Module 7 Renal Cell Carcinoma and Urothelial Bladder

Module 7: Renal Cell Carcinoma and Urothelial Bladder Cancer Susan K Roethke, CRNP, MSN, ANP-BC, AOCNP David I Quinn, MBBS, Ph. D

Disclosures for Ms Roethke Advisory Committee Astellas Speakers Bureau Astellas, Pfizer Inc

Disclosures for Dr Quinn Advisory Committee and Consulting Agreements Astellas, Astra. Zeneca Pharmaceuticals LP, Bayer Health. Care Pharmaceuticals, Bristol-Myers Squibb Company, Celgene Corporation, Dendreon Pharmaceuticals Inc, Exelixis Inc, Genentech, Janssen Biotech Inc, Merck, Novartis, Pfizer Inc, Pharmacyclics LLC, an Abb. Vie Company, Roche Laboratories Inc, Sanofi Genzyme, Seattle Genetics Contracted Research Bayer Health. Care Pharmaceuticals, Bristol-Myers Squibb Company, Genentech, Merck, Pfizer Inc, Pharmacyclics LLC, an Abb. Vie Company, Roche Laboratories Inc, Seattle Genetics Data and Safety Monitoring Board/Committee Children’s Hospital Los Angeles, Eisai Inc, US Biotest Inc
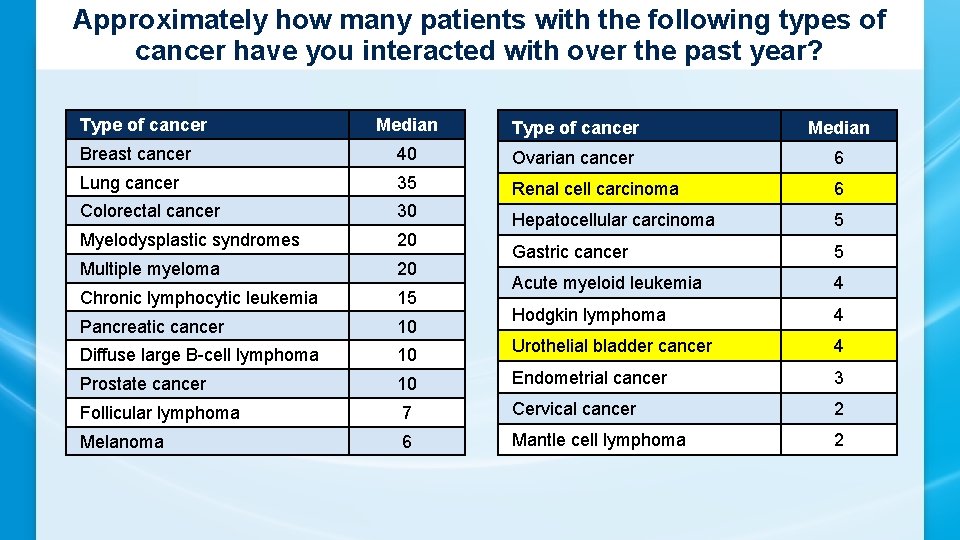
Approximately how many patients with the following types of cancer have you interacted with over the past year? Type of cancer Median Breast cancer 40 Ovarian cancer 6 Lung cancer 35 Renal cell carcinoma 6 Colorectal cancer 30 Myelodysplastic syndromes 20 Hepatocellular carcinoma 5 Multiple myeloma 20 Gastric cancer 5 Chronic lymphocytic leukemia 15 Acute myeloid leukemia 4 Pancreatic cancer 10 Hodgkin lymphoma 4 Diffuse large B-cell lymphoma 10 Urothelial bladder cancer 4 Prostate cancer 10 Endometrial cancer 3 Follicular lymphoma 7 Cervical cancer 2 Melanoma 6 Mantle cell lymphoma 2

Day in the Life: Renal Cell Carcinoma • 68 F, Ipilimumab/nivolumab, Newly married • 46 M, Trying to start axitinib but continues to have admissions for brain metastasis, rapidly declining performance status while we try to start treatment • 50 M, temsirolimus, has been on this drug for 4 years with no evidence of progression • 58 M, Mets to lung, bone, axitinib, HIV positive • 63 M, Nivolumab, He had excellent response to nivolumab and absolutely no side effects/complaints. • 52 M, Ipilimumab/nivolumab, Doing well on current treatment plan • 81 M, brain mets, on hold as is inpatient hospital, he stays in good spirits even as he quickly deteriorates

Day in the Life: Bladder Cancer • 90 F, Gemcitabine, Comorbidities including age, frailty, pain and opioid management, constipation, depression, declining performance status • 78 M, Concurrent radiation and weekly gemcitabine, Asked me about supplements and showed me the reishi mushroom preparation that he bought asking my opinion • 78 M, Intravesical gemcitabine, Frustration with care • 78 F, Bladder cancer and DLBCL, Extreme anxiety • 49 F, 3 rd line tx, now with cord compression neurosurgery, cord compression • 89 M, Concurrent radiation and Gemcitabine, Comorbidities including hydronephrosis, DVT, acute UTI requiring hospitalization, amiodarone with hypothyroidism • 70 M, Bladder cancer and skin cancer squamous cell, Clinical trial, Tolerating treatment well

Renal Cell Carcinoma

Case #3: Metastatic RCC treated with TKI & immunotherapy • A 67 yo female, retired 3 rd grade teacher, initially presented with fever and night sweats, weight loss and palpable left supraclavicular adenopathy (11/2016) • CT C/A/P: supraclavicular and retroperitoneal adenopathy, and left renal mass • Biopsy of supraclav node: poorly differentiated carcinoma with clear cell morphology c/w met RCC • PMH: HTN, cluster headaches/migraines; non celiac gluten sensitivity • Enrolled in phase II trial of axitinib + pembrolizumab, started 1/2017

Case #3 • Challenge: teaching with TKIs on top on immunotherapy, Also patient’s history: Baseline HTN, migraines • Potential overlapping toxicities to differentiate: • Need to assess diarrhea/loose bowels of TKIs vs potential autoimmune colitis • If rash, what’s responsible? • Managing arthralgias/myalgias • Evaluating source/severity of lab abnormalities
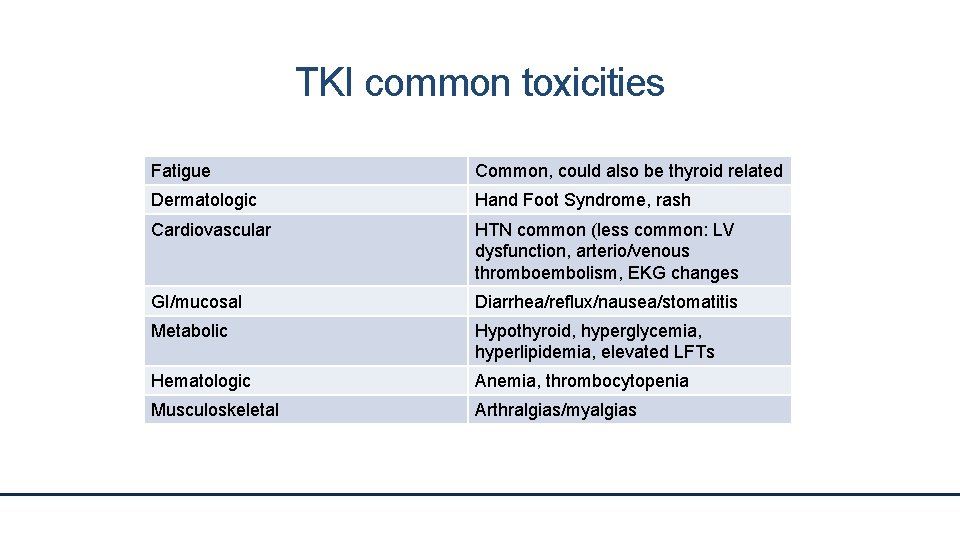
TKI common toxicities Fatigue Common, could also be thyroid related Dermatologic Hand Foot Syndrome, rash Cardiovascular HTN common (less common: LV dysfunction, arterio/venous thromboembolism, EKG changes GI/mucosal Diarrhea/reflux/nausea/stomatitis Metabolic Hypothyroid, hyperglycemia, hyperlipidemia, elevated LFTs Hematologic Anemia, thrombocytopenia Musculoskeletal Arthralgias/myalgias

Case #3 • Patient’s course: developed HTN, HFS, stomatitis, diarrhea, hypothyroidism, h/a’s, myalgias, needed tooth extraction. PLUS: wanted to visit grandchildren & take vacations! MANAGEMENT: • Regular monitoring of CBC, CMP, TSH. And physical exams • HTN: Patient self monitoring BP; added antihypertensives • HFS, stomatitis, diarrhea: dose interruption, supportive measures**, dose reduction of axitinib • Dose interruption axitinib for wound healing, & sometimes for vacations • Myalgias — realized due to pembro; analgesics, stretching, acupuncture • Thyroid replacement — regular monitoring TFTs

**Preventive/Supportive Measures: GI/Mucosal • Diarrhea • Dietary changes, replace fluids • OTC antidiarrheal • Psyllium agents • Reflux • Prescription or OTC H 2 blockers or PPIs (caution) • Dietary changes • Stomatitis • Dietary changes • Sensitive toothpaste • Salt & Soda rinses, “magic mouthwash” (NO antifungal) • Nausea/vomiting • Antiemetics
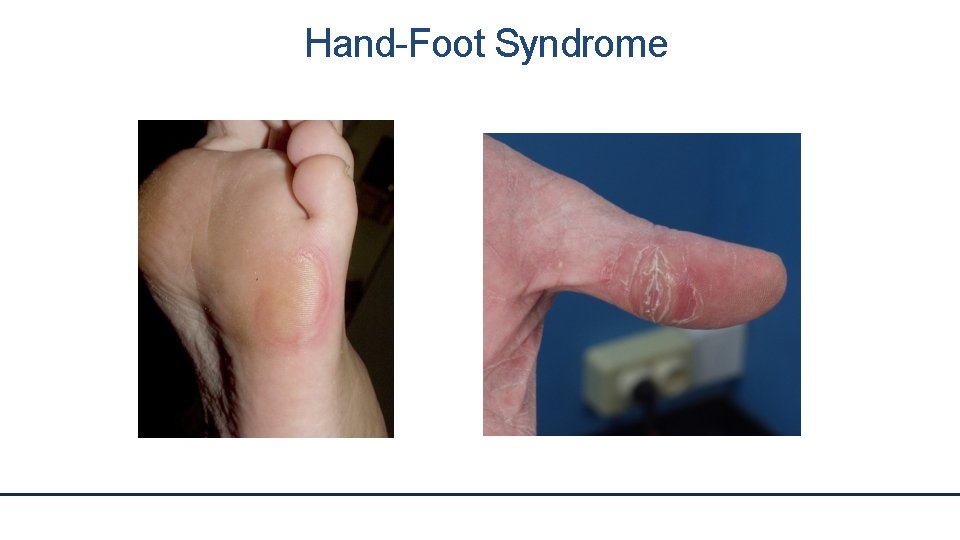
Hand-Foot Syndrome

**Preventive/Supportive Measures: Dermatologic • Hand-foot • Emollients, moisturizers • Topical exfoliating agents, urea containing lotions/creams • Soft/cushioned shoes; gel inserts • Consider podiatric care • Dose interruption/reduction • Rash • Moisturizers • Antidandruff shampoo • Topical antihistamine lotion/mild hydrocortisone ointment • Prevent bacterial superinfection; antibiotic gel • Mild, non-deodorant soaps; avoid hot shower/bath

Case #3 Patient’s course: • “B sx” resolved, adenopathy no longer palpable, scans showed reduced adenopathy and renal mass within 3 -6 months, and have remained stable • Completed 2 years of pembro (as per trial) and continues on axitinib 2 mg BID with intermittent dose interruption to manage HFS for QOL; stomatitis did not recur after down to 2 mg; myalgias resolved after off of pembro

