Mohammed BOUZID Mohamed GHANEM Youcef DEHBI ABOUT GILEAD

Mohammed BOUZID Mohamed GHANEM Youcef DEHBI

ABOUT GILEAD US biopharmarceutical company HQ in Foster City, California Founded in 1987 by Dr MICHAEL RIORDAN under the name of “OLIGOGEN” OLIGOGEN Named after “the balm of Gilead “ Completed its IPO in 1991 Michael L. Riordan Gilead’s primary areas of focus include : viral, cardiovascular and respiratory diseases
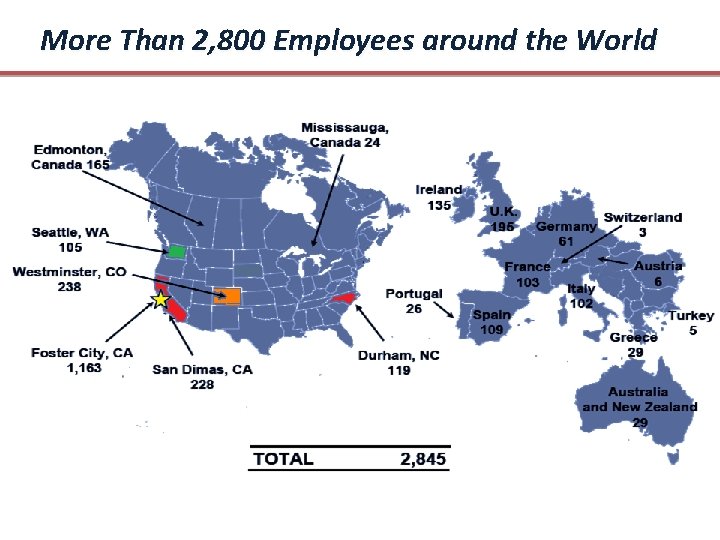
More Than 2, 800 Employees around the World

ANTISENSE : GILEAD’S FIRST STEP
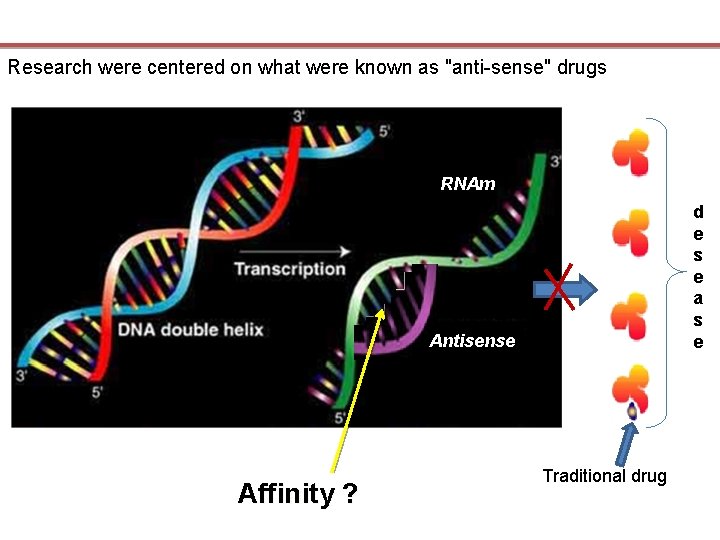
Research were centered on what were known as "anti-sense" drugs RNAm d e s e a s e Antisense Affinity ? Traditional drug

A « propyne group » was added to bases of nucleotids Binding affinity increased by 10 to 100 times but Several problems had to be resolved before making antisense work as drug
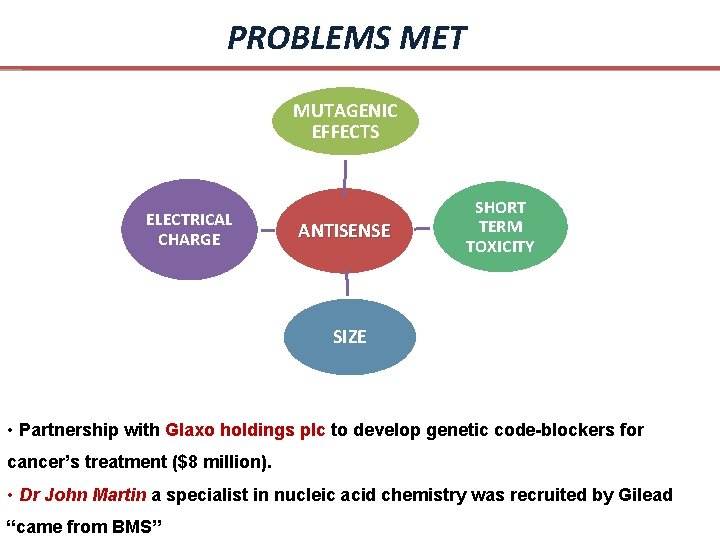
PROBLEMS MET MUTAGENIC EFFECTS ELECTRICAL CHARGE ANTISENSE SHORT TERM TOXICITY SIZE • Partnership with Glaxo holdings plc to develop genetic code-blockers for cancer’s treatment ($8 million). • Dr John Martin a specialist in nucleic acid chemistry was recruited by Gilead “came from BMS”

After several years of research: A strong expertise in nucleic acid chemistry was established The company was not closer to marketable product. Gilead will need an other alternative!

