Module 7 Outbreak Investigation Disease Control Public Health

Module 7: Outbreak Investigation & Disease Control Public Health Surveillance Developed through the APTR Initiative to Enhance Prevention and Population Health Education in collaboration with the Brody School of Medicine at East Carolina University with funding from the Centers for Disease Control and Prevention

Acknowledgments APTR wishes to acknowledge the following individuals that developed this module: Kristina Simeonsson, MD, MSPH Department of Public Health Brody School of Medicine at East Carolina University Julie Daugherty, MPH Department of Public Health Brody School of Medicine at East Carolina University This education module is made possible through the Centers for Disease Control and Prevention (CDC) and the Association for Prevention Teaching and Research (APTR) Cooperative Agreement, No. 5 U 50 CD 300860. The module represents the opinions of the author(s) and does not necessarily represent the views of the Centers for Disease Control and Prevention or the Association for Prevention Teaching and Research.

Presentation Objectives 1. 2. 3. 4. Define surveillance Discuss the uses of surveillance Review notifiable disease surveillance Describe surveillance limitations
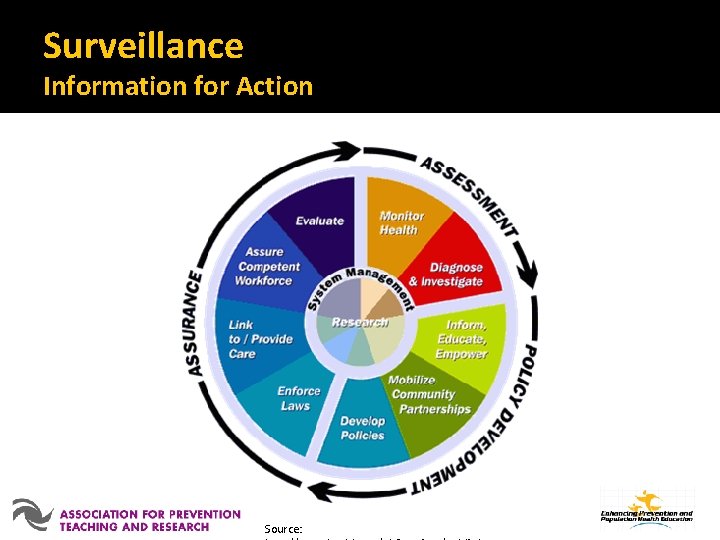
Surveillance Information for Action Source:

Surveillance The ongoing systematic collection, analysis, and interpretation of health data, essential to the planning, implementation, and evaluation of public health practice, closely integrated with the timely dissemination to those who need to know. www. cdc. gov

Sources of Surveillance Data Notifiable diseases Vital records Environmental monitoring systems Animal health data Individuals Laboratories Medical records Over the counter medication sales Registries Surveys population-based provider-based

Types of Surveillance Passive Active Syndromic

Passive Surveillance Laboratories, physicians, or others regularly report cases of disease / death to the local or state health department Case reports based on a standard case definition of that particular disease Deaths reported on standard certificate

Active Surveillance Local or state health departments initiate the collection of information from laboratories, physicians, health care providers or the general population. Achieves more complete and accurate reporting More resource intensive for the public health agency ▪ money ▪ personnel ▪ time

Syndromic Surveillance The ongoing, systematic collection, analysis, interpretation, and application of real-time indicators for disease that allow for detection before public health authorities would otherwise identify them. Common surveillance syndromes Gastrointestinal Influenza-like illness Rash and Fever

Ideal Surveillance System Simple Timely Representative Flexible Sensitive Strong predictive value Acceptable public health care providers Cost-effective

Ideal Surveillance System Simple Timely Representative Flexible Sensitive Strong predictive value Acceptable public health care providers Cost effective

Ideal Surveillance System Sensitivity and Predictive Value Sensitivity Few if any missed cases Increase by having broad case definitions Positive predictive value Almost all case reports received for illnesses meet the surveillance case definition Increase by adopting a more restrictive case definition Hopkins, R. 2005

Uses of Public Health Surveillance Estimate magnitude of the problem Determine geographic distribution of illness Portray the natural history of a disease Detect epidemics / define a problem Generate hypotheses, stimulate research Evaluate control measures Monitor changes in infectious agents Detect changes in health practices Facilitate planning http: //www. cdc. gov/osels/ph_surveillance/nndss/phs/overview. htm