Treatment break allowed trip to London and Italy

Genitourinary Cancer: m. RCC and m. UC Oncology Nursing Retreat Miami, FL November 9 th, 2019 David I Quinn MBBS (Hons) Ph. D FRACP FACP Associate Professor of Medicine Chief, Section of GU Medical Oncology Division of Medical Oncology Medical Director, Norris Cancer Hospital and Clinics Kenneth J. Norris Comprehensive Cancer Center Keck School of Medicine at University of Southern California diquinn@med. usc. edu
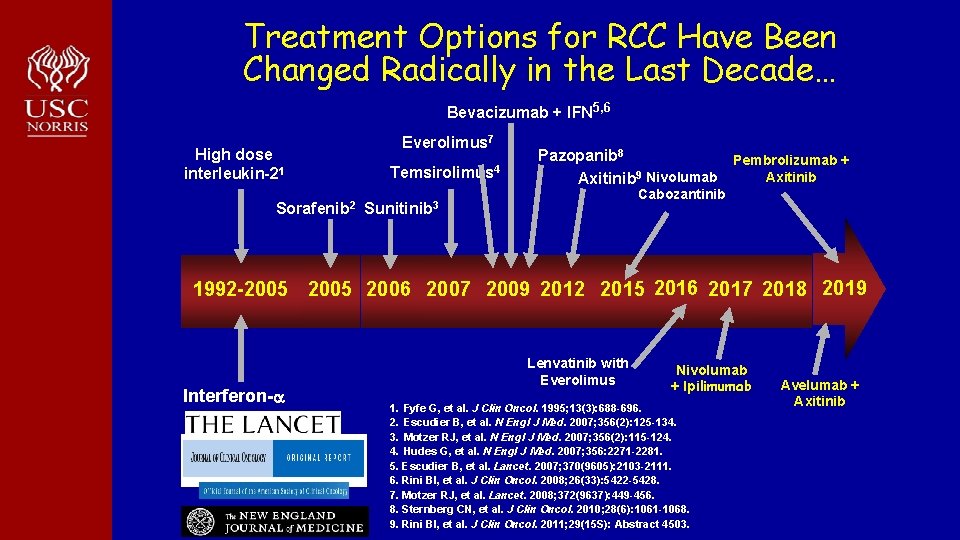
Treatment Options for RCC Have Been Changed Radically in the Last Decade… Bevacizumab + IFN 5, 6 High dose interleukin-21 Sorafenib 2 Everolimus 7 Temsirolimus 4 Pazopanib 8 Pembrolizumab + 9 Axitinib Nivolumab Cabozantinib Sunitinib 3 1992 -2005 2006 2007 2009 2012 2015 2016 2017 2018 2019 Interferon- Lenvatinib with Everolimus Nivolumab + Ipilimumab 1. Fyfe G, et al. J Clin Oncol. 1995; 13(3): 688 -696. 2. Escudier B, et al. N Engl J Med. 2007; 356(2): 125 -134. 3. Motzer RJ, et al. N Engl J Med. 2007; 356(2): 115 -124. 4. Hudes G, et al. N Engl J Med. 2007; 356: 2271 -2281. 5. Escudier B, et al. Lancet. 2007; 370(9605): 2103 -2111. 6. Rini BI, et al. J Clin Oncol. 2008; 26(33): 5422 -5428. 7. Motzer RJ, et al. Lancet. 2008; 372(9637): 449 -456. 8. Sternberg CN, et al. J Clin Oncol. 2010; 28(6): 1061 -1068. 9. Rini BI, et al. J Clin Oncol. 2011; 29(15 S): Abstract 4503. Avelumab + Axitinib