Second step: From antisense to antivirals

How it began ? • Bristol-Myers merged with Squibb in 1989 and cut back on HIV spending. • Martin went back to Bristol-Myers library returned to its european inventors • In 1991, Gilead licensed rights to the anti-viral drug candidates. • Research were redirected to antivirals and antisense program was abandoned later Three marketed products: vistide; Hepsera; viread All patents of Antisens chemistry were sold to ISIS pharmaceutical for $6 M(1998)

Gilead’s objectives Keeping its focus on antivirals To Favour collaboration and partnerships To growth through acquisitions Reducing dvpt costs and commercialisation timeline Satisfy the unmet medical need

Acquisitions REQUIRMENTS & REQUISES
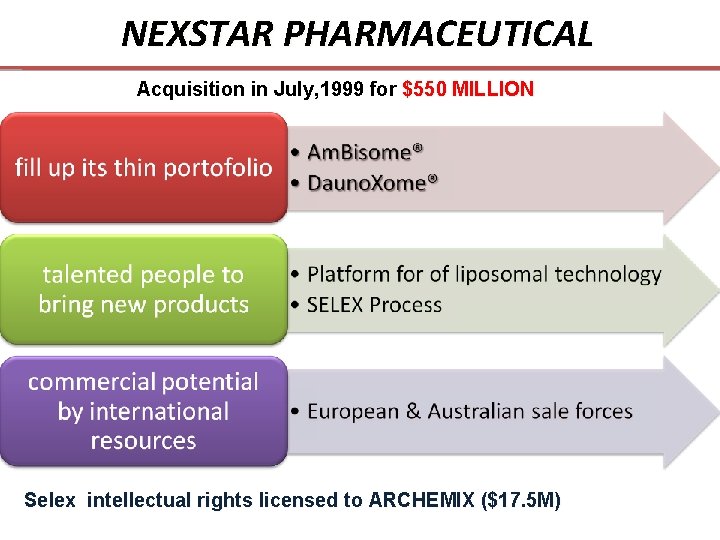
NEXSTAR PHARMACEUTICAL Acquisition in July, 1999 for $550 MILLION Selex intellectual rights licensed to ARCHEMIX ($17. 5 M)

liposomal drug delivery technology: Side effects and administration problems of potent compounds can be solved New patent obtained by new formulation Am. Bisome® Phospholipid bilayer Lower toxicity of amphotericin B Affinity for ergosterol in fungal cell membrane Amphoterician B molecules

Triangle Pharmaceuticals Acquisition in Jan, 2003 for $464 million Keep founds • Triangle researches were focused on therapies for flowing to R&D in HIV and HBV ATV created field Similar and complementary profile to Viread • Co formulation emtricitabine/Viread into one pill U. S. approval, 2004 of TRUVADA® TRUVADA

Corus Pharma Acquisition in Aug, 2006 for $365 MILLION a center of Moving into new • establishing expertise in respiratory therapeutic areas therapeutics for the company. Unmet medical needs • Phase III aztreonam lysine for inhalation for cystic fibrosis treatment CF impacts the lives of more than 70000 people Aztreonam lysine was granted orphan drug status

Raylo Chemicals Inc Nov, 2006 for 107 million Euros improve existing commercial manufacturing processes. • manufacturing of API for investigational and commercial products

Myogen nov. 17, 2006 $2, 5 Billion Inhances growing focus • Research program in non-infection on cardiopulmonary dvpt lung disorders • Darusentan, Ambrisentan in clinical Marketed or early stage dvpt drugs • Flolan marketed since 1995 Unmet medical need • PAH, resistant hypertension

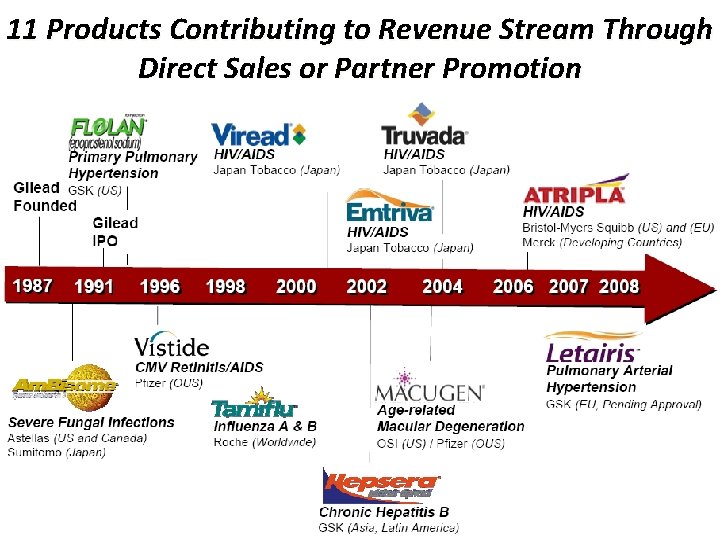
11 Products Contributing to Revenue Stream Through Direct Sales or Partner Promotion

