Module 5 Nebido clinical efficacy Approval Number G

Module 5 Nebido® clinical efficacy Approval Number: G. MKT. GM. MH. 04. 2018. 0513

Nebido®: clinical efficacy • The clinical efficacy of Nebido® has been evaluated in a number of studies in patients with hypogonadism, including those with: – Type 2 diabetes mellitus – Metabolic syndrome – Obesity – Hypogonadism of different etiologies – ED ED, erectile dysfunction 2

Module 5: Nebido® clinical efficacy Body composition 3
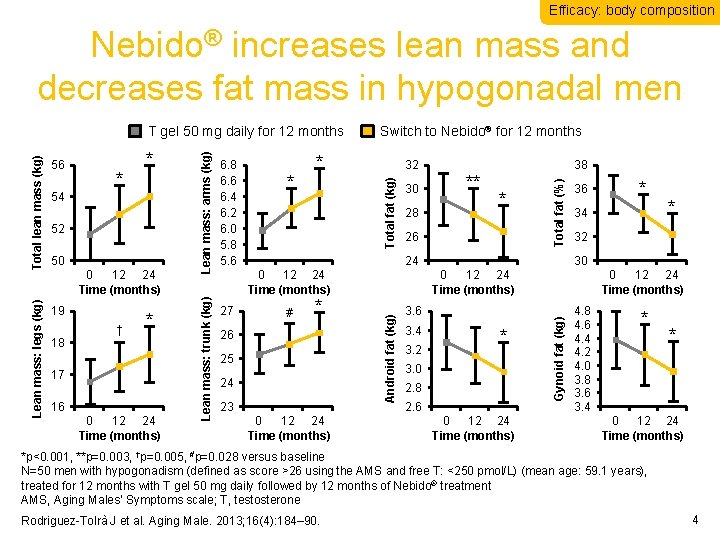
Efficacy: body composition Nebido® increases lean mass and decreases fat mass in hypogonadal men 0 12 24 Time (months) † * 18 17 16 0 12 24 Time (months) 30 28 * 26 24 0 12 24 Time (months) 27 ** 38 Total fat (%) 32 Total fat (kg) * * # * 26 25 24 23 0 12 24 Time (months) 36 3. 2 * 32 30 3. 6 3. 4 * 34 0 12 24 Time (months) * 3. 0 2. 8 2. 6 0 12 24 Time (months) Gynoid fat (kg) 50 6. 8 6. 6 6. 4 6. 2 6. 0 5. 8 5. 6 Switch to Nebido® for 12 months Android fat (kg) 52 Lean mass: arms (kg) 19 54 * * Lean mass: trunk (kg) Total lean mass (kg) 56 Lean mass: legs (kg) T gel 50 mg daily for 12 months 4. 8 4. 6 4. 4 4. 2 4. 0 3. 8 3. 6 3. 4 * * 0 12 24 Time (months) *p<0. 001, **p=0. 003, †p=0. 005, #p=0. 028 versus baseline N=50 men with hypogonadism (defined as score >26 using the AMS and free T: <250 pmol/L) (mean age: 59. 1 years), treated for 12 months with T gel 50 mg daily followed by 12 months of Nebido® treatment AMS, Aging Males’ Symptoms scale; T, testosterone Rodriguez-Tolrà J et al. Aging Male. 2013; 16(4): 184– 90. 4
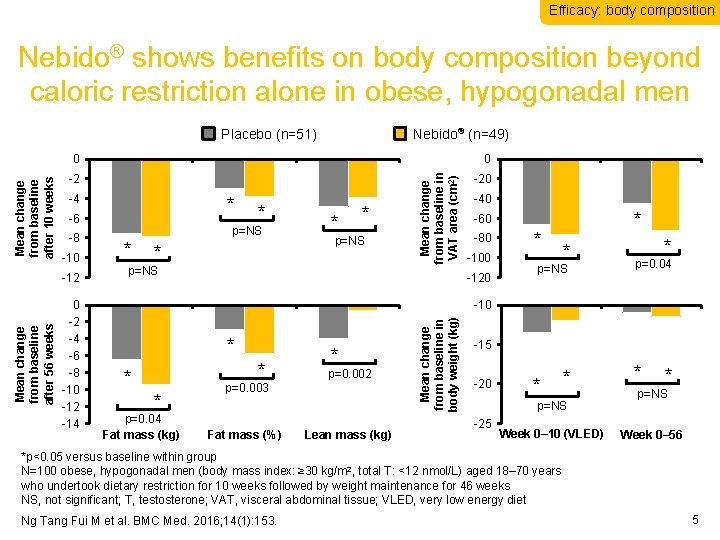
Efficacy: body composition Nebido® shows benefits on body composition beyond caloric restriction alone in obese, hypogonadal men Placebo (n=51) Nebido® (n=49) -4 * -6 -8 -10 0 -2 -4 -6 -8 -10 -12 -14 * p=NS * * p=NS Fat mass (kg) Fat mass (%) * * p=NS Mean change from baseline in VAT area (cm 2) -2 -12 Mean change from baseline after 56 weeks 0 -20 -40 Lean mass (kg) * -60 -80 -100 -120 * * * p=NS p=0. 04 Week 0– 10 Week 0– 56 -10 * * p=0. 04 Fat mass (kg) * p=0. 002 p=0. 003 Fat mass (%) Lean mass (kg) Mean change from baseline in body weight (kg) Mean change from baseline after 10 weeks 0 -15 -20 * * p=NS -25 Week 0– 10 (VLED) Week * * p=NS Week 0– 56 *p<0. 05 versus baseline within group N=100 obese, hypogonadal men (body mass index: ≥ 30 kg/m 2, total T: <12 nmol/L) aged 18– 70 years who undertook dietary restriction for 10 weeks followed by weight maintenance for 46 weeks NS, not significant; T, testosterone; VAT, visceral abdominal tissue; VLED, very low energy diet Ng Tang Fui M et al. BMC Med. 2016; 14(1): 153. 5
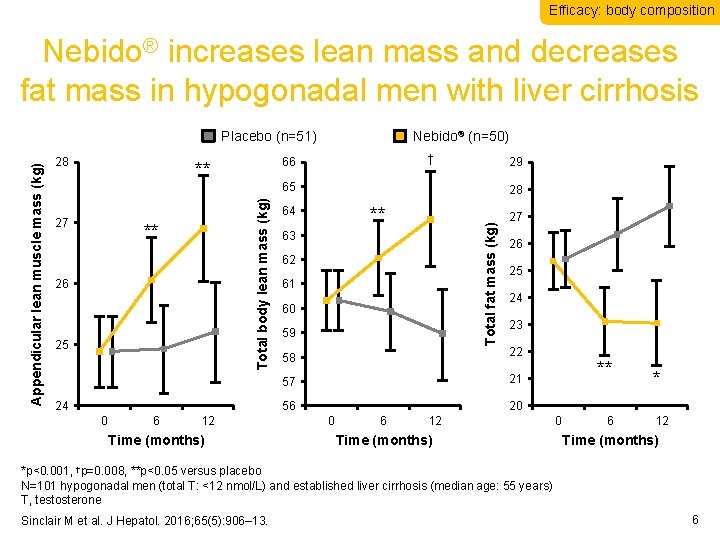
Efficacy: body composition Nebido® increases lean mass and decreases fat mass in hypogonadal men with liver cirrhosis ** 26 25 6 29 65 24 0 † 66 ** 27 Nebido® (n=50) 12 Time (months) 28 ** 64 Total fat mass (kg) 28 Total body lean mass (kg) Appendicular lean muscle mass (kg) Placebo (n=51) 63 62 61 60 59 27 26 25 24 23 58 22 57 21 56 20 0 6 12 Time (months) ** 0 6 * 12 Time (months) *p<0. 001, †p=0. 008, **p<0. 05 versus placebo N=101 hypogonadal men (total T: <12 nmol/L) and established liver cirrhosis (median age: 55 years) T, testosterone Sinclair M et al. J Hepatol. 2016; 65(5): 906– 13. 6

Module 5: Nebido® clinical efficacy Weight loss 7
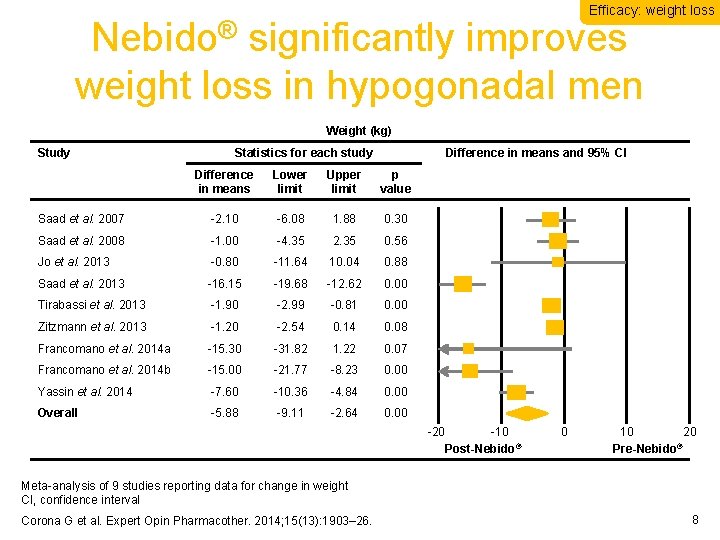
Efficacy: weight loss Nebido® significantly improves weight loss in hypogonadal men Weight (kg) Study Statistics for each study Difference in means and 95% CI Difference in means Lower limit Upper limit p value Saad et al. 2007 -2. 10 -6. 08 1. 88 0. 30 Saad et al. 2008 -1. 00 -4. 35 2. 35 0. 56 Jo et al. 2013 -0. 80 -11. 64 10. 04 0. 88 Saad et al. 2013 -16. 15 -19. 68 -12. 62 0. 00 Tirabassi et al. 2013 -1. 90 -2. 99 -0. 81 0. 00 Zitzmann et al. 2013 -1. 20 -2. 54 0. 14 0. 08 Francomano et al. 2014 a -15. 30 -31. 82 1. 22 0. 07 Francomano et al. 2014 b -15. 00 -21. 77 -8. 23 0. 00 Yassin et al. 2014 -7. 60 -10. 36 -4. 84 0. 00 Overall -5. 88 -9. 11 -2. 64 0. 00 -20 -10 Post-Nebido® 0 10 20 ® Pre-Nebido Meta-analysis of 9 studies reporting data for change in weight CI, confidence interval Corona G et al. Expert Opin Pharmacother. 2014; 15(13): 1903– 26. 8
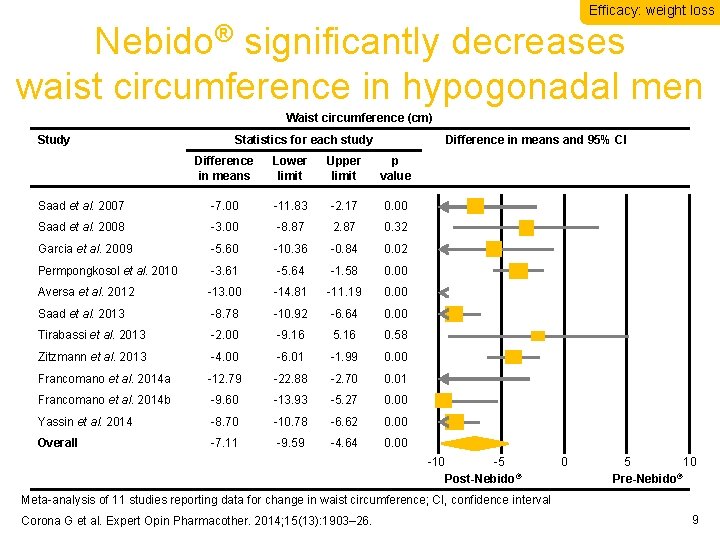
Efficacy: weight loss Nebido® significantly decreases waist circumference in hypogonadal men Waist circumference (cm) Study Statistics for each study Difference in means and 95% CI Difference in means Lower limit Upper limit p value Saad et al. 2007 -7. 00 -11. 83 -2. 17 0. 00 Saad et al. 2008 -3. 00 -8. 87 2. 87 0. 32 Garcia et al. 2009 -5. 60 -10. 36 -0. 84 0. 02 Permpongkosol et al. 2010 -3. 61 -5. 64 -1. 58 0. 00 Aversa et al. 2012 -13. 00 -14. 81 -11. 19 0. 00 Saad et al. 2013 -8. 78 -10. 92 -6. 64 0. 00 Tirabassi et al. 2013 -2. 00 -9. 16 5. 16 0. 58 Zitzmann et al. 2013 -4. 00 -6. 01 -1. 99 0. 00 Francomano et al. 2014 a -12. 79 -22. 88 -2. 70 0. 01 Francomano et al. 2014 b -9. 60 -13. 93 -5. 27 0. 00 Yassin et al. 2014 -8. 70 -10. 78 -6. 62 0. 00 Overall -7. 11 -9. 59 -4. 64 0. 00 -10 -5 Post-Nebido® 0 5 10 ® Pre-Nebido Meta-analysis of 11 studies reporting data for change in waist circumference; CI, confidence interval Corona G et al. Expert Opin Pharmacother. 2014; 15(13): 1903– 26. 9

Module 5: Nebido® clinical efficacy Physical strength 10
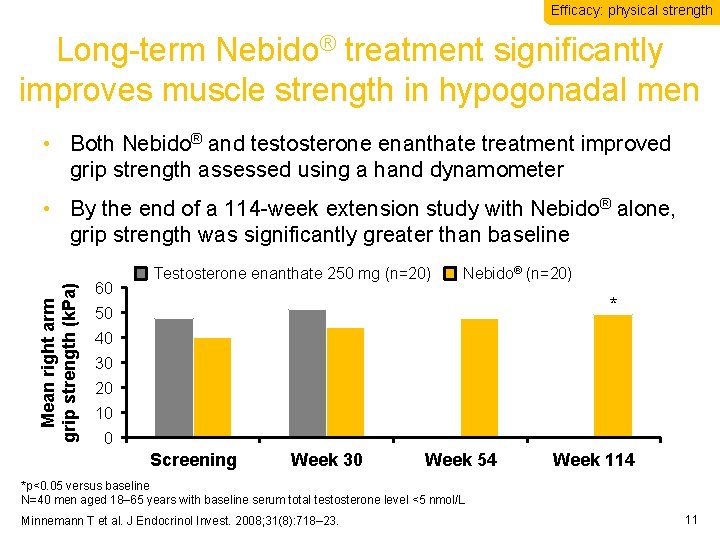
