what does the dissolving (usually a liquid)

Making Solutions • If you have 10 mol of HCl dissolved in 0. 75 L of water, what is the molarity of the solution?

Making Solutions • If you have 10 mol of HCl dissolved in 0. 75 L of water, what is the molarity of the solution? M = mol/ L M = 10 mol/ 0. 75 L = 13. 3 M

Making Solutions • If you have a 6 M solution of HCl that is 300 m. L, how many moles of HCl are dissolved?

Making Solutions • If you have a 6 M solution of HCl that is 300 m. L, how many moles of HCl are dissolved? M = mol/ L 300 m. L x 1 L/ 1, 000 m. L = 0. 3 L 6 M = mol/ 0. 3 L *0. 3 L = *0. 3 L 1. 8 mol

Making Solutions • You’re trying to make a 1. 25 M solution of HCl. If you have 500 g of HCl, what volume must the solution be in L? In m. L?

Making Solutions • You’re trying to make a 1. 25 M solution of HCl. If you have 500 g of HCl, what volume must the solution be in L? In m. L? M = mol/L 500 g x 1 mol/ 36 g = 13. 9 mol 1. 25 M = 13. 9 mol/ L 11. 1 L x 1, 000 m. L/ 1 L = 11, 100 L

Dilutions • Dilution: adding solvent to a solution to decrease the molarity (concentration) • The product of the initial volume and molarity = the product of the final volume and molarity M 1 V 1 = M 2 V 2 M = concentration (molarity) V = volume

Dilutions Example • You have a 250 m. L stock solution of 5 M HCl. You need to make a dilution to 1. 5 M HCl. What must the final volume be? How much water will you have to add?

Dilutions Example • You have a 250 m. L stock solution of 5 M HCl. You need to make a dilution to 1. 5 M HCl. What must the final volume be? How much water will you have to add? M 1 V 1 = M 2 V 2 5 M*250 m. L = 1. 5 MV 2 1, 250 M*m. L = 1. 5 MV 2 1. 5 M 833. 3 m. L = V 2

Molarity of Sugar in Sodas Activity • Your task is to determine the molarity (concentration) of sugar in various beverages. To do this, you must determine: – The amount of sugar in one serving – The volume of one serving • We will make the following assumptions: – All sugar in the beverage is fructose: C 6 H 12 O 6 – There are 30 m. L per 1 fl. oz. • When you finish, read the article on sugar and tooth decay, and relate the molarity of sugar that you found to the article.

Exit Ticket #6

Exit Ticket #6 Show all work, including the original equation. The molar mass of HCl is 36. 5 g/mol. Round your answers to the nearest tenth. 1. How many mol of HCl are in a 2 L solution with a concentration of 6. 5 M? (1 point) 2. How many grams of HCl are required to make a 2 M, 350 m. L solution? (2 points) 3. What volume of HCl is needed to create a 3. 5 M solution from a starting mass of 150 g? (3 points) 4. You dilute a 5. 5 M solution of HCl from 100 m. L to 250 m. L. What is the final concentration? (1 point) 5. You leave a 200 m. L solution of 3 M HCl out overnight, and some of the water evaporates. If the final volume is 125 m. L, what is the final concentration? (2 points) 6. You have a 3 M solution of HCl. You add water to the solution until you reach a final concentration of 1. 2 M. If you started with 175 m. L, how much water did you add? (3 points)
- Slides: 112