HIV / HBV Foster City / Durham
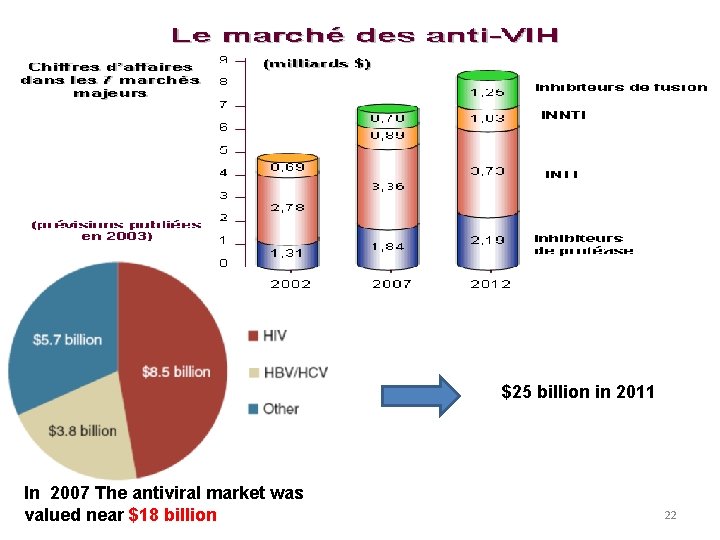
$25 billion in 2011 In 2007 The antiviral market was valued near $18 billion 22
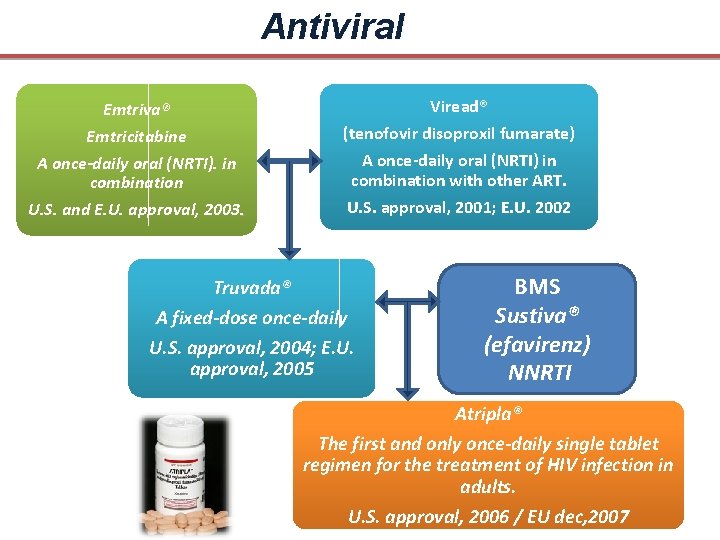
Antiviral Emtriva® Emtricitabine A once-daily oral (NRTI). in combination U. S. and E. U. approval, 2003. Viread® (tenofovir disoproxil fumarate) A once-daily oral (NRTI) in combination with other ART. U. S. approval, 2001; E. U. 2002 Truvada® A fixed-dose once-daily U. S. approval, 2004; E. U. approval, 2005 BMS Sustiva® (efavirenz) NNRTI Atripla® The first and only once-daily single tablet regimen for the treatment of HIV infection in adults. U. S. approval, 2006 / EU dec, 2007
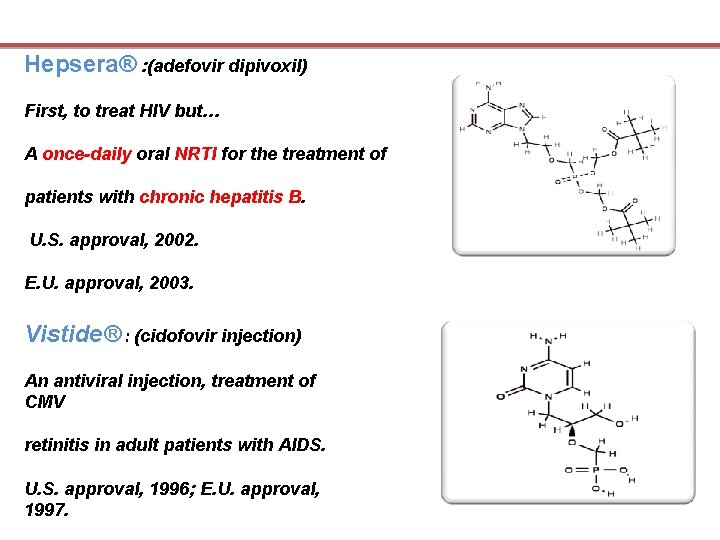
Hepsera® : (adefovir dipivoxil) First, to treat HIV but… A once-daily oral NRTI for the treatment of patients with chronic hepatitis B. U. S. approval, 2002. E. U. approval, 2003. Vistide® : (cidofovir injection) An antiviral injection, treatment of CMV retinitis in adult patients with AIDS. U. S. approval, 1996; E. U. approval, 1997.