Efficacy: physical strength Long-term Nebido® treatment significantly improves muscle strength in hypogonadal men • Both Nebido® and testosterone enanthate treatment improved grip strength assessed using a hand dynamometer Mean right arm grip strength (k. Pa) • By the end of a 114 -week extension study with Nebido® alone, grip strength was significantly greater than baseline 60 Testosterone enanthate 250 mg (n=20) Nebido® (n=20) * 50 40 30 20 10 0 Screening Week 30 Week 54 Week 114 *p<0. 05 versus baseline N=40 men aged 18– 65 years with baseline serum total testosterone level <5 nmol/L Minnemann T et al. J Endocrinol Invest. 2008; 31(8): 718– 23. 11

Module 5: Nebido® clinical efficacy Anemia 12
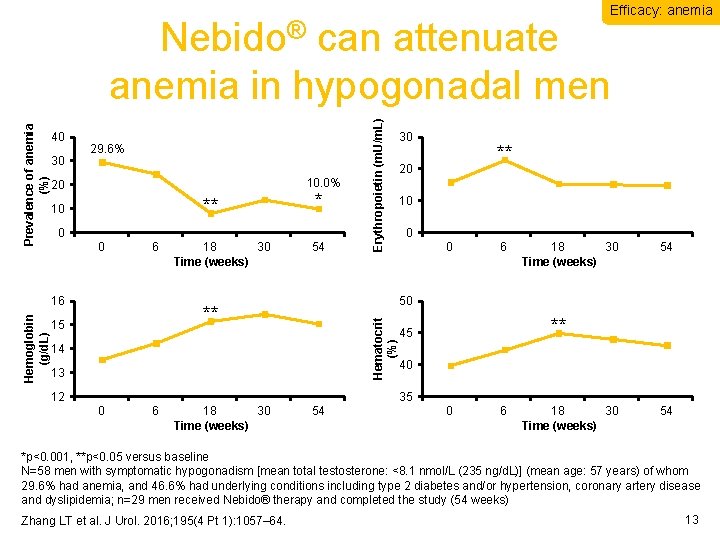
Efficacy: anemia 30 29. 6% 10. 0% 20 ** 10 0 0 6 16 Hemoglobin (g/d. L) * 18 30 Time (weeks) 54 30 ** 20 10 0 0 6 18 30 Time (weeks) 54 50 ** 15 Erythropoietin (m. U/m. L) 40 Hematocrit (%) Prevalence of anemia (%) Nebido® can attenuate anemia in hypogonadal men 14 13 12 ** 45 40 35 0 6 18 30 Time (weeks) 54 *p<0. 001, **p<0. 05 versus baseline N=58 men with symptomatic hypogonadism [mean total testosterone: <8. 1 nmol/L (235 ng/d. L)] (mean age: 57 years) of whom 29. 6% had anemia, and 46. 6% had underlying conditions including type 2 diabetes and/or hypertension, coronary artery disease and dyslipidemia; n=29 men received Nebido® therapy and completed the study (54 weeks) Zhang LT et al. J Urol. 2016; 195(4 Pt 1): 1057– 64. 13

Module 5: Nebido® clinical efficacy Bone mineral density 14
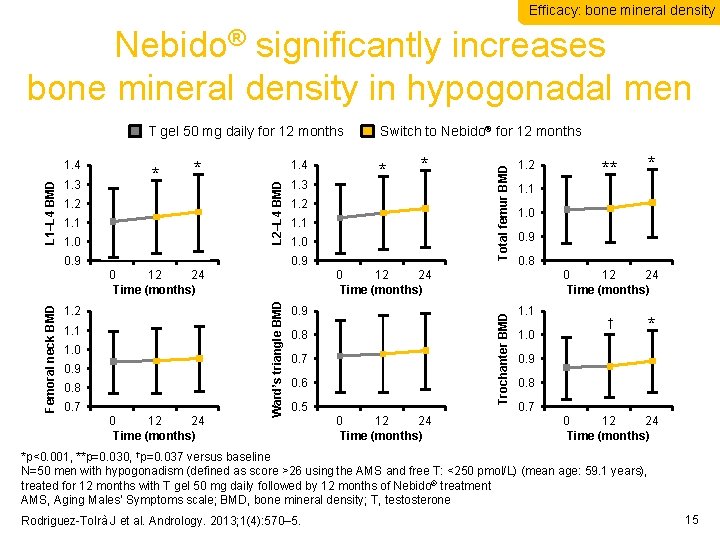
Efficacy: bone mineral density Nebido® significantly increases bone mineral density in hypogonadal men * 1. 2 1. 1 1. 0 1. 4 0. 9 1. 3 * 1. 2 1. 1 1. 0 0. 9 0 12 24 Time (months) 1. 2 1. 1 1. 0 0. 9 0. 8 0. 7 0 12 24 Time (months) 1. 2 0. 9 0. 8 0. 7 0. 6 0. 5 0 12 24 Time (months) ** * 1. 1 1. 0 0. 9 0. 8 0 12 24 Time (months) Ward’s triangle BMD Femoral neck BMD * Trochanter BMD 1. 3 * L 2–L 4 BMD L 1–L 4 BMD 1. 4 Switch to Nebido® for 12 months Total femur BMD T gel 50 mg daily for 12 months 1. 1 1. 0 † * 0. 9 0. 8 0. 7 0 12 24 Time (months) *p<0. 001, **p=0. 030, †p=0. 037 versus baseline N=50 men with hypogonadism (defined as score >26 using the AMS and free T: <250 pmol/L) (mean age: 59. 1 years), treated for 12 months with T gel 50 mg daily followed by 12 months of Nebido® treatment AMS, Aging Males’ Symptoms scale; BMD, bone mineral density; T, testosterone Rodriguez-Tolrà J et al. Andrology. 2013; 1(4): 570– 5. 15
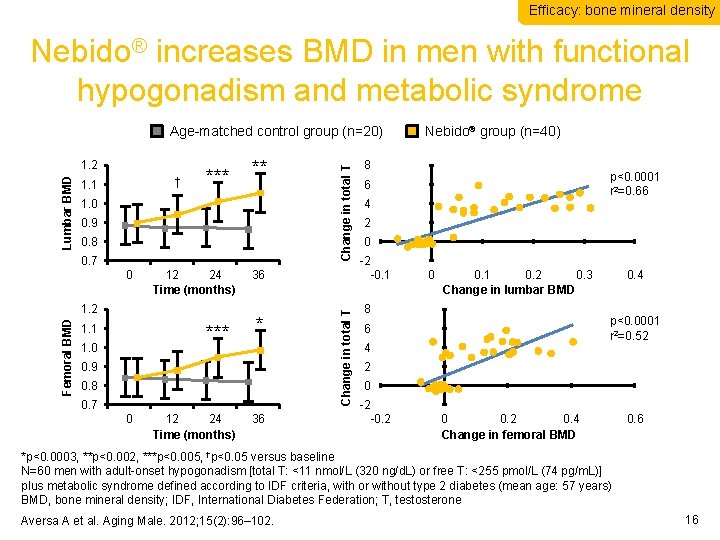
Efficacy: bone mineral density Nebido® increases BMD in men with functional hypogonadism and metabolic syndrome † 1. 1 *** ** 1. 0 0. 9 0. 8 0. 7 0 12 24 Time (months) Femoral BMD 1. 2 1. 1 *** 1. 0 36 * 0. 9 0. 8 0. 7 0 12 24 Time (months) 36 Change in total T Lumbar BMD 1. 2 Change in total T Age-matched control group (n=20) Nebido® group (n=40) 8 p<0. 0001 r 2=0. 66 6 4 2 0 -2 -0. 1 0. 2 0. 3 Change in lumbar BMD 8 0. 4 p<0. 0001 r 2=0. 52 6 4 2 0 -2 -0. 2 0. 4 Change in femoral BMD 0. 6 *p<0. 0003, **p<0. 002, ***p<0. 005, †p<0. 05 versus baseline N=60 men with adult-onset hypogonadism [total T: <11 nmol/L (320 ng/d. L) or free T: <255 pmol/L (74 pg/m. L)] plus metabolic syndrome defined according to IDF criteria, with or without type 2 diabetes (mean age: 57 years) BMD, bone mineral density; IDF, International Diabetes Federation; T, testosterone Aversa A et al. Aging Male. 2012; 15(2): 96– 102. 16
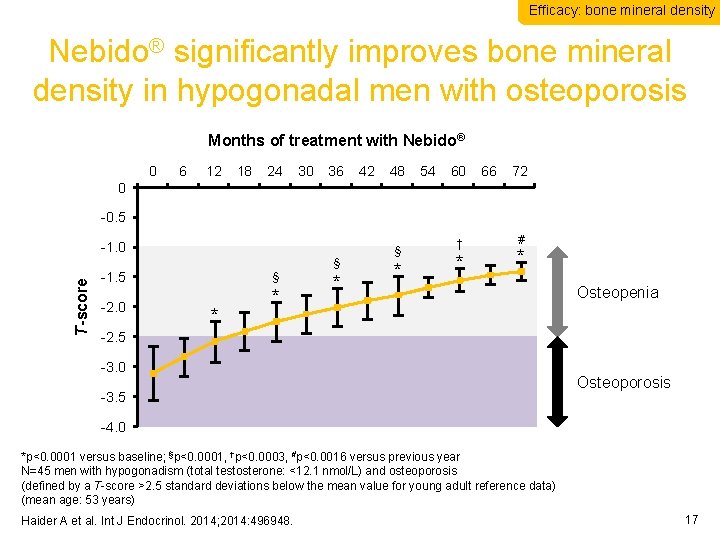
Efficacy: bone mineral density Nebido® significantly improves bone mineral density in hypogonadal men with osteoporosis Months of treatment with Nebido® 0 6 12 18 24 30 36 42 48 54 60 66 72 0. 0 -0. 5 T-score -1. 0 § -1. 5 -2. 0 § * * * § * † * # * Osteopenia -2. 5 -3. 0 -3. 5 Osteoporosis -4. 0 *p<0. 0001 versus baseline; §p<0. 0001, †p<0. 0003, #p<0. 0016 versus previous year N=45 men with hypogonadism (total testosterone: <12. 1 nmol/L) and osteoporosis (defined by a T-score >2. 5 standard deviations below the mean value for young adult reference data) (mean age: 53 years) Haider A et al. Int J Endocrinol. 2014; 2014: 496948. 17

Module 5: Nebido® clinical efficacy Men with metabolic syndrome and/or type 2 diabetes 18

Efficacy: metabolic syndrome & diabetes Nebido® improves clinical parameters in hypogonadal men with diabetes or metabolic syndrome • There is a high prevalence of hypogonadism in men with type 2 diabetes and metabolic syndrome 1, 2 • Studies show that TTh with Nebido® is effective at increasing serum testosterone levels and improving other endpoints including BMD, anthropometry, body composition, metabolic parameters, and sexual function in this patient population 3– 7 • Furthermore, TTh with Nebido® may reduce the occurrence of, or improve, diabetic complications in men with hypogonadism and type 2 diabetes 3, 8 BMD, bone mineral density; TTh, testosterone therapy 1. Traish AM et al. J Androl. 2009; 30(1): 10– 22. 2. Traish AM et al. J Androl. 2009; 30(1): 23– 32. 3. Janjgava S et al. Eur J Med Res. 2014; 19(1): 56. 4. Gianatti EJ et al. Diabetes Care. 2014; 37(8): 2098– 107. 5. Gianatti EJ et al. J Clin Endocrinol Metab. 2014; 99(10): 3821– 8. 6. Francomano D et al. J Endocrinol Invest. 2014; 37(4): 401– 11. 7. Saad F et al. Arch Androl. 2007; 53(6): 353– 7. 8. Kalinchenko S et al. Cardiovasc Diabetol. 2009; 8: 19. 19