Uses of Public Health Surveillance Estimate magnitude of the problem Determine geographic distribution of illness Portray the natural history of a disease Detect epidemics / define a problem Generate hypotheses, stimulate research Evaluate control measures Monitor changes in infectious agents Detect changes in health practices Facilitate planning
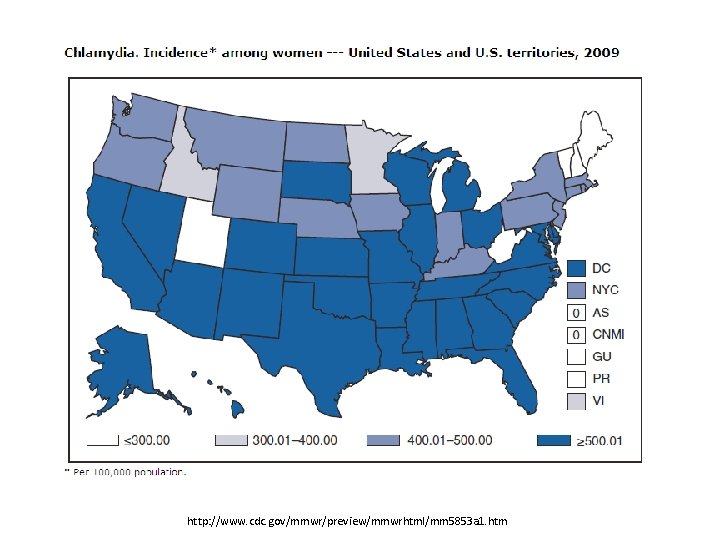
http: //www. cdc. gov/mmwr/preview/mmwrhtml/mm 5853 a 1. htm

Uses of Public Health Surveillance Estimate magnitude of the problem Determine geographic distribution of illness Portray the natural history of a disease Detect epidemics / define a problem Generate hypotheses, stimulate research Evaluate control measures Monitor changes in infectious agents Detect changes in health practices Facilitate planning
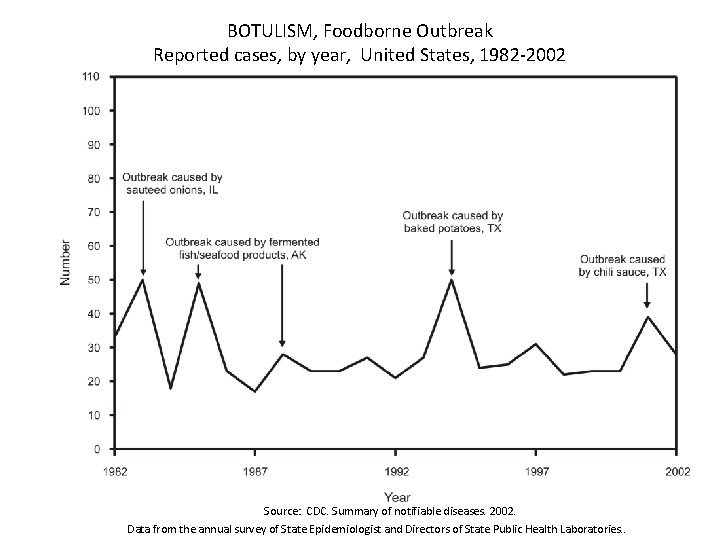
BOTULISM, Foodborne Outbreak Reported cases, by year, United States, 1982 -2002 Source: CDC. Summary of notifiable diseases. 2002. Data from the annual survey of State Epidemiologist and Directors of State Public Health Laboratories. .

Uses of Public Health Surveillance Estimate magnitude of the problem Determine geographic distribution of illness Portray the natural history of a disease Detect epidemics / define a problem Generate hypotheses, stimulate research Evaluate control measures Monitor changes in infectious agents Detect changes in health practices Facilitate planning
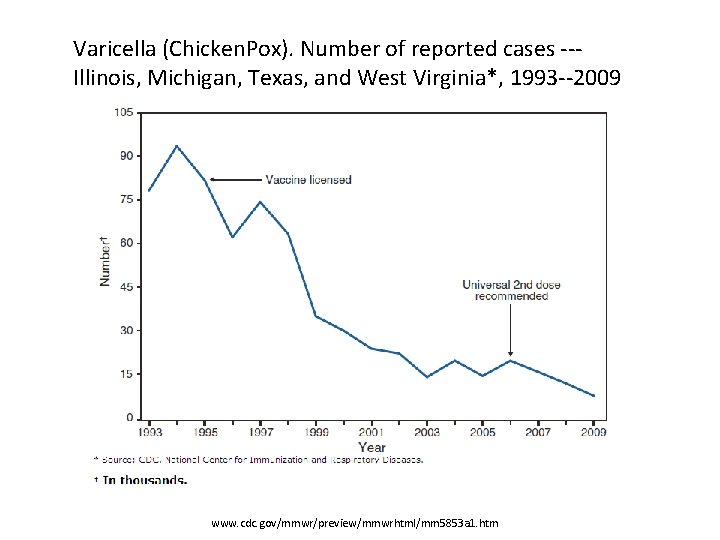
Varicella (Chicken. Pox). Number of reported cases --Illinois, Michigan, Texas, and West Virginia*, 1993 --2009 www. cdc. gov/mmwr/preview/mmwrhtml/mm 5853 a 1. htm