Seattle
Tamiflu® (oseltamivir phosphate) The first orally neuraminidase inhibitor (influenza A and B) FDA: treatment Oct 1999 and for prevention in Nov 2000. Available in Europe since 2002. The product, which was developed by Gilead, is commercialized globally by Hoffmann-La Roche. • Roche pays royalties to Gilead based on a percentage of the net sales generate from Tamiflu worldwide

Others Am. Bisome® : (amphotericin B liposome for injection) A treatment for systemic fungal infections in adults. E. U. approval, 1990; U. S. approval, 1997. Macugen® : (pegaptanib sodium injection) An injection for the treatment of neovascular age-related macular degeneration (also known as "wet" AMD) This product is marketed in the United States by OSI Pharmaceuticals. Royalties to Gilead U. S. approval, 2004.
Cardiopulmonary the new expectation
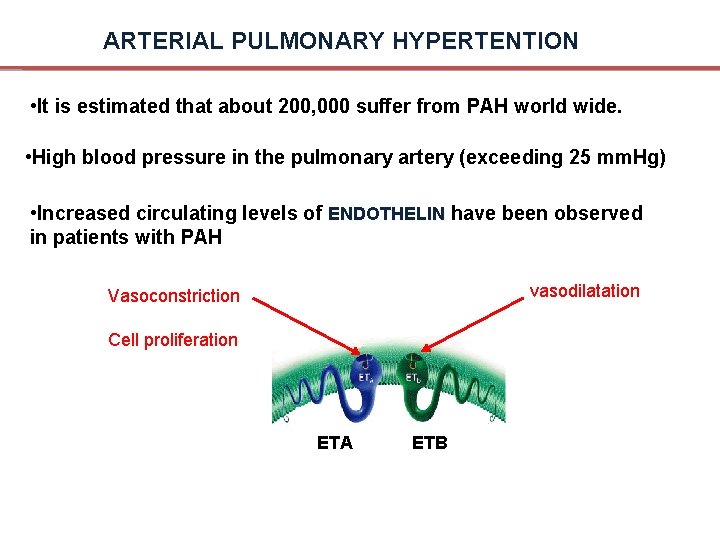
ARTERIAL PULMONARY HYPERTENTION • It is estimated that about 200, 000 suffer from PAH world wide. • High blood pressure in the pulmonary artery (exceeding 25 mm. Hg) • Increased circulating levels of ENDOTHELIN have been observed in patients with PAH vasodilatation Vasoconstriction Cell proliferation ETA ETB
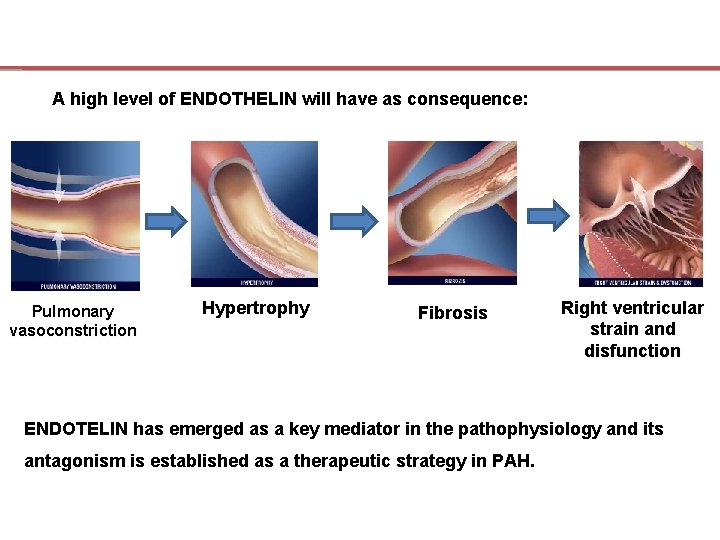
A high level of ENDOTHELIN will have as consequence: Pulmonary vasoconstriction Hypertrophy Fibrosis Right ventricular strain and disfunction ENDOTELIN has emerged as a key mediator in the pathophysiology and its antagonism is established as a therapeutic strategy in PAH.

FIRST IN CLASS : Tracleer®(Actelion Inc) a non selective antagonist orally active drug. In 2007 Tracleer sales were $1, 18 billion. GILEAD’ S PRODUCT LETAIRIS(Ambrisentan) was approved as a selective ETA receptor antagonist (Ambrisentan) orally active drug in 2007. Parity pricing to Tracleer has helped with reimbursement. Main patients sources : • Naïve patients. • Tracleer patients with elevated LFTs(liver toxicity) • SILDENAFIL monotherapy poor responders Commercialized by GSK outside of US ($1 billion expected in 2010)
Flolan® (Epoprostenol sodium for injection) An analogue of prostacyclin PGI 2 Effects vasodilation which in turn lowers the blood pressure A long-term intravenous treatment for primary pulmonary hypertension. U. S. approval, 1995.