Efficacy: metabolic syndrome & diabetes Nebido® significantly improves clinical parameters in hypogonadal men with metabolic syndrome • Nebido® significantly decreased body mass index, body weight and waist circumference, as well as levels of leptin and the HOMA-IR, as a measure of insulin resistance, versus placebo Placebo group (n=71) Change from baseline at 30 weeks p<0. 001 2 0 -2 -4 -6 -8 -10 -12 -14 Nebido® group (n=113) p<0. 001 p=0. 04 +0. 20 -0. 11 -1. 32 -0. 40 -1. 46 -4. 31 -1. 49 -6. 02 -5. 80 -11. 60 Body mass index 2 (kg/m 2) (kg/m ) Weight (kg) Waist circumference (cm) Leptin (μg/L) HOMA-IR N=184 men aged 35– 70 years with metabolic syndrome and hypogonadism [total T: <12 nmol/L (350 ng/d. L) or free T: <225 pmol/L (65 pg/m. L)] HOMA-IR, Homeostasis Model Assessment index of Insulin Resistance; T, testosterone Kalinchenko SY et al. Clin Endocrinol (Oxf). 2010; 73(5): 602– 12. 20
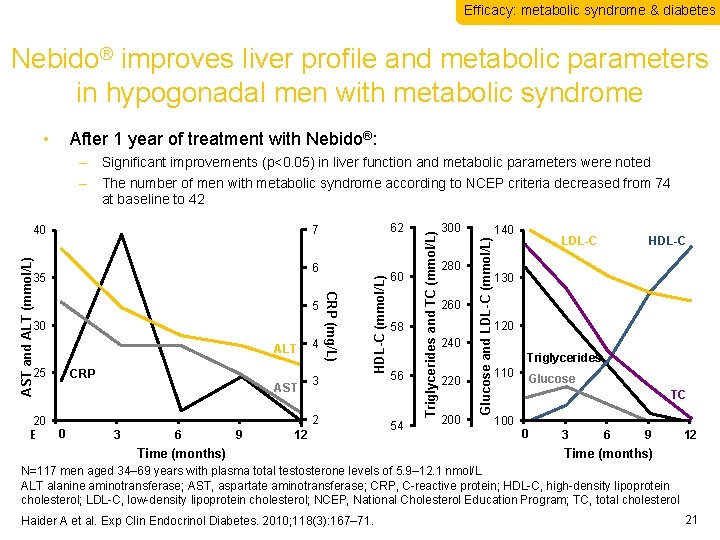
Efficacy: metabolic syndrome & diabetes Nebido® improves liver profile and metabolic parameters in hypogonadal men with metabolic syndrome • After 1 year of treatment with Nebido®: ALT CRP 20 0 Baseline AST 3 6 9 4 60 58 3 56 2 54 12 Time (months) 300 280 260 240 220 200 Glucose and LDL-C (mmol/L) 30 HDL-C (mmol/L) 5 CRP (mg/L) AST and ALT (mmol/L) 6 35 25 62 7 40 Triglycerides and TC (mmol/L) – Significant improvements (p<0. 05) in liver function and metabolic parameters were noted – The number of men with metabolic syndrome according to NCEP criteria decreased from 74 at baseline to 42 140 LDL-C HDL-C 130 120 Triglycerides 110 Glucose TC 100 0 BL Baseline 3 6 9 12 Time (months) N=117 men aged 34– 69 years with plasma total testosterone levels of 5. 9– 12. 1 nmol/L ALT alanine aminotransferase; AST, aspartate aminotransferase; CRP, C-reactive protein; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; NCEP, National Cholesterol Education Program; TC, total cholesterol Haider A et al. Exp Clin Endocrinol Diabetes. 2010; 118(3): 167– 71. 21
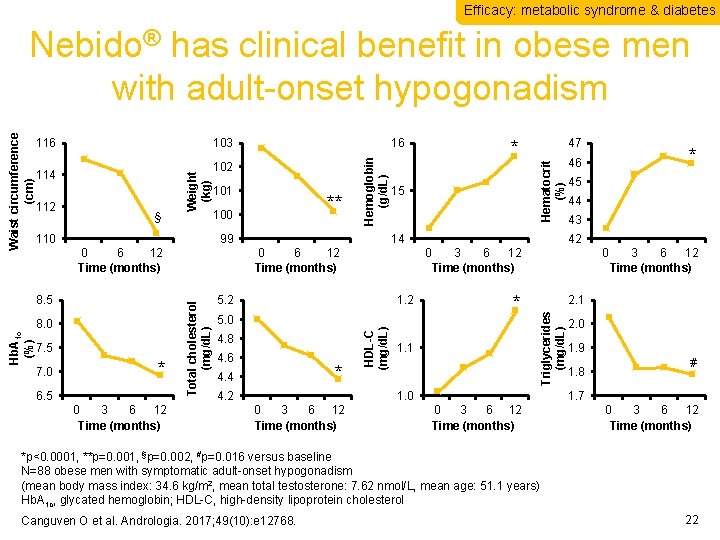
Efficacy: metabolic syndrome & diabetes 110 99 0 6 12 Time (months) 8. 5 Hb. A 1 c (%) 8. 0 7. 5 7. 0 ** 100 * 6. 5 0 3 6 12 Time (months) 47 Hematocrit (%) § 101 15 14 0 6 12 Time (months) 5. 2 1. 2 5. 0 4. 8 4. 6 * 4. 4 4. 2 45 44 43 42 * 1. 1 0 3 6 12 Time (months) 2. 1 1. 0 0 3 6 12 Time (months) * 46 0 3 6 12 Time (months) HDL-C (mg/d. L) 112 102 * Triglycerides (mg/d. L) 114 16 Hemoglobin (g/d. L) 103 Weight (kg) 116 Total cholesterol (mg/d. L) Waist circumference (cm) Nebido® has clinical benefit in obese men with adult-onset hypogonadism 2. 0 1. 9 1. 8 # 1. 7 0 3 6 12 Time (months) *p<0. 0001, **p=0. 001, §p=0. 002, #p=0. 016 versus baseline N=88 obese men with symptomatic adult-onset hypogonadism (mean body mass index: 34. 6 kg/m 2, mean total testosterone: 7. 62 nmol/L, mean age: 51. 1 years) Hb. A 1 c, glycated hemoglobin; HDL-C, high-density lipoprotein cholesterol Canguven O et al. Andrologia. 2017; 49(10): e 12768. 22
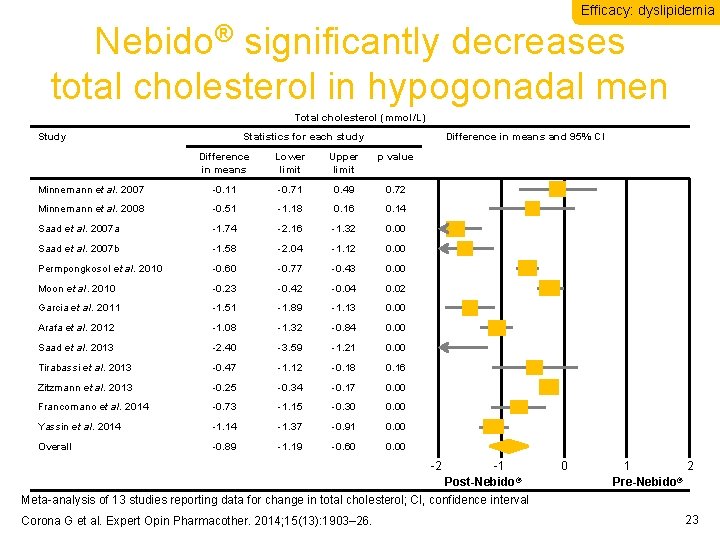
Efficacy: dyslipidemia Nebido® significantly decreases total cholesterol in hypogonadal men Total cholesterol (mmol/L) Study Statistics for each study Difference in means and 95% CI Difference in means Lower limit Upper limit p value Minnemann et al. 2007 -0. 11 -0. 71 0. 49 0. 72 Minnemann et al. 2008 -0. 51 -1. 18 0. 16 0. 14 Saad et al. 2007 a -1. 74 -2. 16 -1. 32 0. 00 Saad et al. 2007 b -1. 58 -2. 04 -1. 12 0. 00 Permpongkosol et al. 2010 -0. 60 -0. 77 -0. 43 0. 00 Moon et al. 2010 -0. 23 -0. 42 -0. 04 0. 02 Garcia et al. 2011 -1. 51 -1. 89 -1. 13 0. 00 Arafa et al. 2012 -1. 08 -1. 32 -0. 84 0. 00 Saad et al. 2013 -2. 40 -3. 59 -1. 21 0. 00 Tirabassi et al. 2013 -0. 47 -1. 12 -0. 18 0. 16 Zitzmann et al. 2013 -0. 25 -0. 34 -0. 17 0. 00 Francomano et al. 2014 -0. 73 -1. 15 -0. 30 0. 00 Yassin et al. 2014 -1. 37 -0. 91 0. 00 Overall -0. 89 -1. 19 -0. 60 0. 00 -2 -1 Post-Nebido® Meta-analysis of 13 studies reporting data for change in total cholesterol; CI, confidence interval Corona G et al. Expert Opin Pharmacother. 2014; 15(13): 1903– 26. 0 1 2 ® Pre-Nebido 23
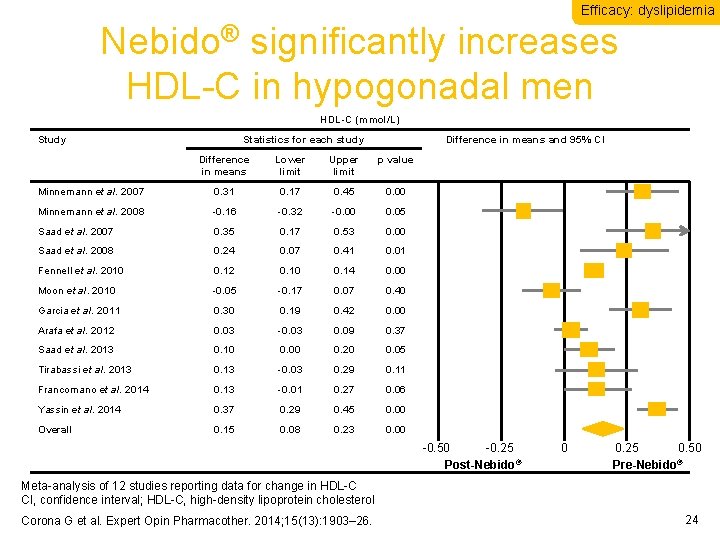
Efficacy: dyslipidemia Nebido® significantly increases HDL-C in hypogonadal men HDL-C (mmol/L) Study Statistics for each study Difference in means and 95% CI Difference in means Lower limit Upper limit p value Minnemann et al. 2007 0. 31 0. 17 0. 45 0. 00 Minnemann et al. 2008 -0. 16 -0. 32 -0. 00 0. 05 Saad et al. 2007 0. 35 0. 17 0. 53 0. 00 Saad et al. 2008 0. 24 0. 07 0. 41 0. 01 Fennell et al. 2010 0. 12 0. 10 0. 14 0. 00 Moon et al. 2010 -0. 05 -0. 17 0. 07 0. 40 Garcia et al. 2011 0. 30 0. 19 0. 42 0. 00 Arafa et al. 2012 0. 03 -0. 03 0. 09 0. 37 Saad et al. 2013 0. 10 0. 00 0. 20 0. 05 Tirabassi et al. 2013 0. 13 -0. 03 0. 29 0. 11 Francomano et al. 2014 0. 13 -0. 01 0. 27 0. 06 Yassin et al. 2014 0. 37 0. 29 0. 45 0. 00 Overall 0. 15 0. 08 0. 23 0. 00 -0. 50 -0. 25 Post-Nebido® 0 0. 25 0. 50 ® Pre-Nebido Meta-analysis of 12 studies reporting data for change in HDL-C CI, confidence interval; HDL-C, high-density lipoprotein cholesterol Corona G et al. Expert Opin Pharmacother. 2014; 15(13): 1903– 26. 24
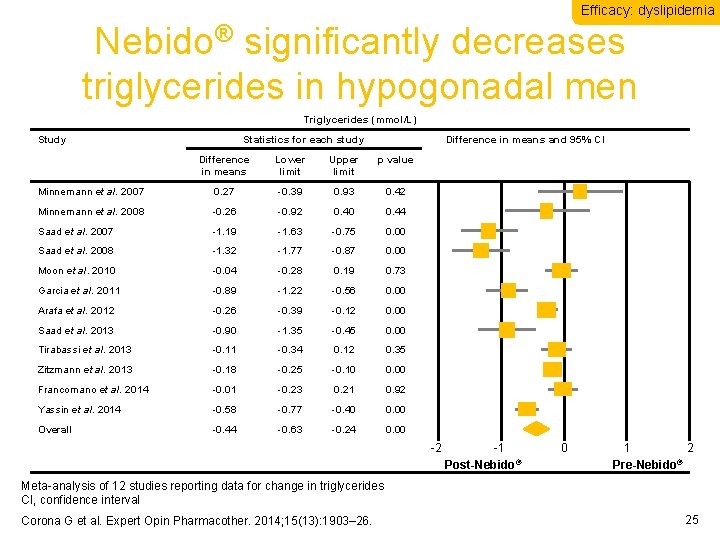