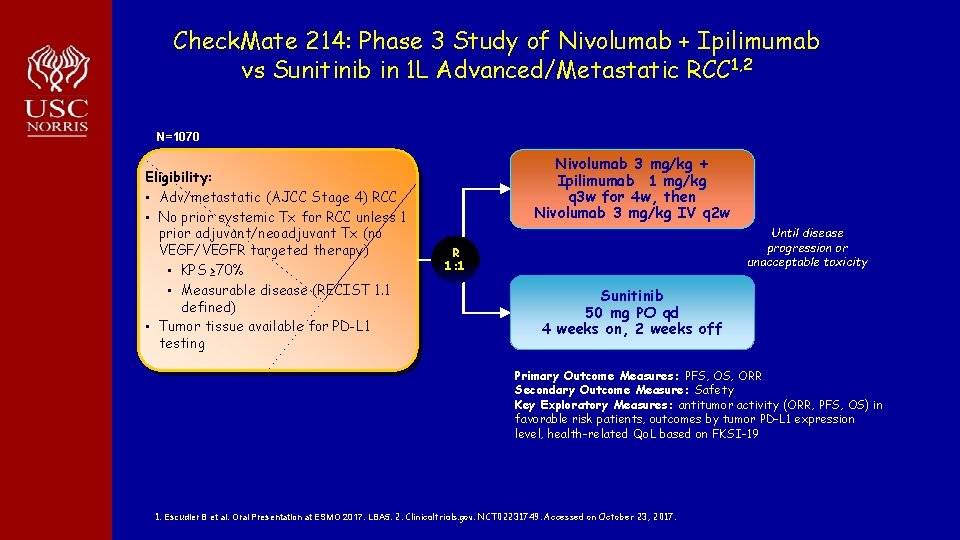
Check. Mate 214: Phase 3 Study of Nivolumab + Ipilimumab vs Sunitinib in 1 L Advanced/Metastatic RCC 1, 2 N=1070 Eligibility: • Adv/metastatic (AJCC Stage 4) RCC • No prior systemic Tx for RCC unless 1 prior adjuvant/neoadjuvant Tx (no VEGF/VEGFR targeted therapy) • KPS ≥ 70% • Measurable disease (RECIST 1. 1 defined) • Tumor tissue available for PD-L 1 testing Nivolumab 3 mg/kg + Ipilimumab 1 mg/kg q 3 w for 4 w, then Nivolumab 3 mg/kg IV q 2 w Until disease progression or unacceptable toxicity R 1: 1 Sunitinib 50 mg PO qd 4 weeks on, 2 weeks off Primary Outcome Measures: PFS, ORR Secondary Outcome Measure: Safety Key Exploratory Measures: antitumor activity (ORR, PFS, OS) in favorable risk patients, outcomes by tumor PD-L 1 expression level, health-related Qo. L based on FKSI-19 1. Escudier B et al. Oral Presentation at ESMO 2017. LBA 5. 2. Clinicaltrials. gov. NCT 02231749. Accessed on October 23, 2017.
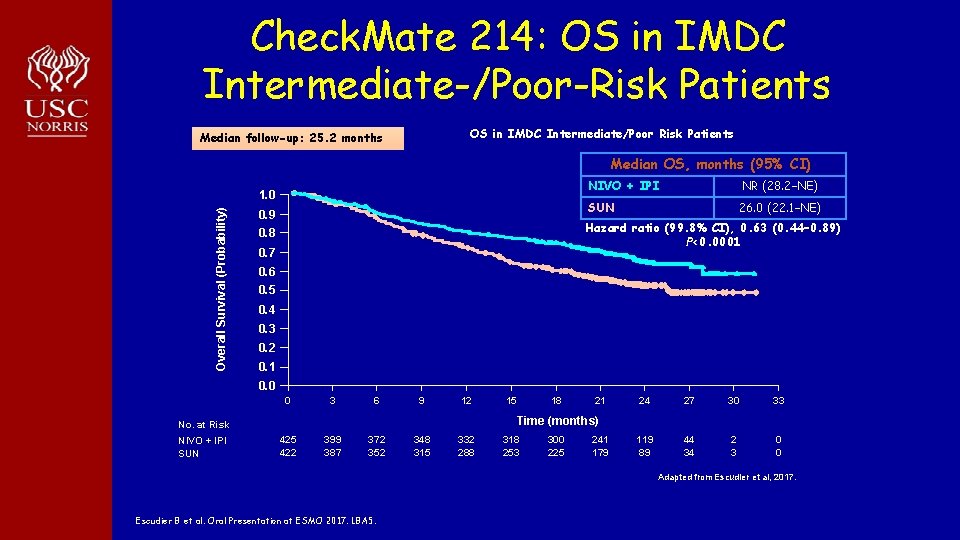
Check. Mate 214: OS in IMDC Intermediate-/Poor-Risk Patients OS in IMDC Intermediate/Poor Risk Patients Median follow-up: 25. 2 months Median OS, months (95% CI)) Overall Survival (Probability) 1. 0 0. 9 NIVO + IPI NR (28. 2–NE) SUN 26. 0 (22. 1–NE) Hazard ratio (99. 8% CI), 0. 63 (0. 44– 0. 89) P<0. 0001 0. 8 0. 7 0. 6 0. 5 0. 4 0. 3 0. 2 0. 1 0. 0 0 3 6 9 12 18 21 24 27 30 33 119 89 44 34 2 3 0 0 Time (months) No. at Risk NIVO + IPI SUN 15 422 399 387 372 352 348 315 332 288 318 253 300 225 241 179 Adapted from Escudier et al, 2017. Escudier B et al. Oral Presentation at ESMO 2017. LBA 5.
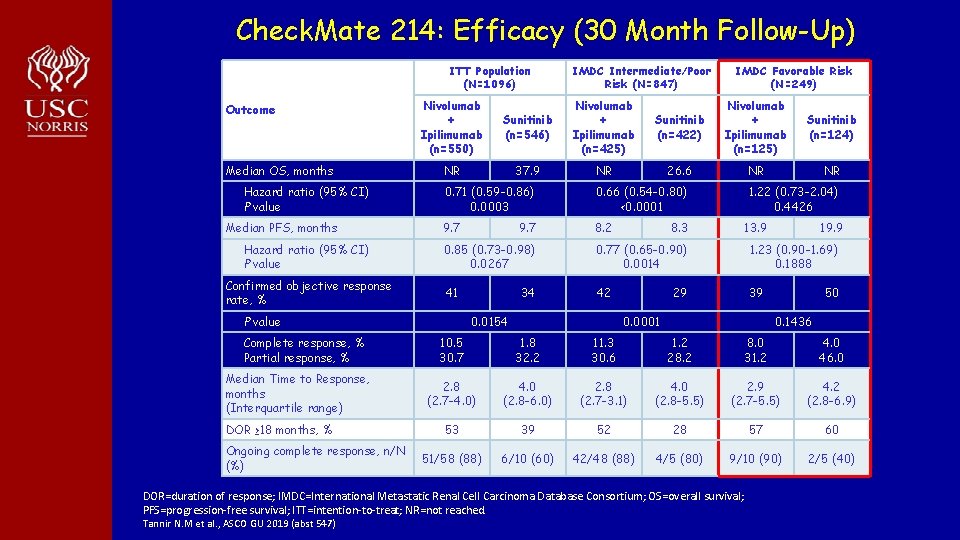
Check. Mate 214: Efficacy (30 Month Follow-Up) ITT Population (N=1096) Outcome Median OS, months Hazard ratio (95% CI) P value Median PFS, months Hazard ratio (95% CI) P value Confirmed objective response rate, % Nivolumab + Ipilimumab (n=550) NR Median Time to Response, months (Interquartile range) DOR ≥ 18 months, % Ongoing complete response, n/N (%) Sunitinib (n=546) Nivolumab + Ipilimumab (n=425) 37. 9 NR Sunitinib (n=422) Sunitinib (n=124) 26. 6 NR NR 0. 66 (0. 54 -0. 80) <0. 0001 9. 7 8. 2 9. 7 IMDC Favorable Risk (N=249) Nivolumab + Ipilimumab (n=125) 0. 71 (0. 59 -0. 86) 0. 0003 1. 22 (0. 73 -2. 04) 0. 4426 8. 3 13. 9 0. 77 (0. 65 -0. 90) 0. 0014 1. 23 (0. 90 -1. 69) 0. 1888 41 42 39 34 0. 0154 29 0. 0001 50 0. 1436 10. 5 30. 7 1. 8 32. 2 11. 3 30. 6 1. 2 28. 2 8. 0 31. 2 4. 0 46. 0 2. 8 (2. 7 -4. 0) 4. 0 (2. 8 -6. 0) 2. 8 (2. 7 -3. 1) 4. 0 (2. 8 -5. 5) 2. 9 (2. 7 -5. 5) 4. 2 (2. 8 -6. 9) 53 39 52 28 57 60 51/58 (88) 6/10 (60) 42/48 (88) 4/5 (80) 9/10 (90) 2/5 (40) DOR=duration of response; IMDC=International Metastatic Renal Cell Carcinoma Database Consortium; OS=overall survival; PFS=progression-free survival; ITT=intention-to-treat; NR=not reached. Tannir N. M et al. , ASCO GU 2019 (abst 547) 19. 9 0. 85 (0. 73 -0. 98) 0. 0267 P value Complete response, % Partial response, % IMDC Intermediate/Poor Risk (N=847)
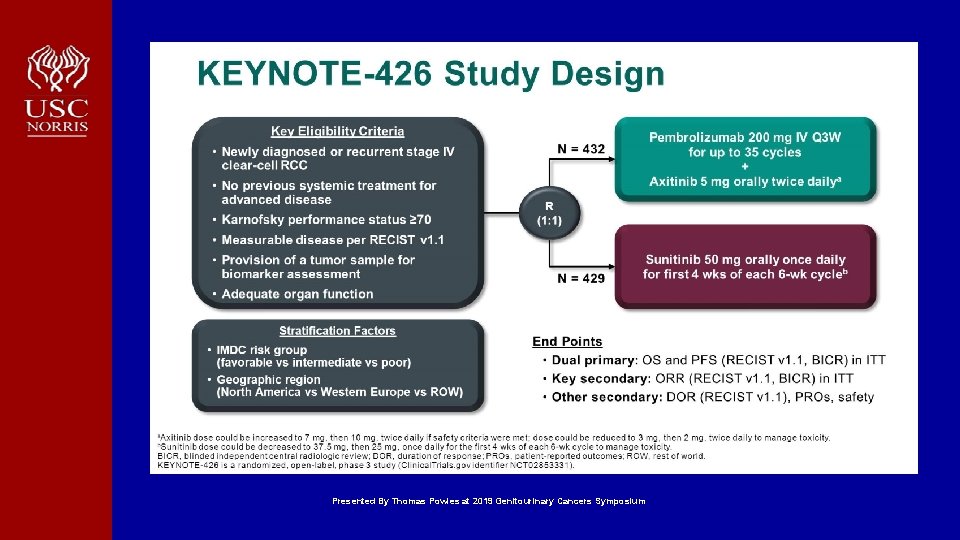
KEYNOTE-426 Study Design Presented By Thomas Powles at 2019 Genitourinary Cancers Symposium
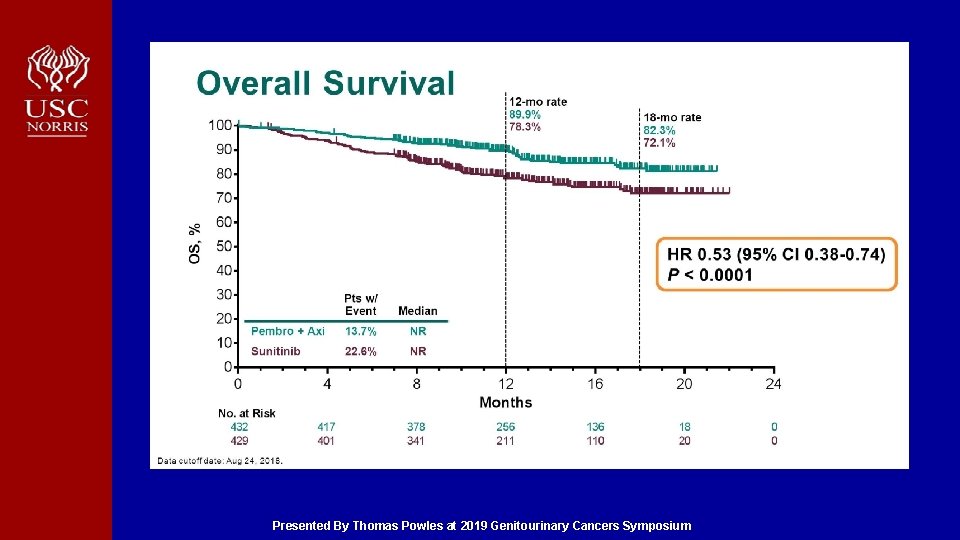
Overall Survival Presented By Thomas Powles at 2019 Genitourinary Cancers Symposium
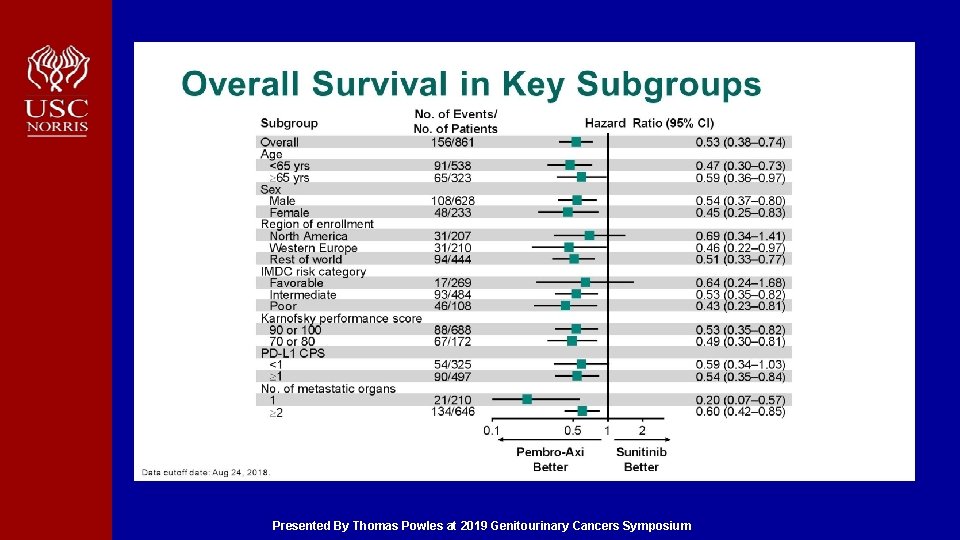
Overall Survival in Key Subgroups Presented By Thomas Powles at 2019 Genitourinary Cancers Symposium
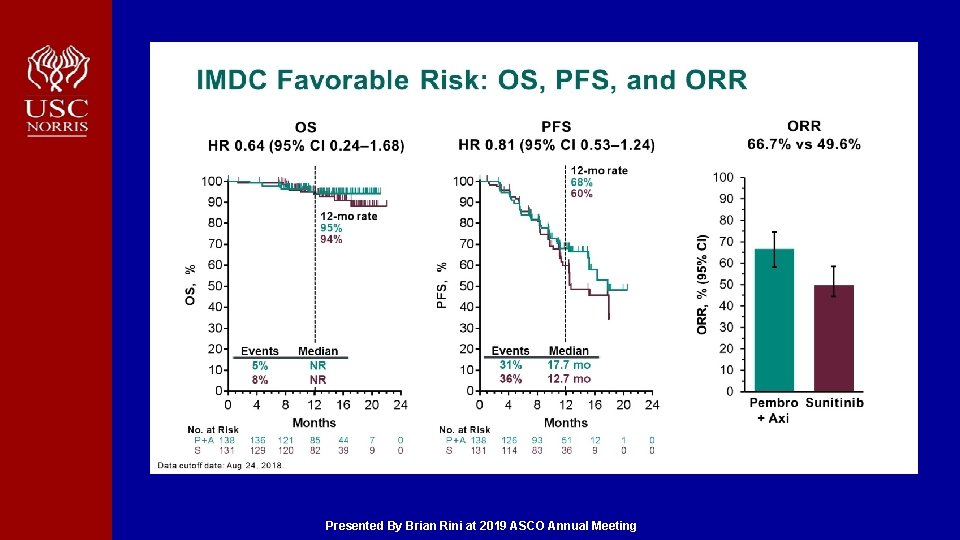
IMDC Favorable Risk: OS, PFS, and ORR Presented By Brian Rini at 2019 ASCO Annual Meeting
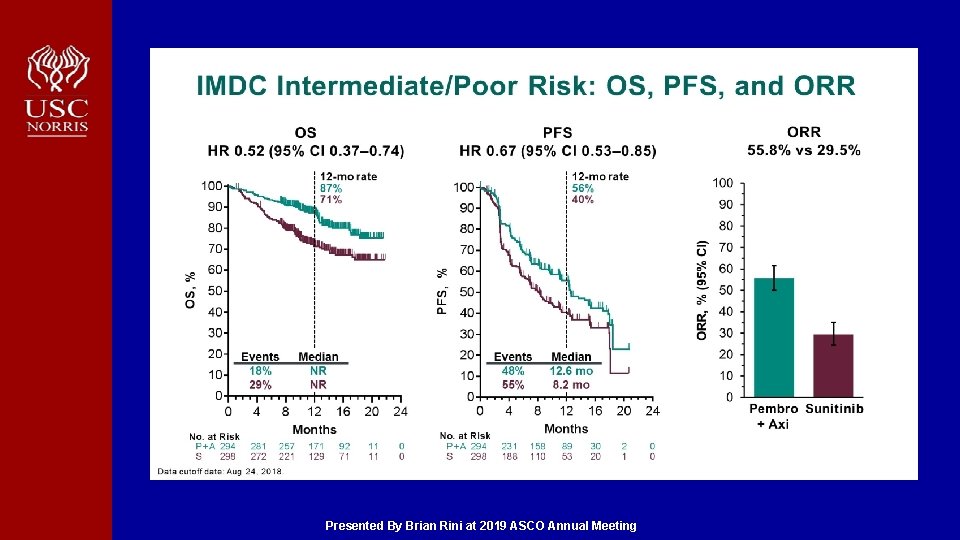
IMDC Intermediate/Poor Risk: OS, PFS, and ORR Presented By Brian Rini at 2019 ASCO Annual Meeting