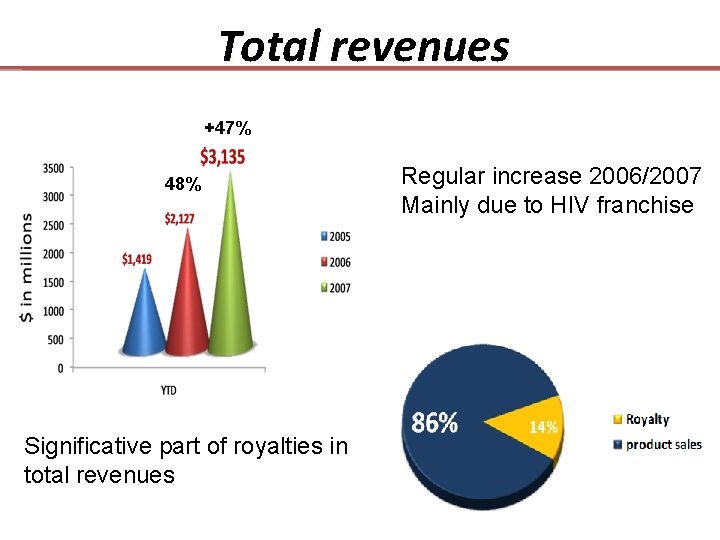
Total revenues +47% 48% Significative part of royalties in total revenues Regular increase 2006/2007 Mainly due to HIV franchise
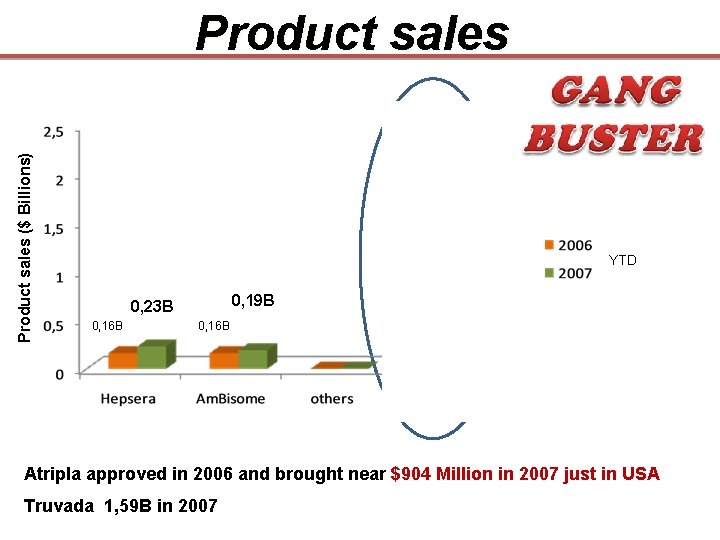
Product sales ($ Billions) 2, 3 B 1, 5 B YTD 0, 19 B 0, 23 B 0, 16 B Atripla approved in 2006 and brought near $904 Million in 2007 just in USA Truvada 1, 59 B in 2007
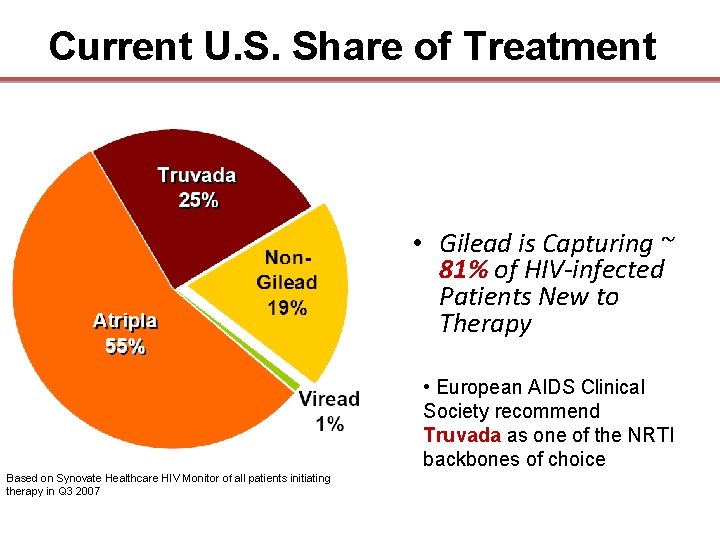
Current U. S. Share of Treatment • Gilead is Capturing ~ 81% of HIV-infected Patients New to Therapy • European AIDS Clinical Society recommend Truvada as one of the NRTI Truvada backbones of choice Based on Synovate Healthcare HIV Monitor of all patients initiating therapy in Q 3 2007
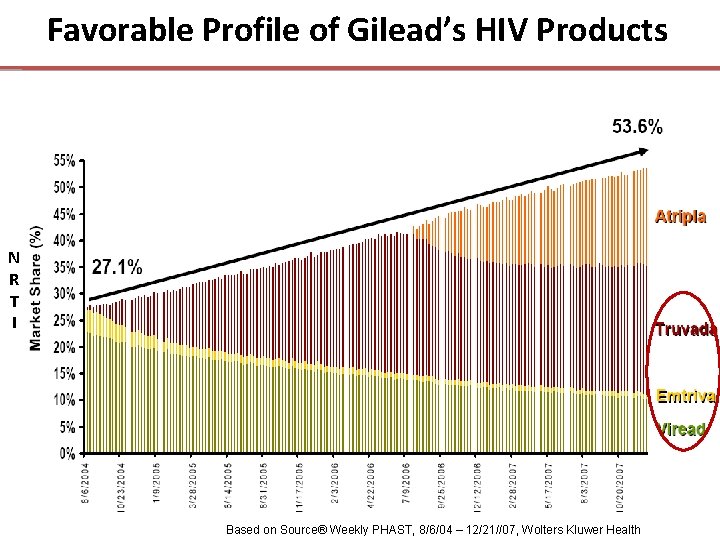
Favorable Profile of Gilead’s HIV Products N R T I Based on Source® Weekly PHAST, 8/6/04 – 12/21//07, Wolters Kluwer Health
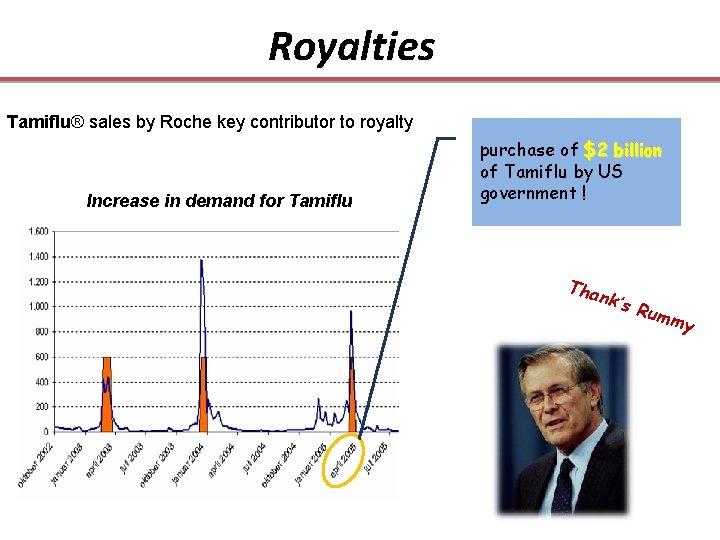
Royalties Tamiflu® sales by Roche key contributor to royalty Increase in demand for Tamiflu purchase of $2 billion of Tamiflu by US government ! Tha nk’s Rum my
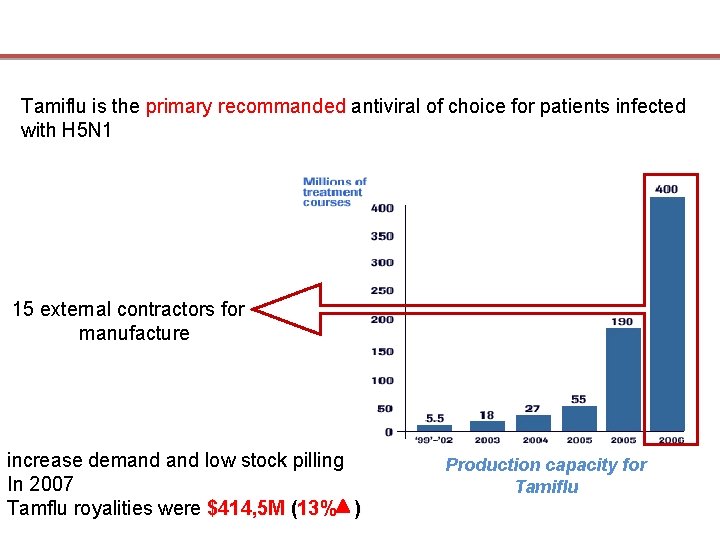
Tamiflu is the primary recommanded antiviral of choice for patients infected with H 5 N 1 15 external contractors for manufacture increase demand low stock pilling In 2007 Tamflu royalities were $414, 5 M (13% ) Production capacity for Tamiflu
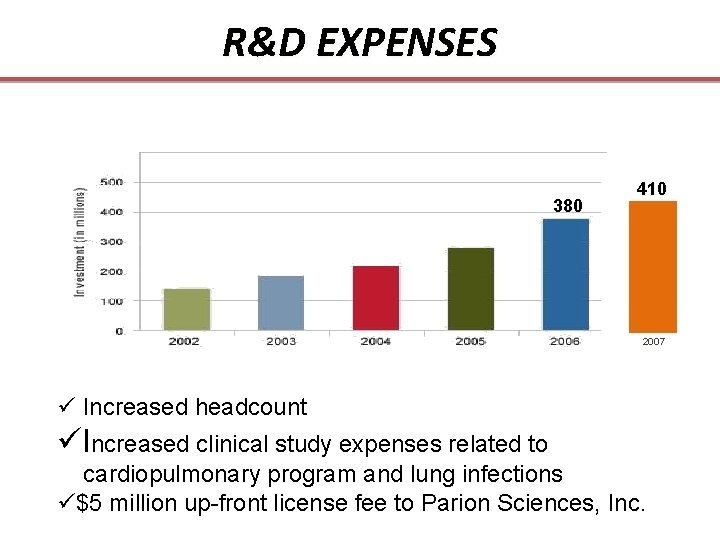
R&D EXPENSES 380 410 2007 ü Increased headcount üIncreased clinical study expenses related to cardiopulmonary program and lung infections ü$5 million up-front license fee to Parion Sciences, Inc.
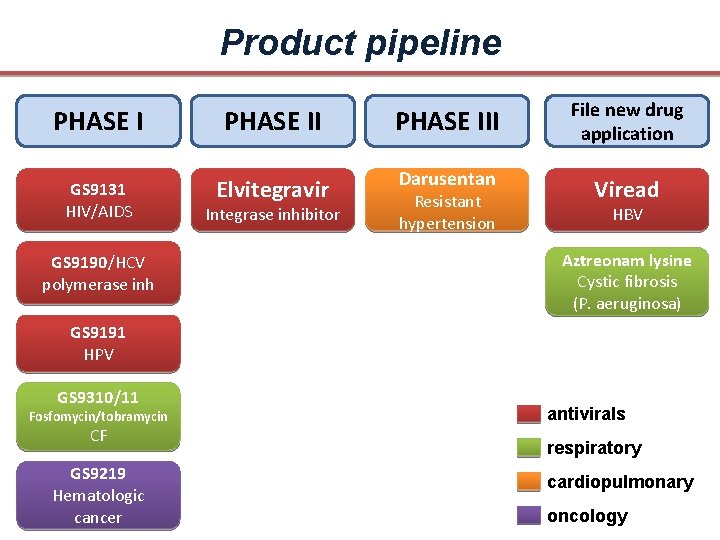
Product pipeline PHASE III File new drug application GS 9131 HIV/AIDS Elvitegravir Darusentan Viread GS 9190/HCV polymerase inh Integrase inhibitor Resistant hypertension HBV Aztreonam lysine Cystic fibrosis (P. aeruginosa) GS 9191 HPV GS 9310/11 Fosfomycin/tobramycin CF GS 9219 Hematologic cancer antivirals respiratory cardiopulmonary oncology