Efficacy: dyslipidemia Nebido® significantly decreases triglycerides in hypogonadal men Triglycerides (mmol/L) Study Statistics for each study Difference in means and 95% CI Difference in means Lower limit Upper limit p value Minnemann et al. 2007 0. 27 -0. 39 0. 93 0. 42 Minnemann et al. 2008 -0. 26 -0. 92 0. 40 0. 44 Saad et al. 2007 -1. 19 -1. 63 -0. 75 0. 00 Saad et al. 2008 -1. 32 -1. 77 -0. 87 0. 00 Moon et al. 2010 -0. 04 -0. 28 0. 19 0. 73 Garcia et al. 2011 -0. 89 -1. 22 -0. 56 0. 00 Arafa et al. 2012 -0. 26 -0. 39 -0. 12 0. 00 Saad et al. 2013 -0. 90 -1. 35 -0. 45 0. 00 Tirabassi et al. 2013 -0. 11 -0. 34 0. 12 0. 35 Zitzmann et al. 2013 -0. 18 -0. 25 -0. 10 0. 00 Francomano et al. 2014 -0. 01 -0. 23 0. 21 0. 92 Yassin et al. 2014 -0. 58 -0. 77 -0. 40 0. 00 Overall -0. 44 -0. 63 -0. 24 0. 00 -2 -1 Post-Nebido® 0 1 2 ® Pre-Nebido Meta-analysis of 12 studies reporting data for change in triglycerides CI, confidence interval Corona G et al. Expert Opin Pharmacother. 2014; 15(13): 1903– 26. 25

Efficacy: metabolic syndrome & diabetes Nebido® but not oral testosterone undecanoate has clinical benefit in hypogonadal men with metabolic syndrome • Only Nebido® was significantly associated with improved metabolic and physical parameters after 6 and 12 months of treatment 6 months Nebido® for 12 months (n=32) 12 months 2 HOMA-IR [mean +SD] 1 0 -1 -2 -3 * Oral testosterone undecanoate for 6 months then switch to Nebido® for next 6 months (n=10) * * 6 months Waist circumference (cm) [mean +SD] Placebo for 12 months (n=10) 12 months 5 0 -5 ** -10 * *p<0. 0001, **p<0. 001 versus baseline N=52 men with hypogonadism [mean total T: <320 ng/d. L (11. 1 nmol/L)] and metabolic syndrome (mean age: 57 years) HOMA-IR, Homeostasis Model Assessment index of Insulin Resistance; SD, standard deviation; T, testosterone Aversa A et al. J Endocrinol Invest. 2010; 33(11): 776– 83. 26
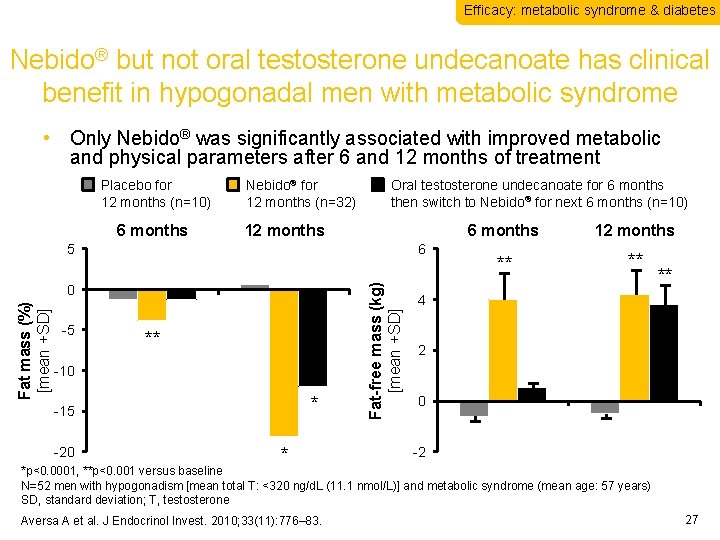
Efficacy: metabolic syndrome & diabetes Nebido® but not oral testosterone undecanoate has clinical benefit in hypogonadal men with metabolic syndrome • Only Nebido® was significantly associated with improved metabolic and physical parameters after 6 and 12 months of treatment Placebo for 12 months (n=10) 6 months Nebido® for 12 months (n=32) Oral testosterone undecanoate for 6 months then switch to Nebido® for next 6 months (n=10) 12 months 6 Fat mass (%) [mean +SD] 0 -5 ** -10 * -15 -20 * Fat-free mass (kg) [mean +SD] 5 6 months 12 months ** ** ** 4 2 0 -2 *p<0. 0001, **p<0. 001 versus baseline N=52 men with hypogonadism [mean total T: <320 ng/d. L (11. 1 nmol/L)] and metabolic syndrome (mean age: 57 years) SD, standard deviation; T, testosterone Aversa A et al. J Endocrinol Invest. 2010; 33(11): 776– 83. 27
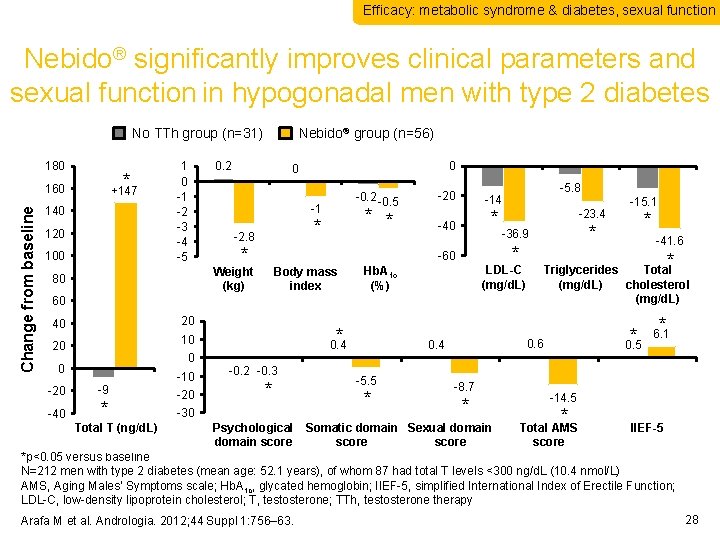
Efficacy: metabolic syndrome & diabetes, sexual function Nebido® significantly improves clinical parameters and sexual function in hypogonadal men with type 2 diabetes No TTh group (n=31) 180 * Change from baseline 160 +147 140 120 100 1 0 -1 -2 -3 -4 -5 Nebido® group (n=56) 0. 2 -0. 5 -1 * * * -2. 8 * -20 -14 -40 * Body mass index -5. 8 -23. 4 20 20 0 0 -20 * 0. 4 10 -9 * -10 -20 -0. 2 -0. 3 * -5. 5 * * 0. 5 0. 6 0. 4 -8. 7 * -41. 6 Triglycerides Total (mg/d. L) cholesterol (mg/d. L) 60 40 * * LDL-C (mg/d. L) Hb. A 1 c (%) -15. 1 * -36. 9 * -60 Weight (kg) 80 0 0 * 6. 1 -14. 5 * -30 Psychological Somatic domain Sexual domain Total AMS IIEF-5 Total T (ng/d. L) Total T domain score (ng/d. L) *p<0. 05 versus baseline N=212 men with type 2 diabetes (mean age: 52. 1 years), of whom 87 had total T levels <300 ng/d. L (10. 4 nmol/L) AMS, Aging Males’ Symptoms scale; Hb. A 1 c, glycated hemoglobin; IIEF-5, simplified International Index of Erectile Function; LDL-C, low-density lipoprotein cholesterol; T, testosterone; TTh, testosterone therapy -40 Arafa M et al. Andrologia. 2012; 44 Suppl 1: 756– 63. 28
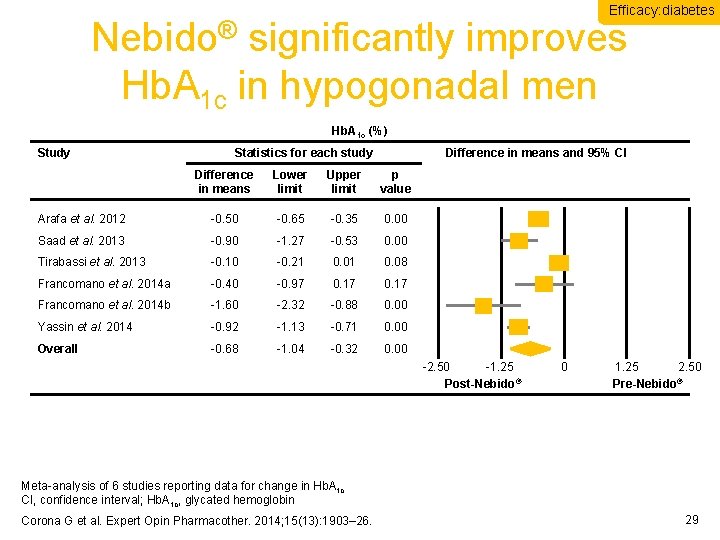
Efficacy: diabetes Nebido® significantly improves Hb. A 1 c in hypogonadal men Hb. A 1 c (%) Study Statistics for each study Difference in means and 95% CI Difference in means Lower limit Upper limit p value Arafa et al. 2012 -0. 50 -0. 65 -0. 35 0. 00 Saad et al. 2013 -0. 90 -1. 27 -0. 53 0. 00 Tirabassi et al. 2013 -0. 10 -0. 21 0. 08 Francomano et al. 2014 a -0. 40 -0. 97 0. 17 Francomano et al. 2014 b -1. 60 -2. 32 -0. 88 0. 00 Yassin et al. 2014 -0. 92 -1. 13 -0. 71 0. 00 Overall -0. 68 -1. 04 -0. 32 0. 00 -2. 50 -1. 25 Post-Nebido® 0 1. 25 2. 50 Pre-Nebido® Meta-analysis of 6 studies reporting data for change in Hb. A 1 c CI, confidence interval; Hb. A 1 c, glycated hemoglobin Corona G et al. Expert Opin Pharmacother. 2014; 15(13): 1903– 26. 29

Module 5: Nebido® clinical efficacy General well-being and sexual function 30
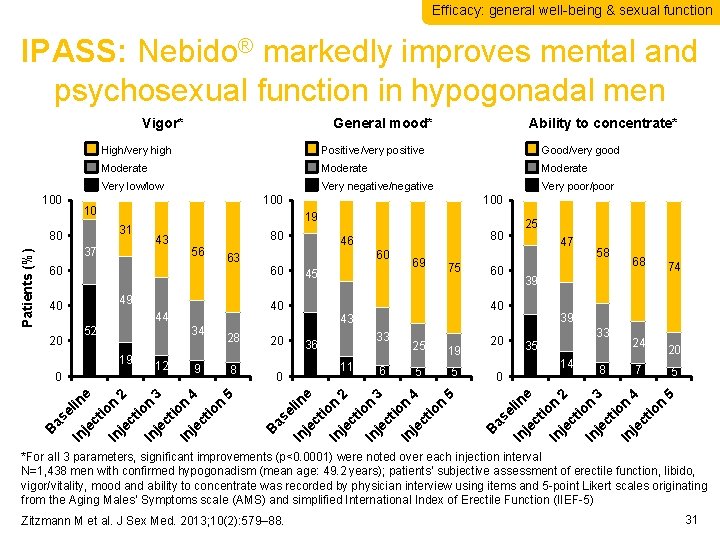
Efficacy: general well-being & sexual function IPASS: Nebido® markedly improves mental and psychosexual function in hypogonadal men Vigor* Good/very good Moderate Very low/low Very negative/negative Very poor/poor 24 5 n In je n 4 7 ct io je In 20 ct io 3 8 n 2 n In je lin as e B 74 5 14 0 n 4 ct io n je In ct io 33 35 e 5 20 ct io 5 68 39 je 19 58 39 In 25 6 3 2 60 5 11 0 ct io 8 33 n 9 47 40 36 je In as e B 12 lin e je ct io n In 2 je ct io n In 3 je ct io n In 4 je ct io n 5 0 20 In 19 28 75 43 e 34 69 45 ct io 20 60 40 44 52 60 n 49 40 46 je 60 63 25 80 In 56 je 43 80 lin 37 19 In 31 100 ct io 100 as e Patients (%) Positive/very positive 10 80 Ability to concentrate* High/very high B 100 General mood* *For all 3 parameters, significant improvements (p<0. 0001) were noted over each injection interval N=1, 438 men with confirmed hypogonadism (mean age: 49. 2 years); patients’ subjective assessment of erectile function, libido, vigor/vitality, mood and ability to concentrate was recorded by physician interview using items and 5 -point Likert scales originating from the Aging Males’ Symptoms scale (AMS) and simplified International Index of Erectile Function (IIEF-5) Zitzmann M et al. J Sex Med. 2013; 10(2): 579– 88. 31