JAVELIN RENAL 101: Phase III Study of Avelumab in Combination With Axitinib vs Sunitinib Monotherapy in Treatment-naïve Patients With Advanced RCC Avelumab 10 mg/kg IV q 2 w • • Selected Eligibility Criteria (N=886) Treatment-naïve, advanced RCC with a clear cell component ≥ 1 measurable lesion as defined by RECIST v 1. 1 Tumor tissue available for PD-L 1 staining ECOG PS 0 or 1 + Axitinib 5 mg po bid (6 -week cycle) R 1: 1 Sunitinib 50 mg po qd (4 weeks on/2 weeks off) Primary Endpoints: PFS assessed by IRC in PD-L 1+ group*, OS in PD-L 1+ group* Key Secondary Endpoints: PFS per IRC in overall population, OS in overall population Secondary Endpoints: ORR, DCR, TTR, DOR, safety, PK, PRO, ADA relationship between treatment response and tumor or blood biomarkers, immune-related PFS, ORR, DCR, TTR, and DOR per immune-related RECIST Stratification: ECOG PS (0 vs 1) and geographic region (US, Canada/Western Europe, rest of the world) Statistical Analyses: • For the primary PFS endpoint, 336 events would provide 90% power to detect a HR of 0. 65 at significance level of 0. 004. • A gatekeeping strategy, which tested the overall population only if PFS in the PD-L 1+ group was significant, was used to control for type 1 error. • Interim analysis was planned for approximately 235 PFS events (70% of total required PFS events) in the PD-L 1+ group. bid=twice a day; ECOG=Eastern Cooperative Oncology Group; PK=pharmacokinetics; PO=orally; PRO=patient-reported outcomes; qd=every day; RECIST=Response Evaluation Criteria in Solid Tumors; TKI=tyrosine kinase inhibitor. * ≥ 1% PD-L 1 expression on tumor-infiltrating immune cells using Ventana PD-L 1 (SP 263) IHC assay Motzer RJ, et al. Presented at ESMO. 2018 (abstr LBA 6_PR). https: //clinicaltrials. gov/ct 2/show/NCT 02684006
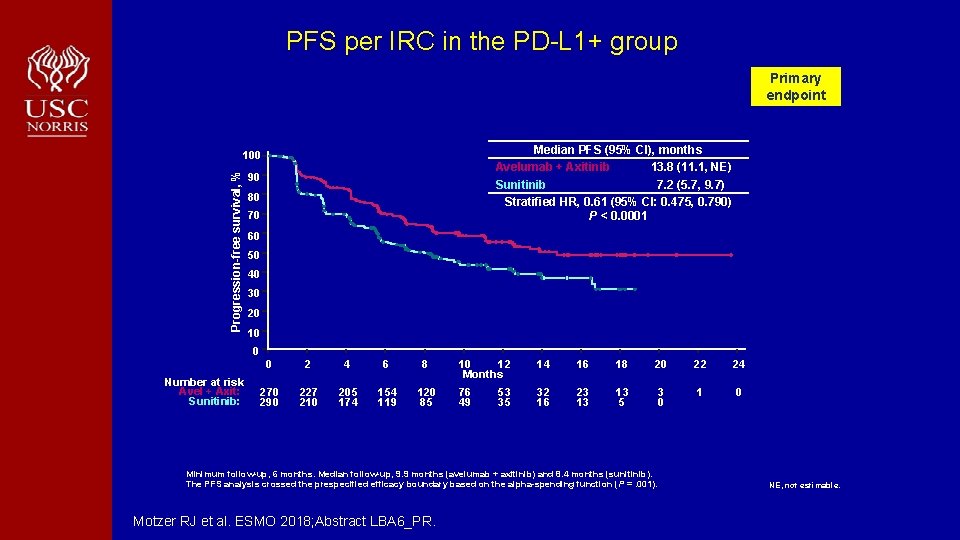
PFS per IRC in the PD-L 1+ group Primary endpoint Median PFS (95% CI), months Avelumab + Axitinib 13. 8 (11. 1, NE) Sunitinib 7. 2 (5. 7, 9. 7) Stratified HR, 0. 61 (95% CI: 0. 475, 0. 790) P < 0. 0001 Progression-free survival, % 100 90 80 70 60 50 40 30 20 10 0 Number at risk Avel + Axit: Sunitinib: 0 2 4 6 8 270 290 227 210 205 174 154 119 120 85 10 12 Months 14 16 18 20 22 24 76 49 32 16 23 13 13 5 3 0 1 0 53 35 Minimum follow-up, 6 months. Median follow-up, 9. 9 months (avelumab + axitinib) and 8. 4 months (sunitinib). The PFS analysis crossed the prespecified efficacy boundary based on the alpha-spending function (P =. 001). Motzer RJ et al. ESMO 2018; Abstract LBA 6_PR. NE, not estimable.
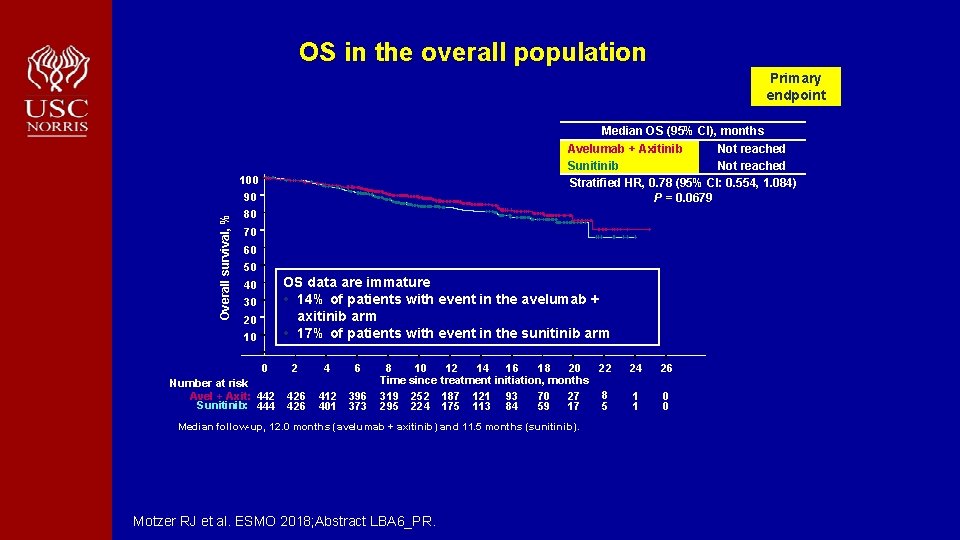
OS in the overall population Primary endpoint Median OS (95% CI), months Avelumab + Axitinib Not reached Sunitinib Not reached Stratified HR, 0. 78 (95% CI: 0. 554, 1. 084) P = 0. 0679 100 Overall survival, % 90 80 70 60 50 OS data are immature • 14% of patients with event in the avelumab + axitinib arm • 17% of patients with event in the sunitinib arm 40 30 20 10 0 Number at risk Avel + Axit: 442 Sunitinib: 444 2 4 6 426 412 401 396 373 8 10 12 14 16 18 20 22 Time since treatment initiation, months 8 319 252 187 121 93 70 27 295 224 175 113 84 59 17 5 Median follow-up, 12. 0 months (avelumab + axitinib) and 11. 5 months (sunitinib). Motzer RJ et al. ESMO 2018; Abstract LBA 6_PR. 24 26 1 1 0 0
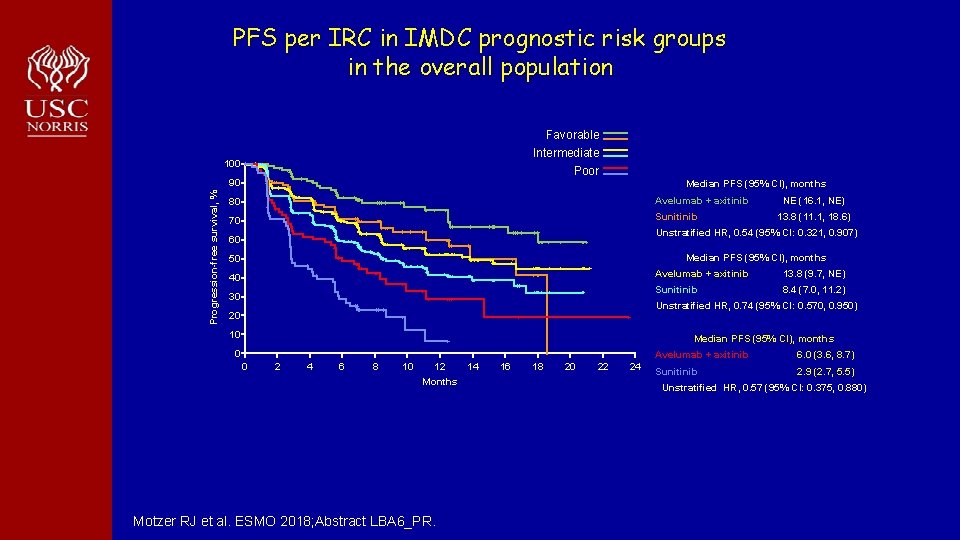
PFS per IRC in IMDC prognostic risk groups in the overall population Favorable Intermediate 100 Poor Progression-free survival, % 90 Median PFS (95% CI), months 80 Avelumab + axitinib 70 Sunitinib NE (16. 1, NE) 13. 8 (11. 1, 18. 6) Unstratified HR, 0. 54 (95% CI: 0. 321, 0. 907) 60 Median PFS (95% CI), months 50 40 30 Avelumab + axitinib 13. 8 (9. 7, NE) Sunitinib 8. 4 (7. 0, 11. 2) Unstratified HR, 0. 74 (95% CI: 0. 570, 0. 950) 20 10 Median PFS (95% CI), months 0 0 2 4 6 8 10 12 Months Motzer RJ et al. ESMO 2018; Abstract LBA 6_PR. 14 16 18 20 22 24 Avelumab + axitinib 6. 0 (3. 6, 8. 7) Sunitinib 2. 9 (2. 7, 5. 5) Unstratified HR, 0. 57 (95% CI: 0. 375, 0. 880)
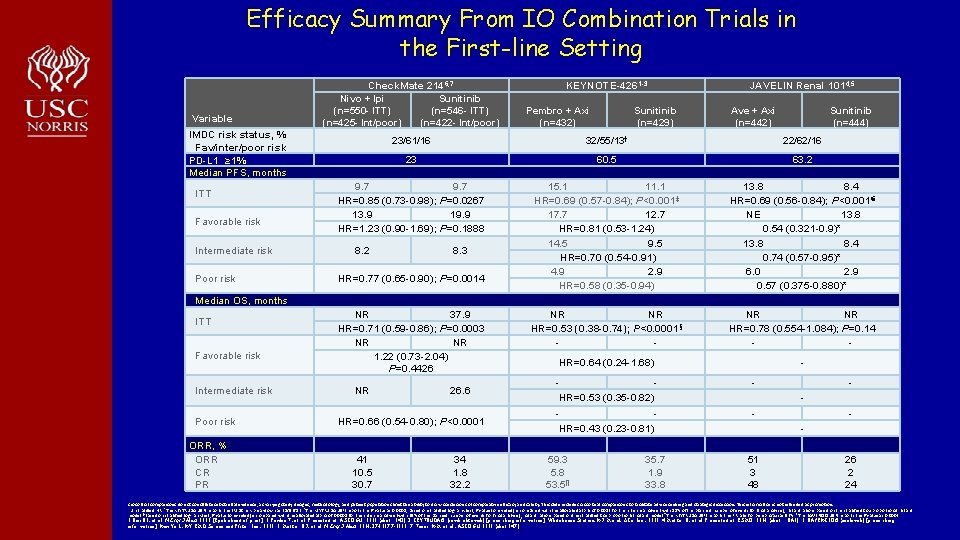