HIV prevalance Run clinical trail GILD Access Program Generics companies 42
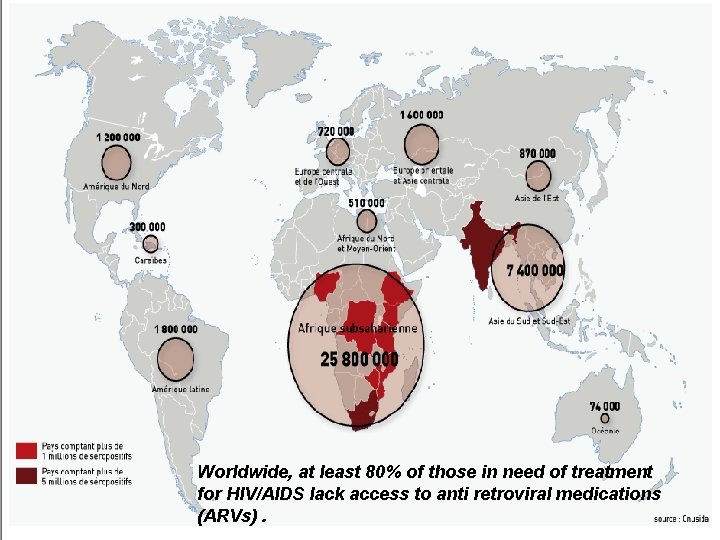
Worldwide, at least 80% of those in need of treatment for HIV/AIDS lack access to anti retroviral medications 43 (ARVs).
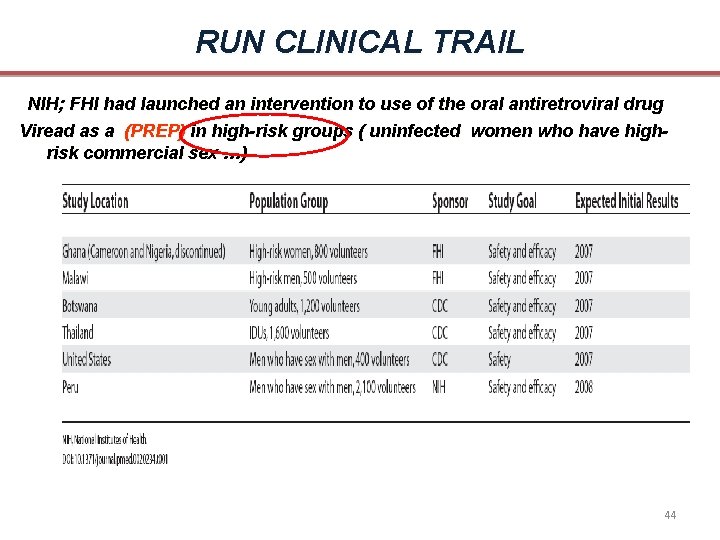
RUN CLINICAL TRAIL NIH; FHI had launched an intervention to use of the oral antiretroviral drug Viread as a (PREP) in high-risk groups ( uninfected women who have highrisk commercial sex …) 44