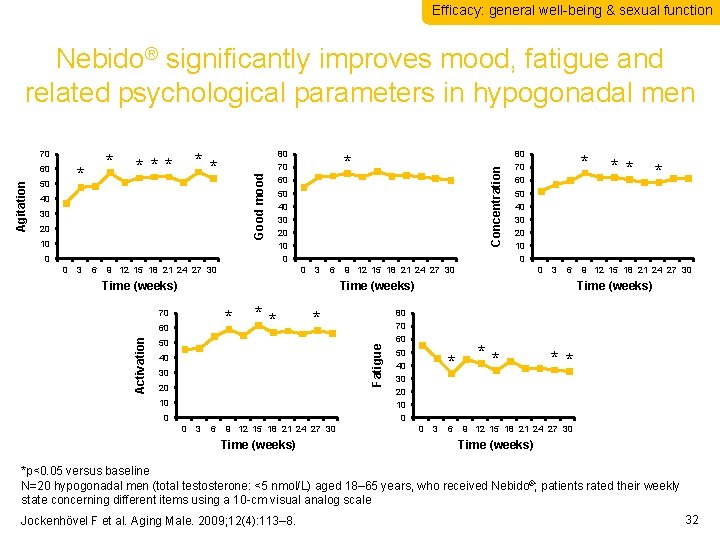
Efficacy: general well-being & sexual function Nebido® significantly improves mood, fatigue and related psychological parameters in hypogonadal men * 50 ** *** 40 30 20 10 0 0 3 6 80 70 60 50 40 30 20 10 0 9 12 15 18 21 24 27 30 * 0 3 6 Time (weeks) * 70 60 ** * 50 Fatigue Activation Concentration Agitation 60 * Good mood 70 40 30 20 10 0 0 3 6 9 12 15 18 21 24 27 30 Time (weeks) ** * 9 12 15 18 21 24 27 30 Time (weeks) 80 70 60 50 40 30 20 10 0 * 0 3 6 ** ** 9 12 15 18 21 24 27 30 Time (weeks) *p<0. 05 versus baseline N=20 hypogonadal men (total testosterone: <5 nmol/L) aged 18– 65 years, who received Nebido®; patients rated their weekly state concerning different items using a 10 -cm visual analog scale Jockenhövel F et al. Aging Male. 2009; 12(4): 113– 8. 32
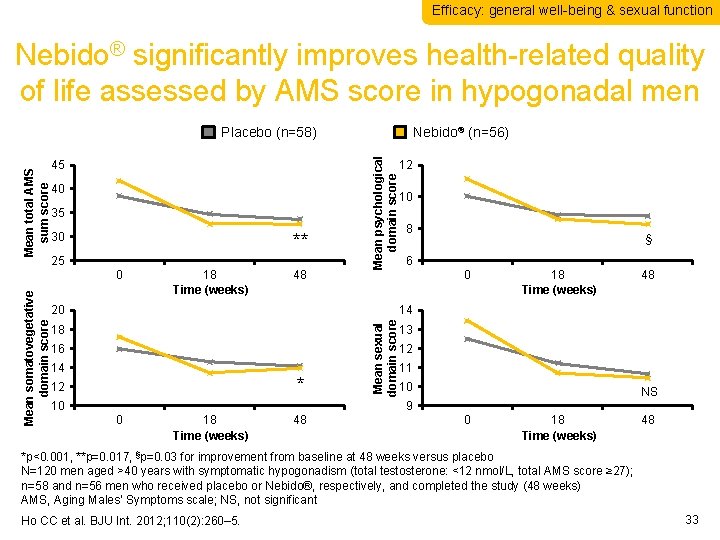
Efficacy: general well-being & sexual function Nebido® significantly improves health-related quality of life assessed by AMS score in hypogonadal men 40 35 30 ** 25 Mean somatovegetative domain score 0 18 Time (weeks) 48 Mean psychological domain score 45 Nebido® (n=56) 20 12 10 8 § 6 0 18 Time (weeks) 48 14 18 16 14 * 12 10 Mean sexual domain score Mean total AMS sum score Placebo (n=58) 13 12 11 10 NS 9 0 18 Time (weeks) 48 *p<0. 001, **p=0. 017, §p=0. 03 for improvement from baseline at 48 weeks versus placebo N=120 men aged >40 years with symptomatic hypogonadism (total testosterone: <12 nmol/L, total AMS score ≥ 27); n=58 and n=56 men who received placebo or Nebido®, respectively, and completed the study (48 weeks) AMS, Aging Males’ Symptoms scale; NS, not significant Ho CC et al. BJU Int. 2012; 110(2): 260– 5. 33
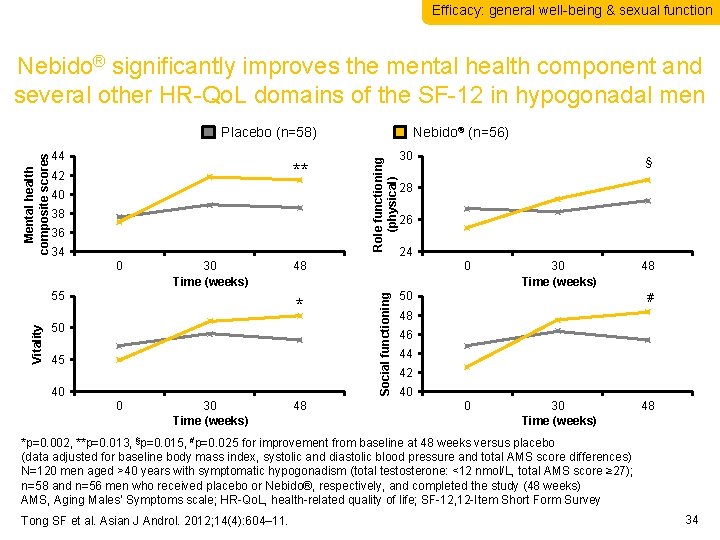
Efficacy: general well-being & sexual function Nebido® significantly improves the mental health component and several other HR-Qo. L domains of the SF-12 in hypogonadal men ** 42 40 38 36 34 0 30 Time (weeks) Vitality 55 50 45 40 30 Time (weeks) 30 48 § 28 26 24 48 * 0 Role functioning (physical) 44 Nebido® (n=56) 0 Social functioning Mental health composite scores Placebo (n=58) 30 Time (weeks) 50 48 # 48 46 44 42 40 0 30 Time (weeks) 48 *p=0. 002, **p=0. 013, §p=0. 015, #p=0. 025 for improvement from baseline at 48 weeks versus placebo (data adjusted for baseline body mass index, systolic and diastolic blood pressure and total AMS score differences) N=120 men aged >40 years with symptomatic hypogonadism (total testosterone: <12 nmol/L, total AMS score ≥ 27); n=58 and n=56 men who received placebo or Nebido®, respectively, and completed the study (48 weeks) AMS, Aging Males’ Symptoms scale; HR-Qo. L, health-related quality of life; SF-12, 12 -Item Short Form Survey Tong SF et al. Asian J Androl. 2012; 14(4): 604– 11. 34
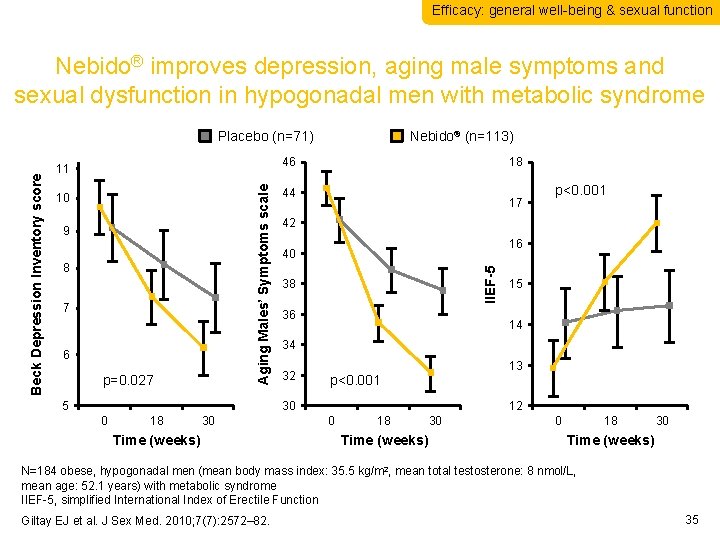
Efficacy: general well-being & sexual function Nebido® improves depression, aging male symptoms and sexual dysfunction in hypogonadal men with metabolic syndrome Nebido® (n=113) 46 10 9 8 7 6 p=0. 027 5 18 44 17 16 40 38 36 18 30 Time (weeks) 15 14 34 32 13 p<0. 001 30 0 p<0. 001 42 IIEF-5 11 Aging Males’ Symptoms scale Beck Depression Inventory score Placebo (n=71) 12 0 18 Time (weeks) 30 0 18 30 Time (weeks) N=184 obese, hypogonadal men (mean body mass index: 35. 5 kg/m 2, mean total testosterone: 8 nmol/L, mean age: 52. 1 years) with metabolic syndrome IIEF-5, simplified International Index of Erectile Function Giltay EJ et al. J Sex Med. 2010; 7(7): 2572– 82. 35
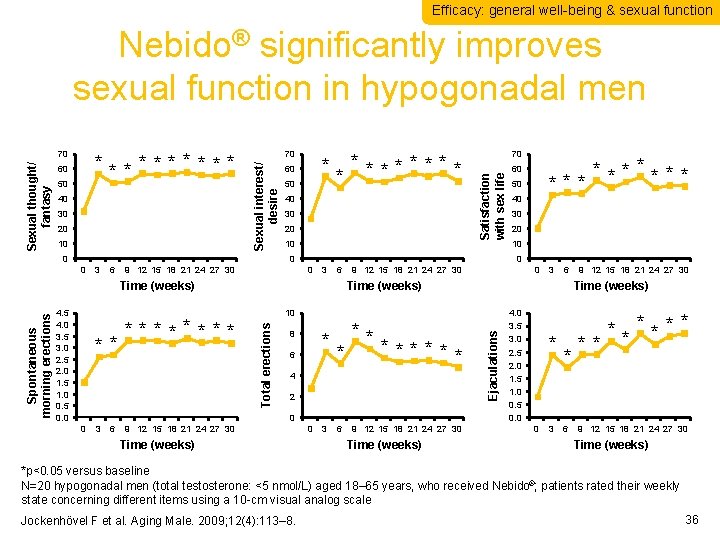
Efficacy: general well-being & sexual function Nebido® significantly improves sexual function in hypogonadal men 60 50 40 30 20 10 70 0 * **** * 60 50 40 30 20 10 0 3 6 9 12 15 18 21 24 27 30 3 6 Time (weeks) ** **** 3 6 40 30 20 10 9 12 15 18 21 24 27 30 0 3 6 ** * ****** * 8 6 4 2 9 12 15 18 21 24 27 30 Time (weeks) 0 3 6 9 12 15 18 21 24 27 30 Time (weeks) 10 0 0 50 Time (weeks) Total erections 4. 5 4. 0 3. 5 3. 0 2. 5 2. 0 1. 5 1. 0 0. 5 0. 0 ******* 60 0 0 Ejaculations 0 Spontaneous morning erections 70 Satisfaction with sex life ***** Sexual interest/ desire Sexual thought/ fantasy 70 4. 0 3. 5 3. 0 2. 5 2. 0 1. 5 1. 0 0. 5 0. 0 * 0 3 * 6 ****** ** 9 12 15 18 21 24 27 30 Time (weeks) *p<0. 05 versus baseline N=20 hypogonadal men (total testosterone: <5 nmol/L) aged 18– 65 years, who received Nebido®; patients rated their weekly state concerning different items using a 10 -cm visual analog scale Jockenhövel F et al. Aging Male. 2009; 12(4): 113– 8. 36
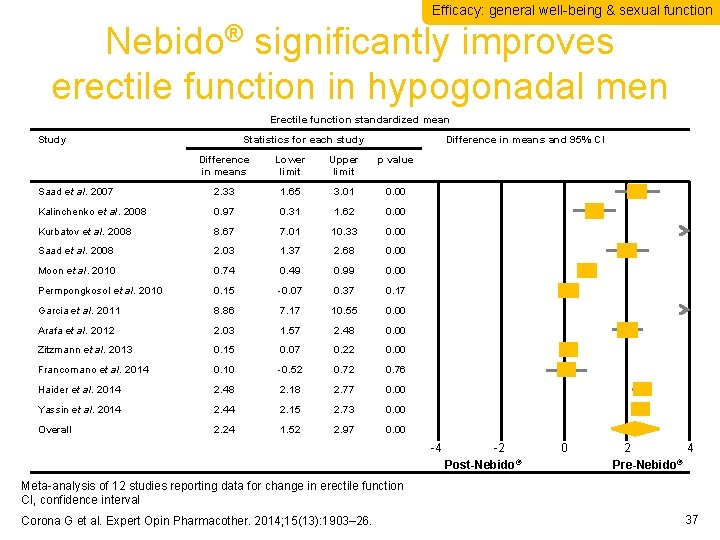
Efficacy: general well-being & sexual function Nebido® significantly improves erectile function in hypogonadal men Erectile function standardized mean Study Statistics for each study Difference in means and 95% CI Difference in means Lower limit Upper limit p value Saad et al. 2007 2. 33 1. 65 3. 01 0. 00 Kalinchenko et al. 2008 0. 97 0. 31 1. 62 0. 00 Kurbatov et al. 2008 8. 67 7. 01 10. 33 0. 00 Saad et al. 2008 2. 03 1. 37 2. 68 0. 00 Moon et al. 2010 0. 74 0. 49 0. 99 0. 00 Permpongkosol et al. 2010 0. 15 -0. 07 0. 37 0. 17 Garcia et al. 2011 8. 86 7. 17 10. 55 0. 00 Arafa et al. 2012 2. 03 1. 57 2. 48 0. 00 Zitzmann et al. 2013 0. 15 0. 07 0. 22 0. 00 Francomano et al. 2014 0. 10 -0. 52 0. 76 Haider et al. 2014 2. 48 2. 18 2. 77 0. 00 Yassin et al. 2014 2. 44 2. 15 2. 73 0. 00 Overall 2. 24 1. 52 2. 97 0. 00 -4 -2 Post-Nebido® 0 2 4 ® Pre-Nebido Meta-analysis of 12 studies reporting data for change in erectile function CI, confidence interval Corona G et al. Expert Opin Pharmacother. 2014; 15(13): 1903– 26. 37