Efficacy Summary From IO Combination Trials in the First-line Setting Variable Check. Mate 2146, 7 Nivo + Ipi Sunitinib (n=550 - ITT) (n=546 - ITT) (n=425 - Int/poor) (n=422 - Int/poor) IMDC risk status, % Fav/inter/poor risk PD-L 1 ≥ 1% Median PFS, months ITT Favorable risk Intermediate risk Poor risk KEYNOTE-4261 -3 Pembro + Axi (n=432) Sunitinib (n=429) JAVELIN Renal 1014, 5 Ave + Axi (n=442) Sunitinib (n=444) 23/61/16 32/55/13† 22/62/16 23 60. 5 63. 2 9. 7 HR=0. 85 (0. 73 -0. 98); P=0. 0267 13. 9 19. 9 HR=1. 23 (0. 90 -1. 69); P=0. 1888 15. 1 11. 1 HR=0. 69 (0. 57 -0. 84); P<0. 001‡ 17. 7 12. 7 HR=0. 81 (0. 53 -1. 24) 14. 5 9. 5 HR=0. 70 (0. 54 -0. 91) 4. 9 2. 9 HR=0. 58 (0. 35 -0. 94) 13. 8 8. 4 HR=0. 69 (0. 56 -0. 84); P<0. 001€ NE 13. 8 0. 54 (0. 321 -0. 9)* 13. 8 8. 4 0. 74 (0. 57 -0. 95)* 6. 0 2. 9 0. 57 (0. 375 -0. 880)* NR NR HR=0. 53 (0. 38 -0. 74); P<0. 0001§ - NR NR HR=0. 78 (0. 554 -1. 084); P=0. 14 - HR=0. 64 (0. 24 -1. 68) - 8. 2 8. 3 HR=0. 77 (0. 65 -0. 90); P=0. 0014 Median OS, months ITT Favorable risk Intermediate risk Poor risk ORR, % ORR CR PR NR 37. 9 HR=0. 71 (0. 59 -0. 86); P=0. 0003 NR NR 1. 22 (0. 73 -2. 04) P=0. 4426 NR 26. 6 HR=0. 66 (0. 54 -0. 80); P<0. 0001 41 10. 5 30. 7 34 1. 8 32. 2 HR=0. 53 (0. 35 -0. 82) HR=0. 43 (0. 23 -0. 81) 59. 3 5. 8 53. 5¶ 35. 7 1. 9 33. 8 - - 51 3 48 26 2 24 Cross-trial comparisons do not constitute substantial evidence, as varying study designs, methodology, and patient populations limit the ability to draw conclusions of comparative efficacy and safety. This slide includes cross-trial comparisons to facilitate advice seeking and strategic discussion. This information is not intended as promotion. *Unstratified HR. †The KEYTRUDA USPI reports this IMDC risk breakdown as 31/56/13. ‡The KEYTRUDA USPI reports the P value as 0. 0001; based on stratified log-rank test; P value (one-sided) is compared with the allocated alpha of 0. 0013 for this interim analysis (with 81% of the planned number of events for final analysis); hazard ratio is based on the stratified Cox proportional hazard model. §Based on stratified log-rank test; P value (one-sided) is compared with the allocated alpha of 0. 0001 for this interim analysis (with 39% of the planned number of events for final analysis); hazard ratio is based on the stratified Cox proportional hazard model. ¶The KEYTRUDA USPI reports the PR rate for pembro/axi as 53%. €The BAVENCIO USPI reports the P value as 0. 0002. 1. Rini BI, et al. N Engl J Med. 2019. [Epub ahead of print]. 2. Powles T, et al. Presented at: ASCO GU. 2019 (abstr 543). 3. KEYTRUDA® (pembrolizumab) [prescribing information]. Whitehouse Station, NJ: Merck & Co Inc. ; 2019. 4. Motzer R, et al. Presented at: ESMO. 2018 (abstr LBA 6). 5. BAVENCIO® (avelumab) [prescribing information]. New York, NY: EMD Serono and Pfizer Inc. ; 2019. 6. Motzer RJ, et al. N Engl J Med. 2018; 378: 1277 -1290. 7. Tannir N. M et al. , ASCO GU 2019 (abst 547).
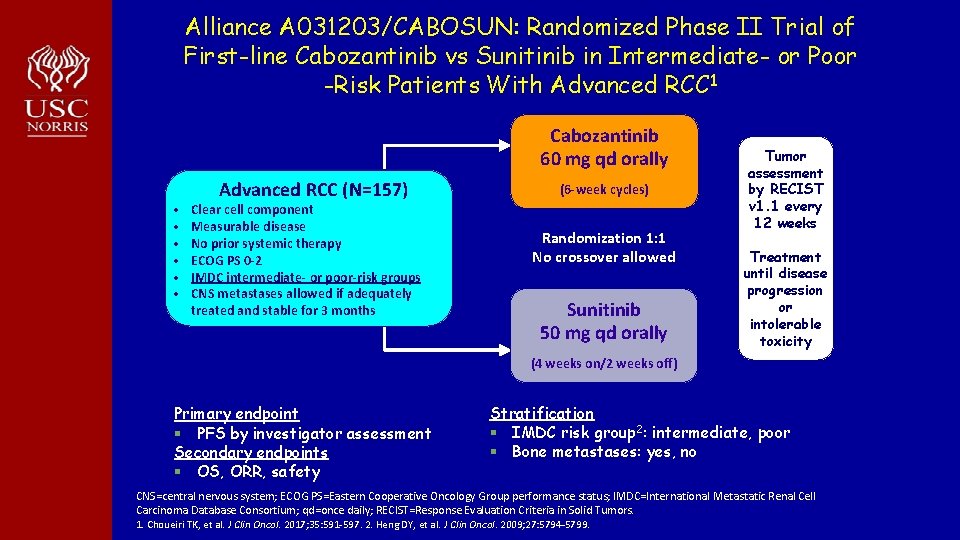
Alliance A 031203/CABOSUN: Randomized Phase II Trial of First-line Cabozantinib vs Sunitinib in Intermediate- or Poor -Risk Patients With Advanced RCC 1 Cabozantinib 60 mg qd orally • • • Advanced RCC (N=157) Clear cell component Measurable disease No prior systemic therapy ECOG PS 0 -2 IMDC intermediate- or poor-risk groups CNS metastases allowed if adequately treated and stable for 3 months (6 -week cycles) Randomization 1: 1 No crossover allowed Sunitinib 50 mg qd orally Tumor assessment by RECIST v 1. 1 every 12 weeks Treatment until disease progression or intolerable toxicity (4 weeks on/2 weeks off) Primary endpoint § PFS by investigator assessment Secondary endpoints § OS, ORR, safety Stratification § IMDC risk group 2: intermediate, poor § Bone metastases: yes, no CNS=central nervous system; ECOG PS=Eastern Cooperative Oncology Group performance status; IMDC=International Metastatic Renal Cell Carcinoma Database Consortium; qd=once daily; RECIST=Response Evaluation Criteria in Solid Tumors. 1. Choueiri TK, et al. J Clin Oncol. 2017; 35: 591 -597. 2. Heng DY, et al. J Clin Oncol. 2009; 27: 5794 -5799.
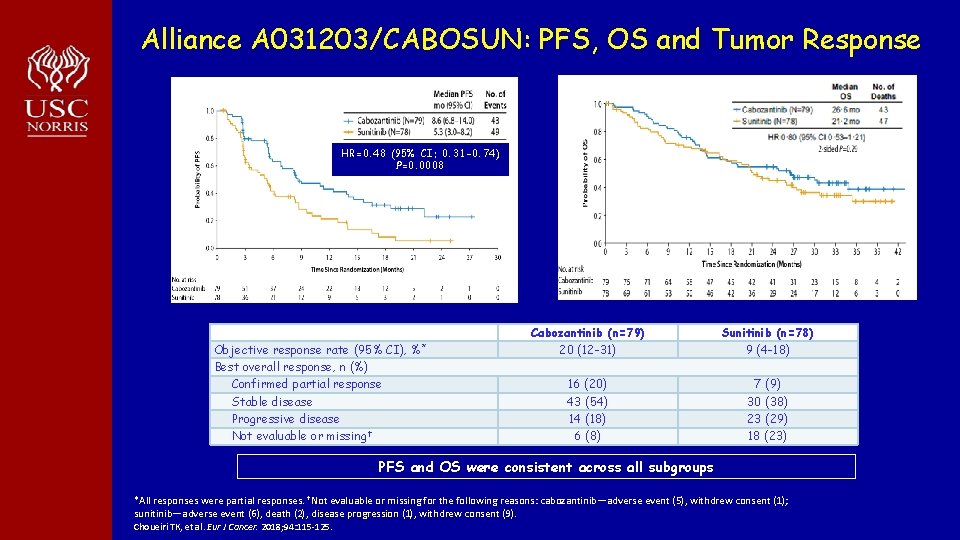
Alliance A 031203/CABOSUN: PFS, OS and Tumor Response Median PFS per IRC HR=0. 48 (95% CI: 0. 31 -0. 74) P=0. 0008 Objective response rate (95% CI), Best overall response, n (%) Confirmed partial response Stable disease Progressive disease Not evaluable or missing† %* Cabozantinib (n=79) 20 (12 -31) Sunitinib (n=78) 9 (4 -18) 16 (20) 43 (54) 14 (18) 6 (8) 7 (9) 30 (38) 23 (29) 18 (23) PFS and OS were consistent across all subgroups *All responses were partial responses. †Not evaluable or missing for the following reasons: cabozantinib—adverse event (5), withdrew consent (1); sunitinib—adverse event (6), death (2), disease progression (1), withdrew consent (9). Choueiri TK, et al. Eur J Cancer. 2018; 94: 115 -125.
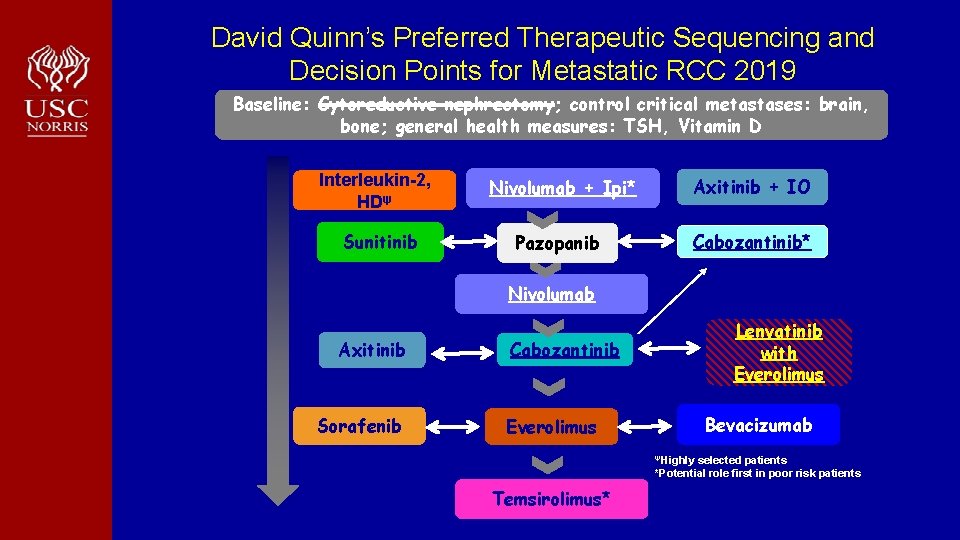
David Quinn’s Preferred Therapeutic Sequencing and Decision Points for Metastatic RCC 2019 Baseline: Cytoreductive nephrectomy; control critical metastases: brain, bone; general health measures: TSH, Vitamin D Interleukin-2, HDψ Sunitinib Nivolumab + Ipi* Axitinib + IO Pazopanib Cabozantinib* Nivolumab Axitinib Sorafenib Lenvatinib with Everolimus Cabozantinib Bevacizumab Everolimus ΨHighly selected patients *Potential role first in poor risk patients Temsirolimus*