Protests against Trials of PREP In 2004 dramatic protest against the Cambodian trial at the XV International AIDS Conference in Bangkok, caught the world’s media attention Reasons cited by the activists : • Inadequate prevention counseling by investigators, • Lack of pre- and post-test HIV counseling, • Nonprovision of medical services. • Safety of tenofovir for longterm use by individuals who are HIV negative has not been established. • participants take all risks and get little of the benefits. 45

consequences • In July 2004, pressure from activist groups and affiliated nongovernmental organizations persuaded the Cambodian Prime Minister to halt the initiation of a PREP trial of tenofovir among Cambodian commercial sex workers • In February 2005, a similar trial in Cameroon, was halted by the Minister of Public Health • March 11, 2005, FHI Nigerian arm of the tenofovir PREP trial discontinued prematurely. 46

GILD Access Program In 2002, provide the drugs throughout the world at reduced prices based on a country’s economic status and HIV prevalence. 35 47
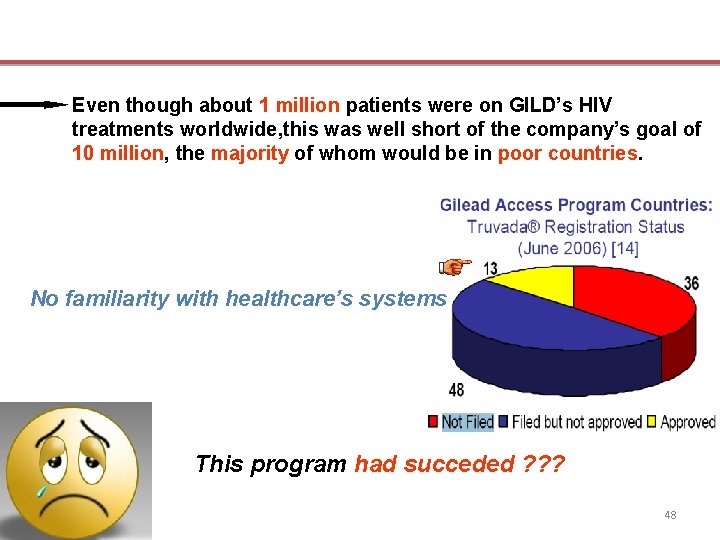
Even though about 1 million patients were on GILD’s HIV treatments worldwide, this was well short of the company’s goal of 10 million, the majority of whom would be in poor countries. No familiarity with healthcare’s systems This program had succeded ? ? ? 48