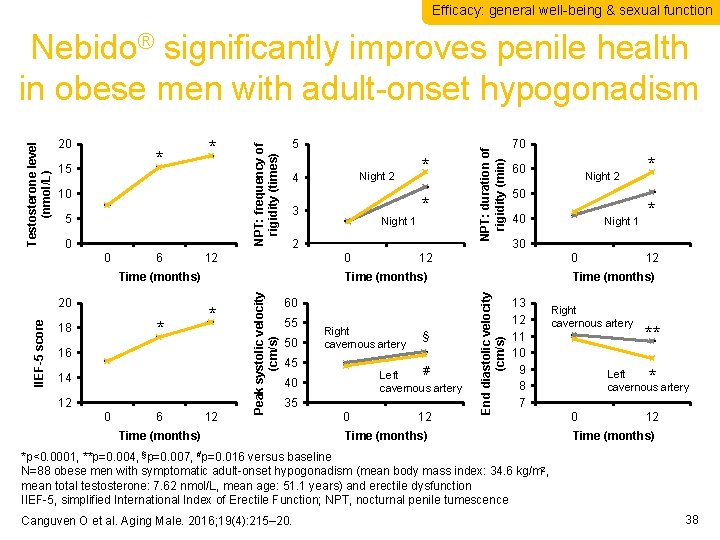
Efficacy: general well-being & sexual function 10 5 0 0 6 5 3 12 0 * 16 14 12 0 6 * 70 60 40 12 55 50 Right cavernous artery 45 § # Left cavernous artery 40 35 Time (months) 0 12 Night 1 * * 30 12 60 Night 2 50 0 Time (months) Peak systolic velocity (cm/s) IIEF-5 score * 18 Night 1 * 2 Time (months) 20 Night 2 4 NPT: duration of rigidity (min) * 15 * 12 Time (months) End diastolic velocity (cm/s) 20 NPT: frequency of rigidity (times) Testosterone level (nmol/L) Nebido® significantly improves penile health in obese men with adult-onset hypogonadism 13 12 11 10 9 8 7 Time (months) Right cavernous artery ** * Left cavernous artery 0 12 Time (months) *p<0. 0001, **p=0. 004, §p=0. 007, #p=0. 016 versus baseline N=88 obese men with symptomatic adult-onset hypogonadism (mean body mass index: 34. 6 kg/m 2, mean total testosterone: 7. 62 nmol/L, mean age: 51. 1 years) and erectile dysfunction IIEF-5, simplified International Index of Erectile Function; NPT, nocturnal penile tumescence Canguven O et al. Aging Male. 2016; 19(4): 215– 20. 38
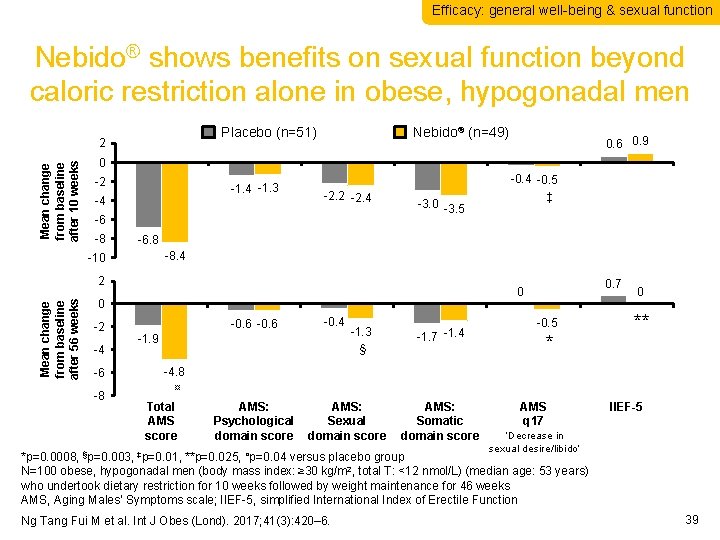
Efficacy: general well-being & sexual function Nebido® shows benefits on sexual function beyond caloric restriction alone in obese, hypogonadal men Placebo (n=51) Mean change from baseline after 10 weeks 2 0. 6 0. 9 0 -2 -1. 4 -1. 3 -4 -2. 2 -2. 4 -6 -8 -10 2 Mean change from baseline after 56 weeks Nebido® (n=49) 0 -2 -4 -6 -8 -3. 0 -3. 5 -0. 4 -0. 5 ‡ -6. 8 -8. 4 Total AMS score AMS: Psychological domain score -0. 6 AMS: Sexual domain score -0. 4 -1. 9 -1. 3 § AMS: Somatic domain score -1. 7 -1. 4 AMS q 17 0 -0. 5 * IIEF-5 0. 7 0 ** -4. 8 ¤ Total AMS score AMS: Psychological domain score AMS: Sexual domain score AMS: Somatic domain score AMS q 17 IIEF-5 ‘Decrease in sexual desire/libido’ *p=0. 0008, §p=0. 003, ‡p=0. 01, **p=0. 025, ¤p=0. 04 versus placebo group N=100 obese, hypogonadal men (body mass index: ≥ 30 kg/m 2, total T: <12 nmol/L) (median age: 53 years) who undertook dietary restriction for 10 weeks followed by weight maintenance for 46 weeks AMS, Aging Males’ Symptoms scale; IIEF-5, simplified International Index of Erectile Function Ng Tang Fui M et al. Int J Obes (Lond). 2017; 41(3): 420– 6. 39
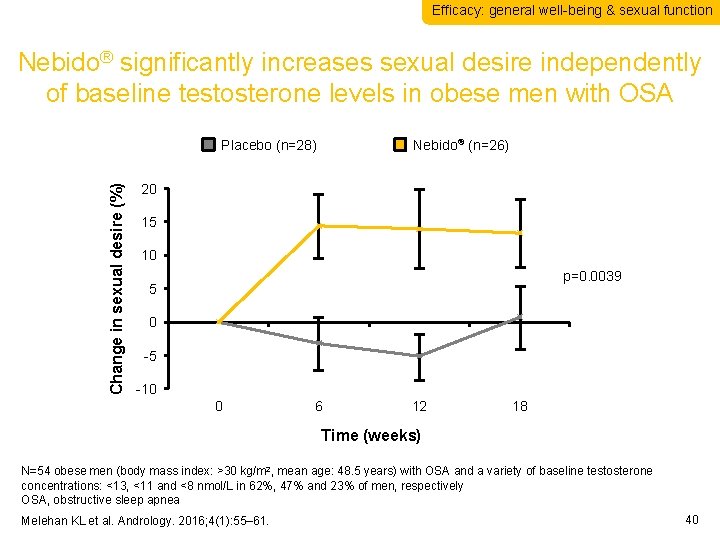
Efficacy: general well-being & sexual function Nebido® significantly increases sexual desire independently of baseline testosterone levels in obese men with OSA Change in sexual desire (%) Placebo (n=28) Nebido® (n=26) 20 15 10 p=0. 0039 5 0 -5 -10 0 6 12 18 Time (weeks) N=54 obese men (body mass index: >30 kg/m 2, mean age: 48. 5 years) with OSA and a variety of baseline testosterone concentrations: <13, <11 and <8 nmol/L in 62%, 47% and 23% of men, respectively OSA, obstructive sleep apnea Melehan KL et al. Andrology. 2016; 4(1): 55– 61. 40
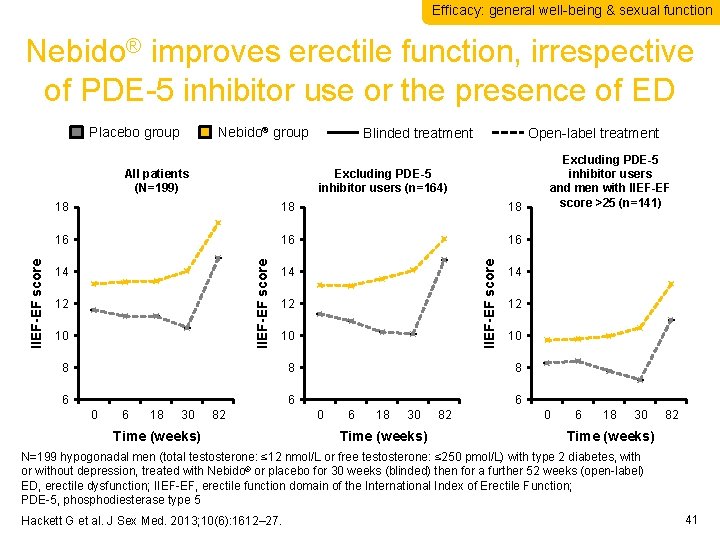
Efficacy: general well-being & sexual function Nebido® improves erectile function, irrespective of PDE-5 inhibitor use or the presence of ED Placebo group Nebido® group Open-label treatment Excluding PDE-5 inhibitor users (n=164) 18 18 16 16 16 14 12 10 IIEF-EF score 18 IIEF-EF score All patients (N=199) Blinded treatment 14 12 10 8 8 6 6 18 30 82 Time (weeks) 14 12 10 8 6 0 Excluding PDE-5 inhibitor users and men with IIEF-EF score >25 (n=141) 6 0 6 18 30 Time (weeks) 82 0 6 18 30 82 Time (weeks) N=199 hypogonadal men (total testosterone: ≤ 12 nmol/L or free testosterone: ≤ 250 pmol/L) with type 2 diabetes, with or without depression, treated with Nebido® or placebo for 30 weeks (blinded) then for a further 52 weeks (open-label) ED, erectile dysfunction; IIEF-EF, erectile function domain of the International Index of Erectile Function; PDE-5, phosphodiesterase type 5 Hackett G et al. J Sex Med. 2013; 10(6): 1612– 27. 41
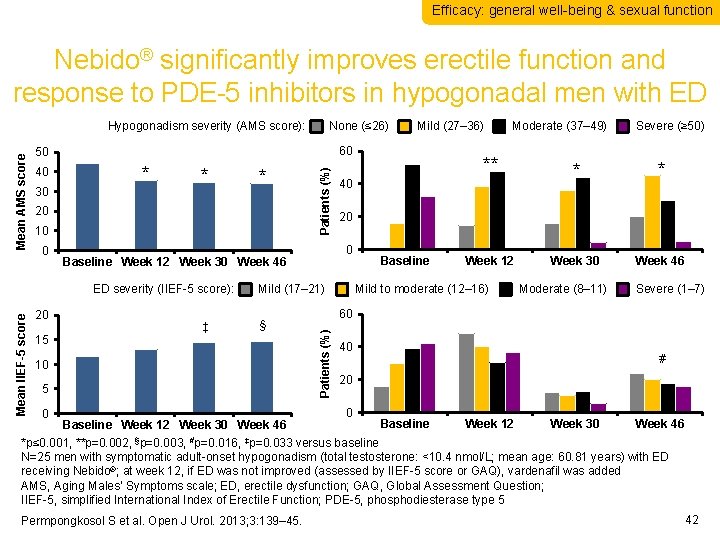
Efficacy: general well-being & sexual function Nebido® significantly improves erectile function and response to PDE-5 inhibitors in hypogonadal men with ED None (≤ 26) 30 * * 20 10 0 20 ‡ 15 10 5 0 Baseline Week 12 Week 30 Week 46 ** Severe (≥ 50) * * Week 30 Week 46 20 Mild (17– 21) § Moderate (37– 49) 40 0 Baseline Week 12 Week 30 Week 46 ED severity (IIEF-5 score): Mean IIEF-5 score * Patients (%) 40 Mild (27– 36) 60 50 Baseline Week 12 Mild to moderate (12– 16) Moderate (8– 11) Severe (1– 7) 60 Patients (%) Mean AMS score Hypogonadism severity (AMS score): 40 # 20 0 Baseline Week 12 Week 30 Week 46 *p≤ 0. 001, **p=0. 002, §p=0. 003, #p=0. 016, ‡p=0. 033 versus baseline N=25 men with symptomatic adult-onset hypogonadism (total testosterone: <10. 4 nmol/L; mean age: 60. 81 years) with ED receiving Nebido®; at week 12, if ED was not improved (assessed by IIEF-5 score or GAQ), vardenafil was added AMS, Aging Males’ Symptoms scale; ED, erectile dysfunction; GAQ, Global Assessment Question; IIEF-5, simplified International Index of Erectile Function; PDE-5, phosphodiesterase type 5 Permpongkosol S et al. Open J Urol. 2013; 3: 139– 45. 42
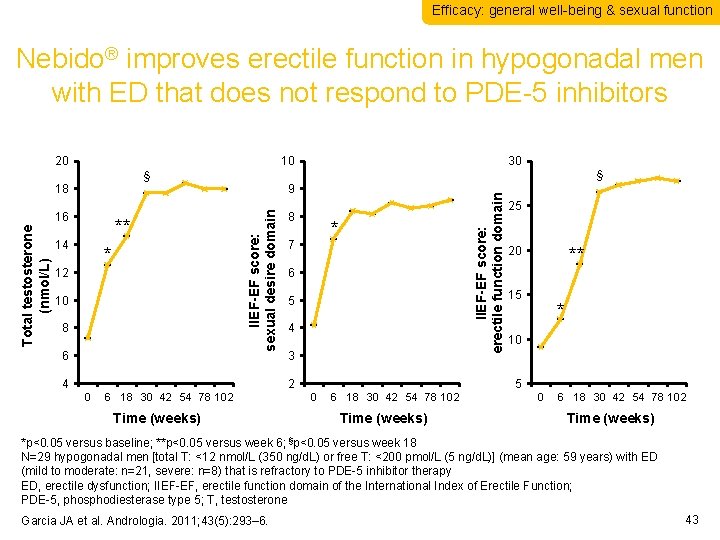