Notifiable Disease Surveillance Foundation is state and local application of the reportable disease surveillance system known as National Notifiable Disease Surveillance System (NNDSS) Infectious reportable diseases Noninfectious reportable diseases ▪ Cancer ▪ Elevated blood lead levels ▪ Pesticide-related illness

National Notifiable Disease Surveillance System (NNDSS) System for passing reports from the local to state health departments, and then on to CDC Role of the Council of State and Territorial Epidemiologists (CSTE) List of disease and laboratory findings of public health interest Case definitions for their surveillance Dissemination of surveillance data Morbidity and Mortality Weekly Report (MMWR) MMWR Annual Summary of Notifiable Diseases
Case Definitions Uniform criteria for reporting cases Clinical, Laboratory, Epidemiologic http: //www. cdc. gov/osels/ph_surveillance/nndss/casedef/hepatiti sacurrent. htm
MMWR Annual Summary of Notifiable Diseases http: //www. cdc. gov/mmwr/PDF/wk/mm 5754. pdf

State Participation in NNDSS Reporting by states to the CDC is voluntary Reporting mandated at state level state legislation or regulation Variation in the lists of reportable diseases exists between states

Example of Physician Report Hepatitis B Resident of long-term care facility hospitalized with acute hepatitis B Physician reports case to local health department (LHD) ▪ LHD nurse remembers several other cases of acute hepatitis B at same facility in past year ▪ Unsuspected outbreak of acute hepatitis B was discovered in the facility

Example of Physician Report Hepatitis B MMWR, March 11, 2005 / 54(09); 220 -223

Example of Physician Report Listeriosis Stillborn infant delivered at hospital diagnosed with listeriosis Physician reports case to LHD ▪ Outbreak of listeriosis was identified among Hispanic females with 12 cases identified over 4 month period � 5 stillbirths � 3 premature births � 2 infected newborns ▪ Case-control study identified common source of exposure

Example of Physician Report Listeriosis MMWR, July 06, 2001 / 50(26); 560 -2
Example of Physician Report Listeriosis Used with permission from Robert E. Whitwam

Limitations of Surveillance Incomplete data Overwhelming volumes of data from a variety of sources make management complex Uneven application of information technology Paper versus electronic reporting Timeliness Reporting time requirement Reporting burden Completeness Unreported cases Incomplete reports

CDC’s Burden of Illness Pyramid: Underascertainment of Foodborne Illness in Notifiable Diseases Surveillance Source: Adapted from Centers for Disease Control and Prevention, “Food. Net Surveillance - Burden of Illness Pyramid. ” http: //www. cdc. gov/foodnet/surveillance_pages/burden_pyramid. htm. Accessed October 3, 2011.

Limitations of Surveillance Shigella 100 persons infected with shigella 76 symptomatic 28 consulted a healthcare provider 9 submitted stool cultures 7 had positive results 6 reported to the local health department 5 reported nationally to CDC Rosenberg et al, 1977

Reasons for Failure to Report 1. 2. 3. 4. 5. 6. 7. Lack of awareness of legal requirement Lack if knowledge of which conditions are reportable Lack of knowledge of how or to whom to report Assumption that someone else will report the case Intentional failure to report to protect patient privacy Insufficient reward for reporting Insufficient penalty for not reporting Doyle et al, 2002

Limitations of Syndromic Surveillance Costly Staff expertise required Formal evaluations of syndromic surveillance systems are incomplete

Limitations of Syndromic Surveillance Inadequate specificity = false alarms Software recognizes key words Uses resources in investigation Inadequate sensitivity = failure to detect outbreaks or bioterrorism events Outbreak is too small Population disperses after exposure, cluster not evident

Summary Surveillance data has many practical uses Notifiable disease surveillance is fundamental to prevention and control efforts List of notifiable diseases varies by state Significant improvements are being made in advancing surveillance and reporting

Collaborating Institutions Center for Public Health Continuing Education University at Albany School of Public Health Department of Community & Family Medicine Duke University School of Medicine

Advisory Committee Mike Barry, CAE Lorrie Basnight, MD Nancy Bennett, MD, MS Ruth Gaare Bernheim, JD, MPH Amber Berrian, MPH James Cawley, MPH, PA-C Jack Dillenberg, DDS, MPH Kristine Gebbie, RN, Dr. PH Asim Jani, MD, MPH, FACP Denise Koo, MD, MPH Suzanne Lazorick, MD, MPH Rika Maeshiro, MD, MPH Dan Mareck, MD Steve Mc. Curdy, MD, MPH Susan M. Meyer, Ph. D Sallie Rixey, MD, MEd Nawraz Shawir, MBBS

APTR Sharon Hull, MD, MPH President Allison L. Lewis Executive Director O. Kent Nordvig, MEd Project Representative
- Slides: 40