Gilead & Generics companies In 2005 Aspen Pharmacare Holdings Ltd. non-exclusive rights to manufacture Viread and Truvada for all of the 98 poorest countries and to distribute the drugs in every country in Africa. In 2005, GILD partnered with Pharma. Chem Technologies Ltd. to open a manufacturing facility for Viread in the Bahamas ease access in lowincome countries in the Caribbean and Latin America In 2007 Gilead provide voluntary, non-exclusive licenses to 10 Indian generic pharmaceutical companies for the manufacture of viread and truvada 49
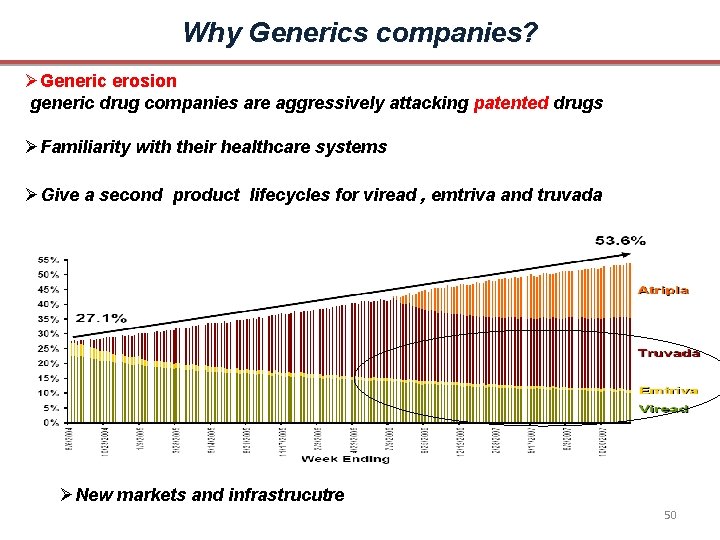
Why Generics companies? Generic erosion generic drug companies are aggressively attacking patented drugs Familiarity with their healthcare systems Give a second product lifecycles for viread , emtriva and truvada New markets and infrastrucutre 50

51
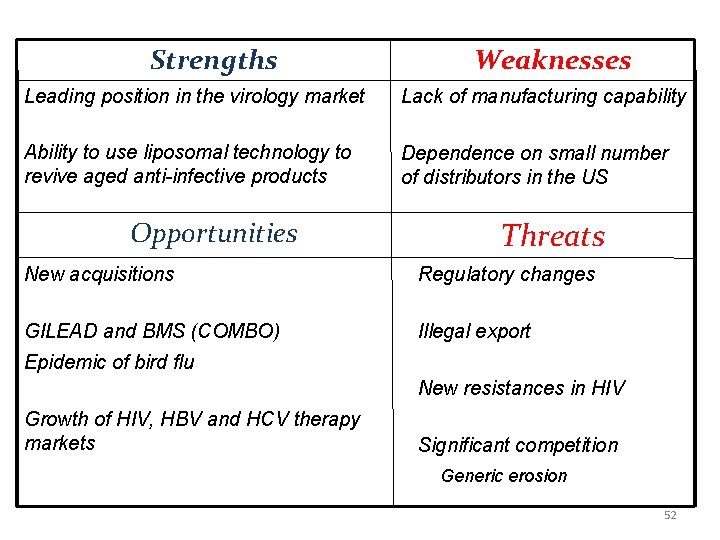
Strengths Weaknesses Leading position in the virology market Lack of manufacturing capability Ability to use liposomal technology to revive aged anti-infective products Dependence on small number of distributors in the US Opportunities Threats New acquisitions Regulatory changes GILEAD and BMS (COMBO) Illegal export Epidemic of bird flu New resistances in HIV Growth of HIV, HBV and HCV therapy markets Significant competition Generic erosion 52

conclusion Gilead is a biopharmaceutical company which is growing through its strategic acquisitions and partnership slowly but certainly. . . Paul Berg, Ph. D Nobel Prize Winner John C. Martin, Ph. D President and Chief Executive Officer James M. Denny Chairman 53


references • • PLo. S Medicine | www. plosmedicine. org Bio. Century ; Unleashing market forces Gilead Sciences, Inc. Company Profile Reference Code: 2499 Publication Date: Jan 2007 Company-History. html • http: //seekingalpha. com/symbol/gild • 26 th Annual JPMorgan Healthcare Conference January 7, 2008 • Q 3 2007 Earnings results Oct, 18 , 2007 Archemix http: //www. archemix. com (accessed December 2002) • • EUROPEAN RESPIRATORY REVIEW, 2007; 16: 13 -18. doi: 10. 1183/09059180. 00010204 • http: //www. fundinguniverse. com/co mpany-histories/Gilead-Sciences-Inc- www. gilead. com
- Slides: 55