Metastatic Renal cell cancer: First line Summary In 2019 Most first line choices involve a CPI either IO + IO or IO + axitinib The decision between these approaches relates to relative efficacy and toxicity from therapy at different time points There is a role for VEGFr. TKI alone in patients not suitable for IO therapy possibly in good risk patients Cabozantinib in patients with bone metastases Observation and HDIL 2 also have roles in selected patients

Urothelial Bladder Cancer

Case study #1 Metastatic urothelial carcinoma (MUC) • 63 yo man presented with gross hematuria 2/2016, found to have left upper tract (ureteropelvic junction) urothelial carcinoma with extensive adenopathy, involving the retroperitoneum, left supraclavicular, and left distal paraesophageal region. • Treated with 6 cycles gemcitabine and cisplatin with excellent response, but still residual disease; also developed significant neuropathy. • Presented to us for enrollment in a Phase II clinical trial of maintenance pembrolizumab vs placebo, after first line chemotherapy. Started 8/2016 • PMH: HTN, GERD, chronic sinusitis • Social: Works in maintenance; lives >100 miles from us

Case #1 MUC • Treatment started on trial 8/2016, Q 3 weeks • Scans 4/2017 showed progressive disease, study unblinded and he transitioned to treatment with pembrolizumab. • PMH: GERD, hyperlipidemia, HTN, chemo-induced peripheral neuropathy, chronic sinusitis, former smoker. • Social: married, lives with wife; works in maintenance. Lives >100 miles from us!
110 miles

: Patient Education with immunotherapies • Establish means of communication—and reconfirm! • Stress importance of calling with new onset symptoms: • Cough, chest pain, dyspnea • Diarrhea or severe abdominal pain • Severe nausea/vomiting, right sided abdominal pain, jaundice, easy bruising/bleeding • New onset fatigue, palpitations, hair loss, skin changes, increased thirst • Fever, urinary tract infection symptoms Reminders!! And for 63 yo patient specifically: Side effects differ from traditional chemotherapy and often treatable, however though are overall less common, side effects can occur and be severe

Immunotherapy Toxicities • Rash • Anorexia • Nausea/Vomiting • Fatigue • Elevated LFTs • Arthralgias/Myalgias • Hyper/Hypothyroidism

Immune Related Adverse Effects

Name an –itis, Any -itis Organ System Reported Toxicities Integumentary Hives, Eczema, Vitiligo, Pemphigus, Lichenoid Reactions Gastrointestinal Enterocolitis, Pancreatitis, Gastritis, Celiac Disease Hepatic Autoimmune Hepatitis, Sclerosing Cholangitis, Primary Biliary Cirrhosis Renal Interstitial Nephritis, Nephrotic Syndrome, Autoimmune Nephropathy Pulmonary Pneumonitis, Interstitial Lung Disease, Pleuritis Cardiac Myocarditis, Pericarditis, Cardiomyopathy Endocrine Hypo/Hyperthyroidism, Hypophysitis, Adrenal Insufficiency Neurologic Encephalitis, Guillain-Barre Syndrome, Myasthenia Gravis, Mononeuritis, Autoimmune inner ear disease Hematologic Hemolytic Anemia, Immune Thrombocytopenic Purpura, Thrombotic Thrombocytopenic Purpura, Hemophilia, Evans Syndrome Rheumatologic Polyarthritis, Systemic Lupus Erythematosus, Antiphospholipid Syndrome

RED FLAGS • Any new signs or symptoms – Most important: cough, diarrhea, rash, extreme fatigue, headache, chest pain • New onset sign or symptom impacting daily living in any way • Labs – – Creatinine >1. 5 x over baseline AST/ALT >3 x ULN and/or Tbili >1. 5 x ULN Glucose >200 Do NOT need to act immediately for abnormal TSH

Management of Immune Related AEs • Immune related AEs typically occur within the first 32 weeks of tx; most within the first 16 weeks, but can occur at any time • No evidence that intervening with steroids curtails antitumor efficacy of agent



Patient Chart Advisories Patient name, DOB, MRN

Case Study #1 • By 4/2017, patient had disease progression on scans, so trial was unblinded, and he was started on pembrolizumab • Developed progressive joint aches, could be due to treatment or OA • 6 months later, developed pruritic rash on arms, upper chest, neck • Also episodes of sinusitis with cough and sometimes DOE– evaluated more than once!
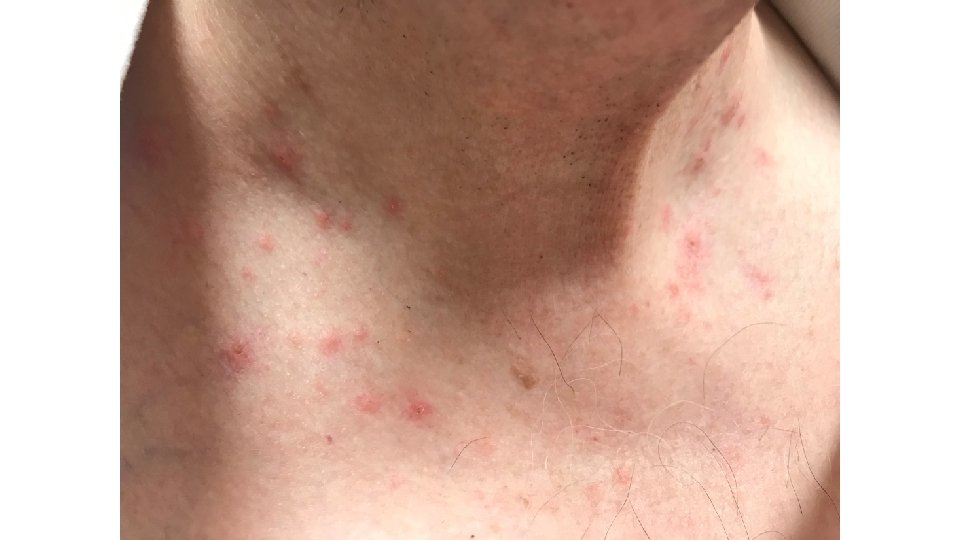
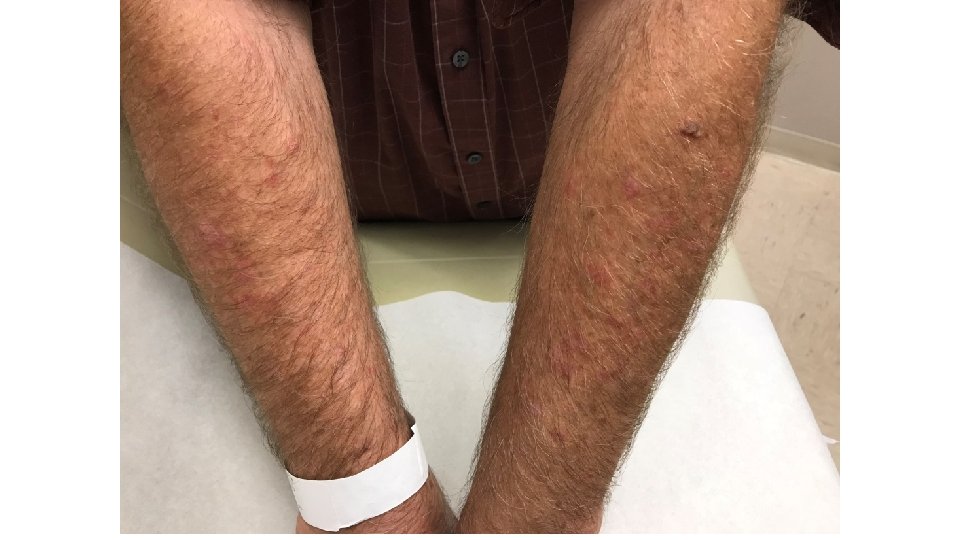

Case Study #1 • Triamcinolone 0. 1% cream prescribed for rash; faded, waxed and waned over time • Completed 2 years treatment 4/2019, and now on surveillance • CT C/A/P 8/2019: 1. Stable urothelial thickening of the left renal pelvis and most proximal ureter which is presumably the patient's treated urothelial carcinoma. 2. Stable treated mediastinal, retroperitoneal, and iliac chain metastatic lymphadenopathy. 3. No new metastatic disease to the chest, abdomen, or pelvis.
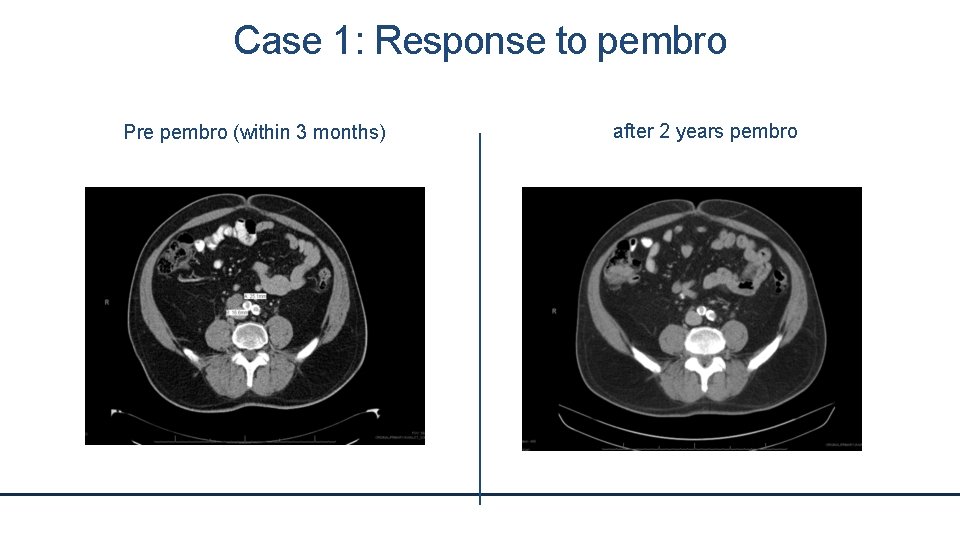
Case 1: Response to pembro Pre pembro (within 3 months) after 2 years pembro

Genitourinary Cancer: m. RCC and m. UC Oncology Nursing Retreat Miami, FL November 9 th, 2019 David I Quinn MBBS (Hons) Ph. D FRACP FACP Associate Professor of Medicine Chief, Section of GU Medical Oncology Division of Medical Oncology Medical Director, Norris Cancer Hospital and Clinics Kenneth J. Norris Comprehensive Cancer Center Keck School of Medicine at University of Southern California diquinn@med. usc. edu
Timeline for systemic therapy development in Nivolumab urothelial cancer… Durvalumab Avelumab Bajorin Criteria 12 Taxanes 4 Methotrexate Cisplatin 1976 Pembrolizumab Neoadjuvant MVAC KN-04514 & phase III SWOG 871010 KN-052 MVAC Phase IIIs vs. CDDP Gemcitabine 6 1 MVAC & CISCA 3 Erdafitinib Vinflunine 11 1981 1990 1985 1993 1998 2000 2003 20092016 20172018 -82 -92 Doxorubicin CMV 2 Ifosfamide 5 Trimodality Chemo Vinblastine dd. MVAC vs. MVAC phase III 9 -radiation RTOG GC vs. MVAC 89 -037 phase III 8 Im. Vigor 210 cohort 1 & 2 Atezolizumab 13 . . . limited progress in the last decade … until recently 1. Sternberg CN et al. J Urol 133: 403 -7, 1985 2. Harker WG et al. J Clin Oncol 3: 1463 -70, 1985 3. Logothetis CJ et al. J Clin Oncol 8: 1050 -5, 1990 4. Roth BJ et al J Clin Oncol 12: 2264 -70, 1994 5. Witte RS et al J Clin Oncol 15: 589 -93, 1997 6. Stadler WM et al J Clin Oncol 15: 3394 -8, 1997 7. Shipley WU et al. J Clin Oncol 16: 3576 -83, 1998 8. Von der Maase H. et al. J Clin Oncol 18: 3068, 2000 9. Sternberg CN et al. J Clin Oncol 19: 2638 -1646, 2001 10. Grossman HB. et al. N Engl J Med 349: 859 -66, 2003 11. Bellmunt J et al. J Clin Oncol 10: 1850 -5, 2009 12. Bajorin DF et al J Clin Oncol 17: 3173 -81, 1999 13. Rosenberg J et al Lancet 2016 14. Bellmunt J et al. N Engl J Med 2017 Enfortumab vedotin Ramucirumab