Efficacy: general well-being & sexual function Nebido® improves erectile function in hypogonadal men with ED that does not respond to PDE-5 inhibitors 20 10 30 § § Total testosterone (nmol/L) 16 ** 14 * 12 10 8 6 4 8 IIEF-EF score: erectile function domain 9 IIEF-EF score: sexual desire domain 18 * 7 6 5 4 3 2 0 6 18 30 42 54 78 102 Time (weeks) 25 20 ** 15 * 10 5 0 6 18 30 42 54 78 102 Time (weeks) *p<0. 05 versus baseline; **p<0. 05 versus week 6; §p<0. 05 versus week 18 N=29 hypogonadal men [total T: <12 nmol/L (350 ng/d. L) or free T: <200 pmol/L (5 ng/d. L)] (mean age: 59 years) with ED (mild to moderate: n=21, severe: n=8) that is refractory to PDE-5 inhibitor therapy ED, erectile dysfunction; IIEF-EF, erectile function domain of the International Index of Erectile Function; PDE-5, phosphodiesterase type 5; T, testosterone Garcia JA et al. Andrologia. 2011; 43(5): 293– 6. 43
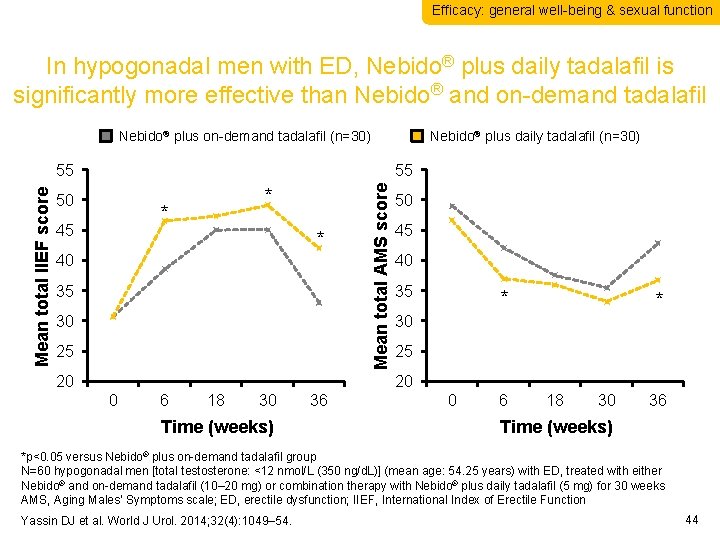
Efficacy: general well-being & sexual function In hypogonadal men with ED, Nebido® plus daily tadalafil is significantly more effective than Nebido® and on-demand tadalafil Nebido® plus on-demand tadalafil (n=30) 55 50 * * 45 * 40 35 30 25 20 Mean total AMS score Mean total IIEF score 55 Nebido® plus daily tadalafil (n=30) 50 45 40 35 * * 30 25 20 0 6 18 30 Time (weeks) 36 0 6 18 30 36 Time (weeks) *p<0. 05 versus Nebido® plus on-demand tadalafil group N=60 hypogonadal men [total testosterone: <12 nmol/L (350 ng/d. L)] (mean age: 54. 25 years) with ED, treated with either Nebido® and on-demand tadalafil (10– 20 mg) or combination therapy with Nebido® plus daily tadalafil (5 mg) for 30 weeks AMS, Aging Males’ Symptoms scale; ED, erectile dysfunction; IIEF, International Index of Erectile Function Yassin DJ et al. World J Urol. 2014; 32(4): 1049– 54. 44
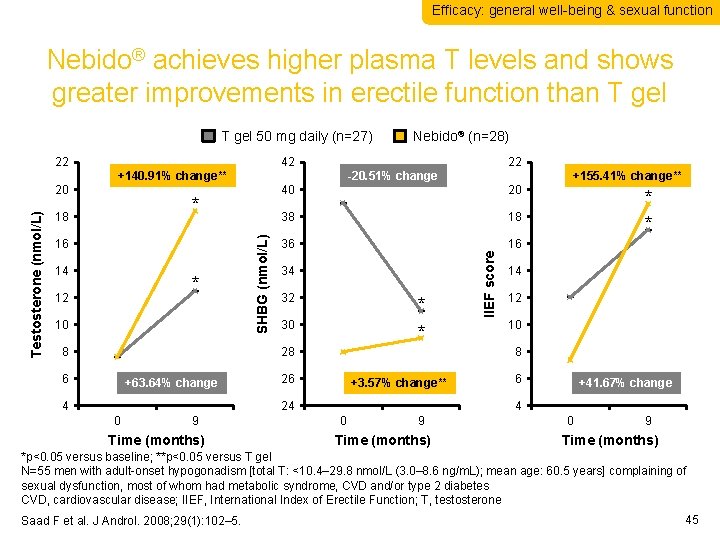
Efficacy: general well-being & sexual function Nebido® achieves higher plasma T levels and shows greater improvements in erectile function than T gel 50 mg daily (n=27) * 18 16 14 * 12 10 8 40 20 38 18 36 16 34 32 * * 30 28 6 +63. 64% change 4 9 Time (months) +155. 41% change** * * 14 12 10 8 26 +3. 57% change** 24 0 22 -20. 51% change IIEF score 20 Testosterone (nmol/L) 42 +140. 91% change** SHBG (nmol/L) 22 Nebido® (n=28) 6 +41. 67% change 4 0 9 Time (months) *p<0. 05 versus baseline; **p<0. 05 versus T gel N=55 men with adult-onset hypogonadism [total T: <10. 4– 29. 8 nmol/L (3. 0– 8. 6 ng/m. L); mean age: 60. 5 years] complaining of sexual dysfunction, most of whom had metabolic syndrome, CVD and/or type 2 diabetes CVD, cardiovascular disease; IIEF, International Index of Erectile Function; T, testosterone Saad F et al. J Androl. 2008; 29(1): 102– 5. 45
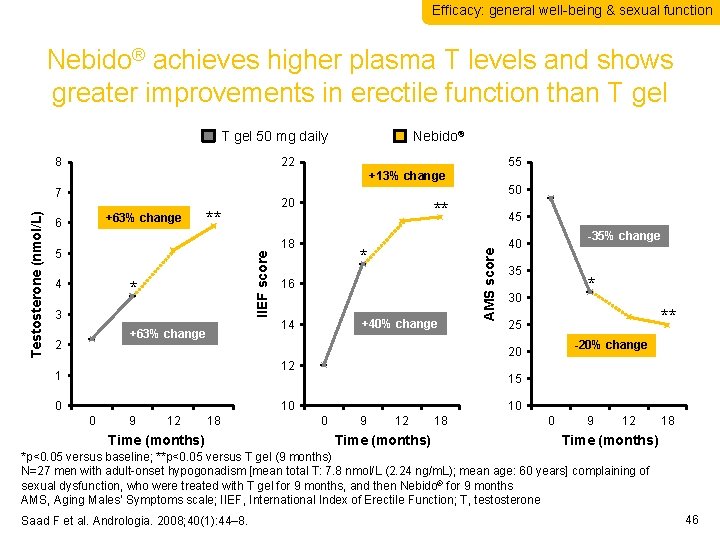
Efficacy: general well-being & sexual function Nebido® achieves higher plasma T levels and shows greater improvements in erectile function than T gel 50 mg daily 8 22 55 +13% change 50 +63% change 6 20 ** ** 18 4 IIEF score 5 * 3 16 +40% change 14 +63% change 2 * 45 AMS score 7 Testosterone (nmol/L) Nebido® -35% change 40 35 * 30 ** 25 -20% change 20 12 1 15 0 10 0 9 12 18 Time (months) 10 0 9 12 Time (months) 18 0 9 12 18 Time (months) *p<0. 05 versus baseline; **p<0. 05 versus T gel (9 months) N=27 men with adult-onset hypogonadism [mean total T: 7. 8 nmol/L (2. 24 ng/m. L); mean age: 60 years] complaining of sexual dysfunction, who were treated with T gel for 9 months, and then Nebido® for 9 months AMS, Aging Males’ Symptoms scale; IIEF, International Index of Erectile Function; T, testosterone Saad F et al. Andrologia. 2008; 40(1): 44– 8. 46
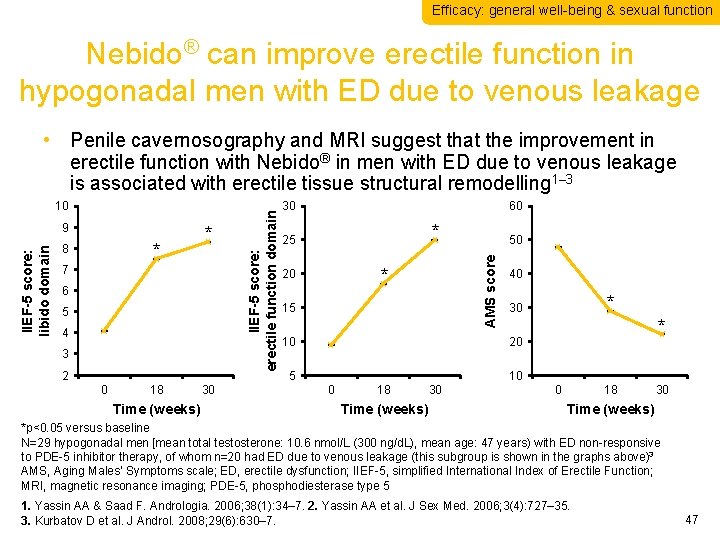
Efficacy: general well-being & sexual function Nebido® can improve erectile function in hypogonadal men with ED due to venous leakage IIEF-5 score: libido domain 9 * 8 * 7 6 5 4 3 2 0 18 Time (weeks) 30 30 60 * 25 * 20 50 AMS score 10 IIEF-5 score: erectile function domain • Penile cavernosography and MRI suggest that the improvement in erectile function with Nebido® in men with ED due to venous leakage is associated with erectile tissue structural remodelling 1– 3 15 40 10 20 5 10 0 18 Time (weeks) 30 * 0 18 30 Time (weeks) *p<0. 05 versus baseline N=29 hypogonadal men [mean total testosterone: 10. 6 nmol/L (300 ng/d. L), mean age: 47 years) with ED non-responsive to PDE-5 inhibitor therapy, of whom n=20 had ED due to venous leakage (this subgroup is shown in the graphs above)3 AMS, Aging Males’ Symptoms scale; ED, erectile dysfunction; IIEF-5, simplified International Index of Erectile Function; MRI, magnetic resonance imaging; PDE-5, phosphodiesterase type 5 1. Yassin AA & Saad F. Andrologia. 2006; 38(1): 34– 7. 2. Yassin AA et al. J Sex Med. 2006; 3(4): 727– 35. 3. Kurbatov D et al. J Androl. 2008; 29(6): 630– 7. 47
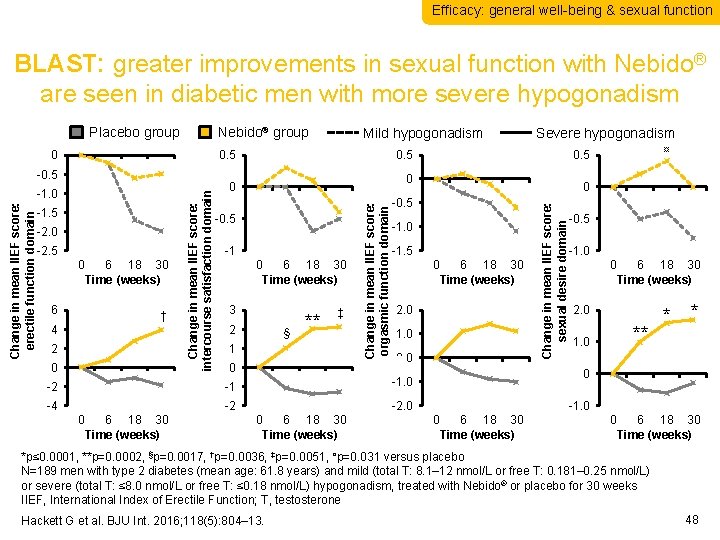
Efficacy: general well-being & sexual function BLAST: greater improvements in sexual function with Nebido® are seen in diabetic men with more severe hypogonadism Nebido® group 0. 5 Change in mean IIEF score: erectile function domain -1. 0 -1. 5 -2. 0 -2. 5 0 6 6 18 30 Time (weeks) † 4 2 0 Change in mean IIEF score: intercourse satisfaction domain -0. 5 -1 0 6 18 30 Time (weeks) 3 2 § ** ‡ 1 0 -1. 5 0 1. 0 0. 0 -4 -2 -2. 0 0 6 18 30 Time (weeks) 2. 0 -1. 0 6 18 30 Time (weeks) 0. 0 -0. 5 -1 ¤ 0. 5 0. 0 0 -2 0 Severe hypogonadism 0. 5 Change in mean IIEF score: orgasmic function domain 0. 0 Mild hypogonadism Change in mean IIEF score: sexual desire domain Placebo group -0. 5 -1. 0 0 6 18 30 Time (weeks) 2. 0 ** 1. 0 * * 0. 0 -1. 0 0 6 18 30 Time (weeks) *p≤ 0. 0001, **p=0. 0002, §p=0. 0017, †p=0. 0036, ‡p=0. 0051, ¤p=0. 031 versus placebo N=189 men with type 2 diabetes (mean age: 61. 8 years) and mild (total T: 8. 1– 12 nmol/L or free T: 0. 181– 0. 25 nmol/L) or severe (total T: ≤ 8. 0 nmol/L or free T: ≤ 0. 18 nmol/L) hypogonadism, treated with Nebido® or placebo for 30 weeks IIEF, International Index of Erectile Function; T, testosterone Hackett G et al. BJU Int. 2016; 118(5): 804– 13. 48