KEYNOTE-045 Study Design (NCT 02256436) Key Eligibility Criteria • Urothelial carcinoma of the renal pelvis, ureter, bladder, or urethra • Transitional cell predominant • PD after 1 -2 lines of platinum-based chemotherapy or recurrence <12 mo after perioperative platinum-based therapy • ECOG performance status 0 -2 • Provision of tumor sample for biomarker assessment • • a. In Stratification Factors ECOG performance status (0/1 vs 2) Hemoglobin level (<10 vs ≥ 10 g/d. L) Liver metastases (yes vs no) Time from last chemotherapy dose (<3 vs ≥ 3 mo) Pembrolizumab 200 mg IV Q 3 W R 1: 1 Paclitaxel 175 mg/m 2 Q 3 W OR Docetaxel 75 mg/m 2 Q 3 W OR Vinflunine 320 mg/m 2 Q 3 W • Dual primary end points: OS and PFS a • Key secondary end points: ORR, DOR, safety • Response: RECIST v 1. 1 by blinded, independent central review • Both unselected and biomarker-selected patients total ITT population and in patients with combined positive score ≥ 10%.
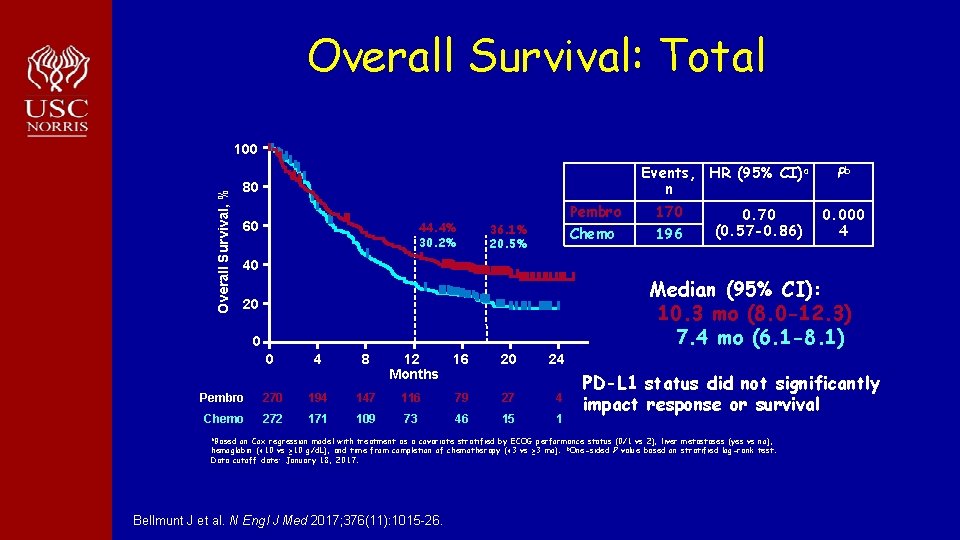
Overall Survival: Total Overall Survival, % 100 80 60 44. 4% 30. 2% Pembro Chemo 36. 1% 20. 5% Events, HR (95% CI)a Pb n 170 0. 000 (0. 57 -0. 86) 4 196 40 Median (95% CI): 10. 3 mo (8. 0 -12. 3) 7. 4 mo (6. 1 -8. 1) 20 0 0 4 8 12 Months 16 20 24 Pembro 270 194 147 116 79 27 4 Chemo 272 171 109 73 46 15 1 a. Based PD-L 1 status did not significantly impact response or survival on Cox regression model with treatment as a covariate stratified by ECOG performance status (0/1 vs 2), liver metastases (yes vs no), hemoglobin (<10 vs ≥ 10 g/d. L), and time from completion of chemotherapy (<3 vs ≥ 3 mo). b. One-sided P value based on stratified log-rank test. Data cutoff date: January 18, 2017. Bellmunt J et al. N Engl J Med 2017; 376(11): 1015 -26.
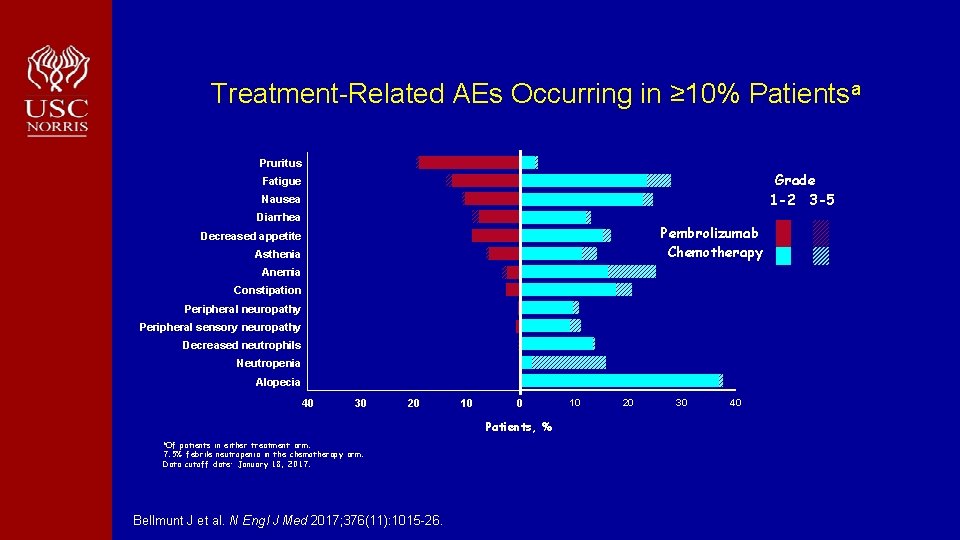
Treatment-Related AEs Occurring in ≥ 10% Patientsa Pruritus Grade 1 -2 3 -5 Fatigue Nausea Diarrhea Pembrolizumab Chemotherapy Decreased appetite Asthenia Anemia Constipation Peripheral neuropathy Peripheral sensory neuropathy Decreased neutrophils Neutropenia Alopecia 40 30 20 10 0 Patients, % a. Of patients in either treatment arm. 7. 5% febrile neutropenia in the chemotherapy arm. Data cutoff date: January 18, 2017. Bellmunt J et al. N Engl J Med 2017; 376(11): 1015 -26. 10 20 30 40
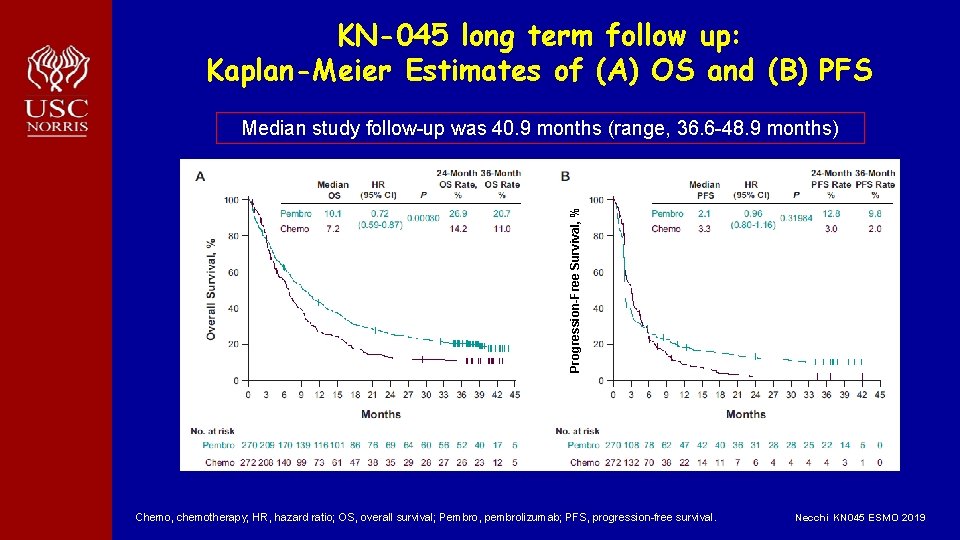
KN-045 long term follow up: Kaplan-Meier Estimates of (A) OS and (B) PFS Progression-Free Survival, % Median study follow-up was 40. 9 months (range, 36. 6 -48. 9 months) Chemo, chemotherapy; HR, hazard ratio; OS, overall survival; Pembro, pembrolizumab; PFS, progression-free survival. Necchi KN 045 ESMO 2019
First line trials
![IMvigor 210: Study Design § Single-arm phase II study with 2 cohorts[1] Pts with IMvigor 210: Study Design § Single-arm phase II study with 2 cohorts[1] Pts with](http://slidetodoc.com/presentation_image_h2/688c6c28c9e54a04277f2fc8cd5ddd33/image-62.jpg)
IMvigor 210: Study Design § Single-arm phase II study with 2 cohorts[1] Pts with inoperable advanced or metastatic UC, predominantly TCC histology, evaluable tumor tissue for PD-L 1 testing (N = 429) § Cohort 1 study Cohort 1 (n = 119)[2]: previously untreated, cisplatin ineligible* Atezolizumab 1200 mg IV Q 3 W until PD Cohort 2 (n = 310)[3]: prior platinum treatment Atezolizumab 1200 mg IV Q 3 W until loss of benefit *≥ 1 of the following: ECOG PS 2; grade ≥ 2 hearing loss or peripheral neuropathy; renal impairment (e. GFRCG: > 30, < 60 m. L/min). – Primary endpoint: confirmed ORR by RECIST v 1. 1 (per central, independent review) – Secondary endpoints: Do. R, PFS, OS, safety 1. Clinical. Trials. gov. NCT 02108652. 2. Balar AV, et al. ASCO 2016. Abstract LBA 4500. 3. Dreicer R, et al. ASCO 2016. Abstract 4515.

First line cisplatin ineligible IMvigor 210 (Cohort 1): Response Outcome IC 0 (n = 39) IC 1 (n = 48) IC 2/3 (n = 32) All Pts (N = 119) ORR, * % § CR § PR 21 8 13 23 6 17 28 6 22 24 7 17 Median time to response, † mos 2. 0 (1. 8 -6. 0) 2. 4 (1. 8 -4. 8) 2. 1 (2. 0 -4. 1) -- Reduced tumor burden, % 63 (n = 30) 51 (n = 39) 65 (n = 26) 59 (n = 95) Higher PD-L 1 expression • Median Do. R not reached in any PD-L 1 IC subgroup DID NOT appear to • Responses seen in all analyzed subgroups including poor enrich response baseline prognostic factors, for pts 80 yrs of age or older *19 pts with missing/unevaluable responses included in ORR analysis. †Responders only. ‒ ORR similar for all cisplatin-ineligible criteria subgroups Balar AV, et al. ASCO 2016. Abstract LBA 4500.
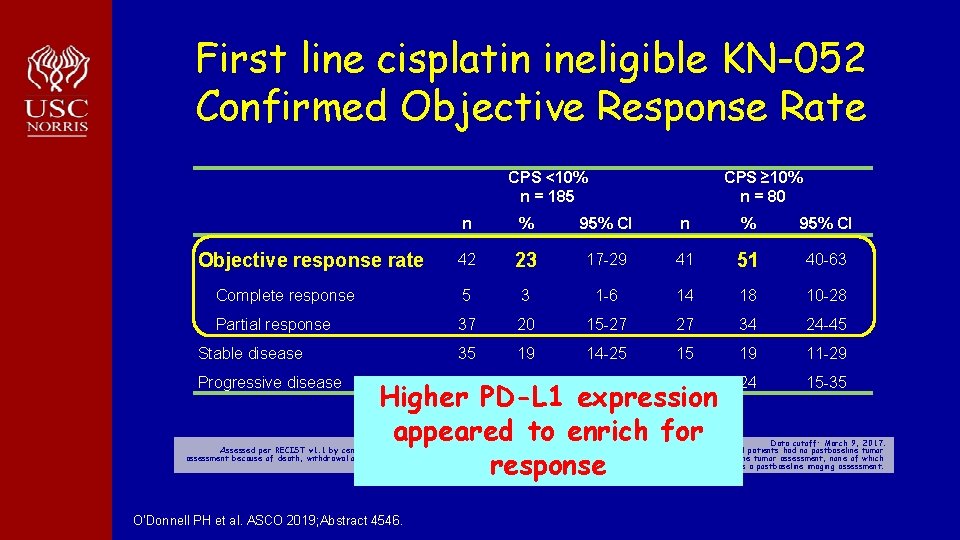
First line cisplatin ineligible KN-052 Confirmed Objective Response Rate CPS <10% n = 185 CPS ≥ 10% n = 80 n % 95% CI 42 23 17 -29 41 51 40 -63 Complete response 5 3 1 -6 14 18 10 -28 Partial response 37 20 15 -27 27 34 24 -45 Stable disease 35 19 14 -25 15 19 11 -29 Progressive disease 86 47 37 -54 19 24 15 -35 Objective response rate Higher PD-L 1 expression appeared to enrich for response Data cutoff: March 9, 2017. Assessed per RECIST v 1. 1 by central imaging vendor review. 361/370 patients had CPS and ORR data. For CPS <10%, 17 additional patients had no postbaseline tumor assessment because of death, withdrawal of consent, loss to follow-up, or start of new anticancer therapy, and 5 patients had ≥ 1 postbaseline tumor assessment, none of which were evaluable. For CPS ≥ 10%, 5 additional patients did not have a postbaseline imaging assessment. O'Donnell PH et al. ASCO 2019; Abstract 4546.

Combination trials

IMvigor 130 (WO 30070): Phase III Atezolizumab vs Atezolizumab + Platinum-Based Chemotherapy in Untreated Locally Advanced or Metastatic UC – Study Design Atezolizumab† + carboplatin, ‡ gemcitabine§ or Atezolizumab† + cisplatin, || gemcitabine§ Locally advanced or metastatic UC (Estimated N=1200*) R 1: 1: 1 Atezolizumab† Disease progression¶/ unacceptable toxicity# Placebo + carboplatin, ‡ gemcitabine§ or Placebo + cisplatin, || gemcitabine§ No crossover allowed from control to either atezolizumab arm • Key inclusion criteria: First-line platinum-eligible; evaluable for tumor PD-L 1 expression; no prior CT for inoperable, locally advanced, or metastatic UC; ECOG ≤ 2 • Primary endpoints: PFS (investigator-assessed), OS, safety • Secondary endpoints: ORR, DOR, QOL, PK, ATA *China extension cohort to accrue until total of 240 patients are enrolled in China (including global enrollment phase and China extension cohort). † 1200 mg q 3 w (d 1). ‡AUC=4. 5 q 3 w (d 1). § 1000 mg/m 2 (d 1 and d 8). ||70 mg/m 2 q 3 w (d 1). ¶Per RECIST v 1. 1. #In specific circumstances atezolizumab treatment may continue beyond disease progression. RECIST=Response Evaluation Criteria in Solid Tumors. WO 30070 Protocol. Version 3. September 2016. https: //clinicaltrials. gov/ct 2/show/NCT 02807636. Accessed 10/07/16.

KEYNOTE-361: Phase III Pembrolizumab With or Without Platinum-Based Combination Chemotherapy Versus Chemotherapy in Patients With Advanced or Metastatic UC Advanced/unresectable or metastatic UC (Estimated N=990) Pembrolizumab† + carboplatin, ‡ gemcitabine§ or Pembrolizumab† + cisplatin, || gemcitabine§ R 1: 1: 1 Pembrolizumab† Disease progression¶/ unacceptable toxicity# Placebo + carboplatin, ‡ gemcitabine§ or Placebo + cisplatin, || gemcitabine§ • Key inclusion criteria: No prior systemic chemotherapy for advanced or metastatic UC (exception of neoadjuvant and adjuvant Pt-based CT); ECOG ≤ 2 • Primary endpoints: PFS (investigator-assessed), OS • Secondary endpoints: Safety, ORR, DCR, PFS as assessed by BICR • Estimated primary completion date: March 2019 † 200 mg q 3 w (d 1), max 35 doses. ‡AUC=5 or 4. 5 q 3 w (d 1). § 1000 mg/m 2 (d 1 and d 8). ||70 mg/m 2 q 3 w (d 1). ¶Per RECIST v 1. 1. #In specific circumstances pembrolizumab treatment may continue beyond disease progression. RECIST=Response Evaluation Criteria in Solid Tumors. WO 30070 Protocol. Version 3. September 2016. https: //clinicaltrials. gov/ct 2/show/NCT 02807636. Accessed 10/07/16. https: //clinicaltrials. gov/ct 2/show/NCT 02853305. Accessed 11/8/16.
![Update [6/20/2018]: The FDA is restricting the use of pembrolizumab and atezolizumab for patients Update [6/20/2018]: The FDA is restricting the use of pembrolizumab and atezolizumab for patients](http://slidetodoc.com/presentation_image_h2/688c6c28c9e54a04277f2fc8cd5ddd33/image-68.jpg)
Update [6/20/2018]: The FDA is restricting the use of pembrolizumab and atezolizumab for patients with locally advanced or metastatic urothelial cancer who are not eligible for cisplatin-containing therapy. • Data show lower survival in some patients with low levels of cancer protein PD-L 1 compared to chemotherapy alone • Early data from two clinical trials show reduced survival with pembrolizumab and atezolizumab when used as first-line treatments for urothelial cancer (cancer of the bladder and urinary tract) in cancers with low levels of a protein called PD-L 1. The data indicate that pembrolizumab and atezolizumab may not work as well as chemotherapy medicines in this group of patients. • As a result, the FDA labels of both drugs have been revised to reflect the restricted indications. The restricted indications for patients with urothelial cancer not eligible for cisplatin-containing therapy read as follows: In patients who: • Are not eligible for cisplatin-containing therapy, and whose tumors express PD-L 1, or • Are not eligible for any platinum-containing therapy regardless of level of tumor PDL 1 expression

IMvigor 130: a phase III study of atezolizumab with or without platinum-based chemotherapy in previously untreated metastatic urothelial carcinoma Enrique Grande, 1 Matthew D Galsky, 2 José Ángel Arranz Arija, 3 Maria De Santis, 4 Ian D Davis, 5 Ugo De Giorgi, 6 Marina Mencinger, 7 Eiji Kikuchi, 8 Xavier García-del-Muro, 9 Mahmut Gumus, 10 Mustafa Özgüroğlu, 11 Arash Rezazadeh Kalebasty, 12 Se Hoon Park, 13 Boris Alekseev, 14 Fabio Augusto Schutz, 15 Jian-Ri Li, 16 Almut Mecke, 17 Sanjeev Mariathasan, 18 Ann. Christine Thåström, 18 Aristotelis Bamias 19 1 MD Anderson Cancer Center Madrid, Spain; 2 Icahn School of Medicine at Mount Sinai/Tisch Cancer Institute, New York, NY, USA; 3 Hospital General Universitario Gregorio Marañón, Madrid, Spain; 4 Charité University Hospital, Berlin, Germany, and Department of Urology, Medical University, Vienna, Austria; 5 Eastern Health/Monash University, Melbourne, Australia; 6 Istituto Scientifico Romagnolo per lo Studio e la Cura dei Tumori (IRST), IRCCS, Meldola, Italy; 7 Institute of Oncology Ljubljana, Slovenia; 8 Keio University, Tokyo, Japan; 9 Catalan Institute of Oncology, IDIBELL, University of Barcelona, Spain; 10 Istanbul Medeniyet University, Goztepe Research Hospital, Istanbul, Turkey; 11 Istanbul University-Cerrahpaşa, Cerrahpasa School of Medicine, Istanbul, Turkey; 12 Norton Cancer Institute, Louisville, KY, USA; 13 Sungkyunkwan University Samsung Medical Center, Seoul, Korea; 14 P. Herzen Oncology Research Institute, Moscow, Russia; 15 Beneficência Portuguesa de São Paulo , São Paulo, Brazil; 16 Taichung Veterans General Hospital/Hungkuang University, Taichung, Taiwan; 17 F. Hoffmann-La Roche Ltd, Basel, Switzerland; 18 Genentech, Inc. , South San Francisco, CA, USA; 19 National and Kapodistrian University of Athens, Greece esmo. org
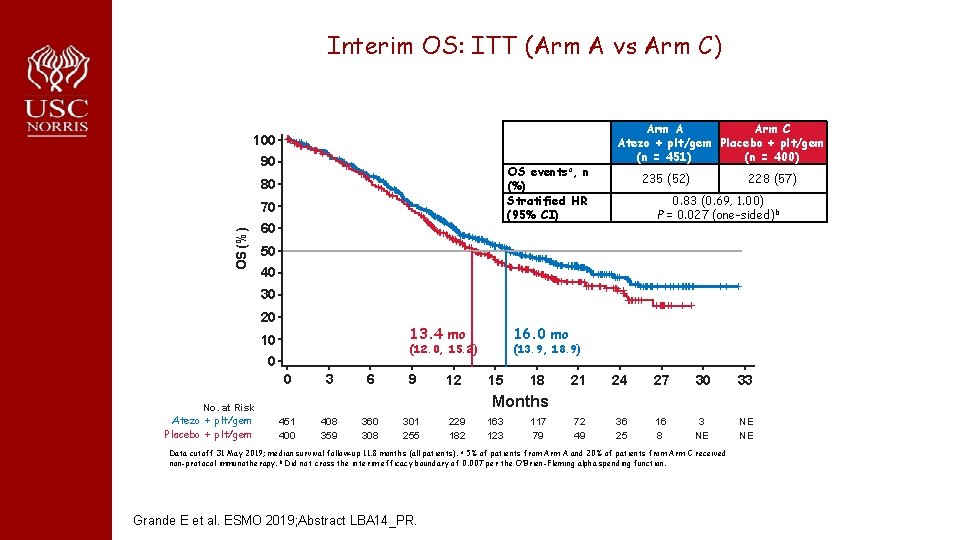
Interim OS: ITT (Arm A vs Arm C) 100 90 OS eventsa, n (%) Stratified HR (95% CI) 80 OS (%) 70 60 Arm A Arm C Atezo + plt/gem Placebo + plt/gem (n = 451) (n = 400) 235 (52) 228 (57) 0. 83 (0. 69, 1. 00) P = 0. 027 (one-sided) b 50 40 30 20 13. 4 mo 10 (12. 0, 15. 2) 0 0 No. at Risk Atezo + plt/gem Placebo + plt/gem 16. 0 mo 3 6 9 12 (13. 9, 18. 9) 15 18 21 24 27 30 33 72 49 36 25 16 8 3 NE NE NE Months 451 400 408 359 360 308 301 255 229 182 163 123 117 79 Data cutoff 31 May 2019; median survival follow-up 11. 8 months (all patients). a 5% of patients from Arm A and 20% of patients from Arm C received non-protocol immunotherapy. b Did not cross the interim efficacy boundary of 0. 007 per the O’Brien-Fleming alpha spending function. Grande E et al. ESMO 2019; Abstract LBA 14_PR.

Interim OS: PD-L 1 status (Arm B vs Arm C) OS (%) PD-L 1 IC 0/1 Months Atezo Placebo + OS events, n (%) Unstratified HR (95% CI) Arm B Atezo (n = 272) 158 (58) Arm C Placebo + plt/gem (n = 274) 156 (57) 1. 07 (0. 86, 1. 33)

PD-1 / PD-L 1 therapy in Metastatic Urothelial Cancer: Summary First line: • In cisplatin-ineligible patients we need to test tumor tissue for PD-L 1 – If the tumor has high expression then CPI is appropriate – If low then best chemotherapy available or symptomatic care • In cisplatin-eligible patients: give cisplatin In platinum pre-treated patients: • Single agent CPI is standard of care. Clinical trials remain good options.

Special Issues in Oncology Care Personalized oncology: Delivering bad news to patients
- Slides: 73