Module 5: Nebido® clinical efficacy LUTS 49
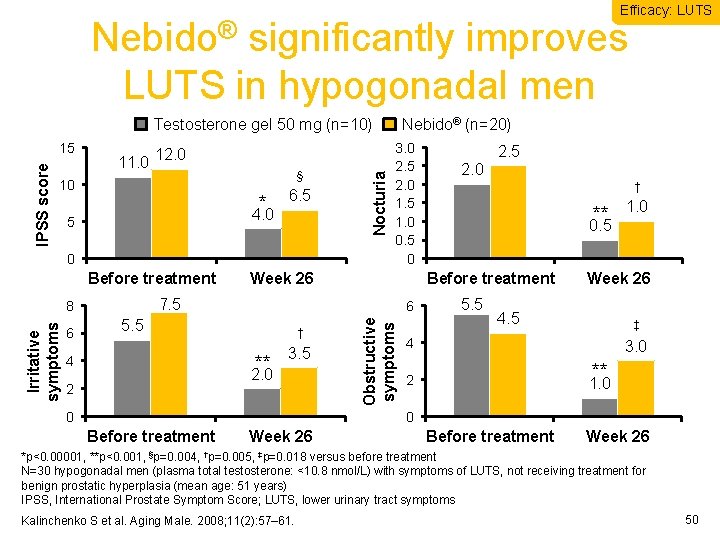
Efficacy: LUTS Nebido® significantly improves LUTS in hypogonadal men 11. 0 12. 0 10 § * 6. 5 4. 0 5 0 Before treatment Irritative symptoms 3. 0 2. 5 2. 0 1. 5 1. 0 0. 5 0. 0 Week 26 6 5. 5 † ** 4 3. 5 2. 0 2. 5 2. 0 † ** 0. 5 Before treatment 7. 5 8 6 Nebido® (n=20) Obstructive symptoms IPSS score 15 Nocturia Testosterone gel 50 mg (n=10) 5. 5 1. 0 Week 26 4. 5 ‡ 4 3. 0 ** 2 1. 0 0 Before treatment Week 26 *p<0. 00001, **p<0. 001, §p=0. 004, †p=0. 005, ‡p=0. 018 versus before treatment N=30 hypogonadal men (plasma total testosterone: <10. 8 nmol/L) with symptoms of LUTS, not receiving treatment for benign prostatic hyperplasia (mean age: 51 years) IPSS, International Prostate Symptom Score; LUTS, lower urinary tract symptoms Kalinchenko S et al. Aging Male. 2008; 11(2): 57– 61. 50
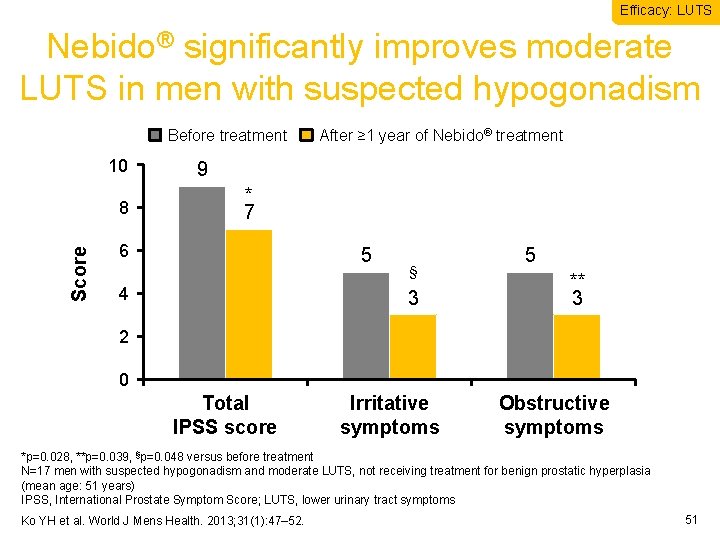
Efficacy: LUTS Nebido® significantly improves moderate LUTS in men with suspected hypogonadism Before treatment 10 Score 8 After ≥ 1 year of Nebido® treatment 9 * 7 6 5 4 § 3 5 ** 3 2 0 Total IPSS score Irritative symptoms Obstructive symptoms *p=0. 028, **p=0. 039, §p=0. 048 versus before treatment N=17 men with suspected hypogonadism and moderate LUTS, not receiving treatment for benign prostatic hyperplasia (mean age: 51 years) IPSS, International Prostate Symptom Score; LUTS, lower urinary tract symptoms Ko YH et al. World J Mens Health. 2013; 31(1): 47– 52. 51
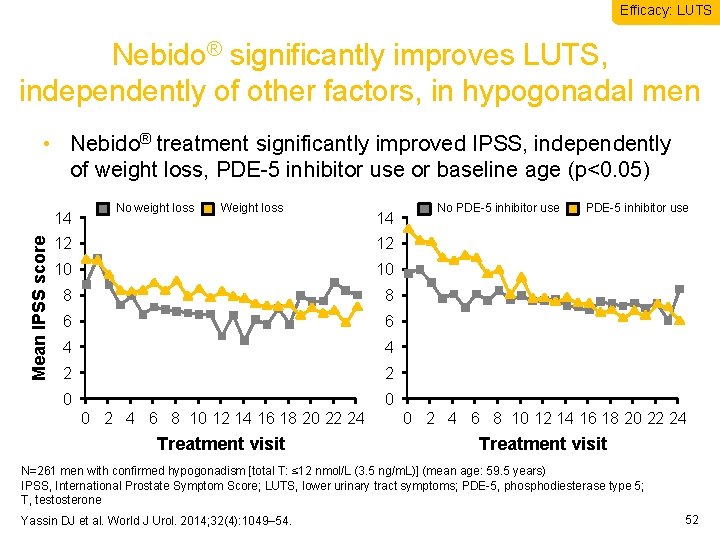
Efficacy: LUTS Nebido® significantly improves LUTS, independently of other factors, in hypogonadal men • Nebido® treatment significantly improved IPSS, independently of weight loss, PDE-5 inhibitor use or baseline age (p<0. 05) Mean IPSS score 14 No weight loss Weight loss 14 12 12 10 10 8 8 6 6 4 4 2 2 0 0 No PDE-5 inhibitor use 0 2 4 6 8 10 12 14 16 18 20 22 24 Treatment visit N=261 men with confirmed hypogonadism [total T: ≤ 12 nmol/L (3. 5 ng/m. L)] (mean age: 59. 5 years) IPSS, International Prostate Symptom Score; LUTS, lower urinary tract symptoms; PDE-5, phosphodiesterase type 5; T, testosterone Yassin DJ et al. World J Urol. 2014; 32(4): 1049– 54. 52
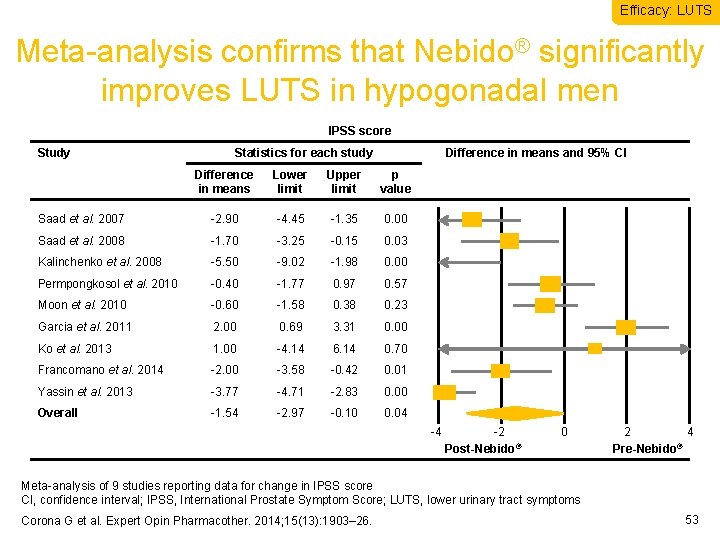
Efficacy: LUTS Meta-analysis confirms that Nebido® significantly improves LUTS in hypogonadal men IPSS score Study Statistics for each study Difference in means and 95% CI Difference in means Lower limit Upper limit p value Saad et al. 2007 -2. 90 -4. 45 -1. 35 0. 00 Saad et al. 2008 -1. 70 -3. 25 -0. 15 0. 03 Kalinchenko et al. 2008 -5. 50 -9. 02 -1. 98 0. 00 Permpongkosol et al. 2010 -0. 40 -1. 77 0. 97 0. 57 Moon et al. 2010 -0. 60 -1. 58 0. 38 0. 23 Garcia et al. 2011 2. 00 0. 69 3. 31 0. 00 Ko et al. 2013 1. 00 -4. 14 6. 14 0. 70 Francomano et al. 2014 -2. 00 -3. 58 -0. 42 0. 01 Yassin et al. 2013 -3. 77 -4. 71 -2. 83 0. 00 Overall -1. 54 -2. 97 -0. 10 0. 04 -4 -2 Post-Nebido® 0 2 4 ® Pre-Nebido Meta-analysis of 9 studies reporting data for change in IPSS score CI, confidence interval; IPSS, International Prostate Symptom Score; LUTS, lower urinary tract symptoms Corona G et al. Expert Opin Pharmacother. 2014; 15(13): 1903– 26. 53

Module 5: Nebido® clinical efficacy Long-term studies 54
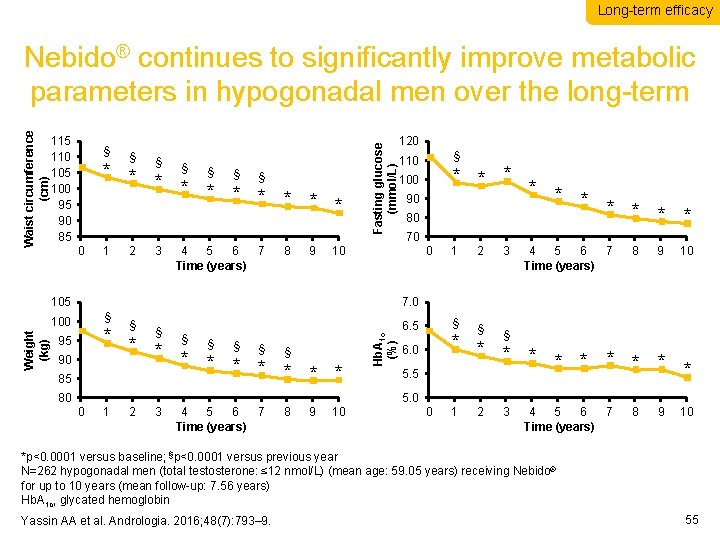
Long-term efficacy 115 110 105 100 95 90 85 § § * * * * 0 1 2 3 § 4 5 6 7 Time (years) 8 9 Fasting glucose (mmol/L) Waist circumference (cm) Nebido® continues to significantly improve metabolic parameters in hypogonadal men over the long-term § 110 * * * * * 100 90 80 70 105 1 2 3 § § 2 3 4 5 6 7 Time (years) 8 9 10 7. 0 § § § * * * * 95 90 85 80 § 6. 5 Hb. A 1 c (%) § 100 Weight (kg) 120 * * * * * 6. 0 5. 5 5. 0 0 1 2 3 4 5 6 7 Time (years) 8 9 10 0 1 4 5 6 7 Time (years) 8 9 10 *p<0. 0001 versus baseline; §p<0. 0001 versus previous year N=262 hypogonadal men (total testosterone: ≤ 12 nmol/L) (mean age: 59. 05 years) receiving Nebido® for up to 10 years (mean follow-up: 7. 56 years) Hb. A 1 c, glycated hemoglobin Yassin AA et al. Andrologia. 2016; 48(7): 793– 9. 55
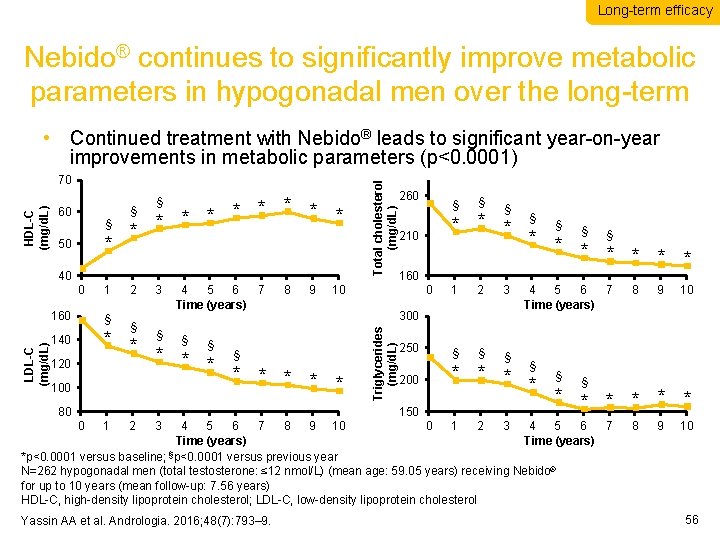
Long-term efficacy Nebido® continues to significantly improve metabolic parameters in hypogonadal men over the long-term 60 § * 50 * * * * * § § 40 0 LDL-C (mg/d. L) 160 1 § 2 3 4 5 6 7 Time (years) § 8 9 * * § § * * * * 140 120 100 80 260 § § 1 2 3 250 § § § 200 * * 160 0 300 150 0 1 2 3 § * * * § § * * * * 210 10 Triglycerides (mg/d. L) HDL-C (mg/d. L) 70 Total cholesterol (mg/d. L) • Continued treatment with Nebido® leads to significant year-on-year improvements in metabolic parameters (p<0. 0001) 4 5 6 7 Time (years) § § 9 10 § * * * 4 5 6 7 8 9 10 0 1 2 3 4 5 6 7 Time (years) § *p<0. 0001 versus baseline; p<0. 0001 versus previous year N=262 hypogonadal men (total testosterone: ≤ 12 nmol/L) (mean age: 59. 05 years) receiving Nebido® for up to 10 years (mean follow-up: 7. 56 years) HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol Yassin AA et al. Andrologia. 2016; 48(7): 793– 9. 8 8 9 10 56
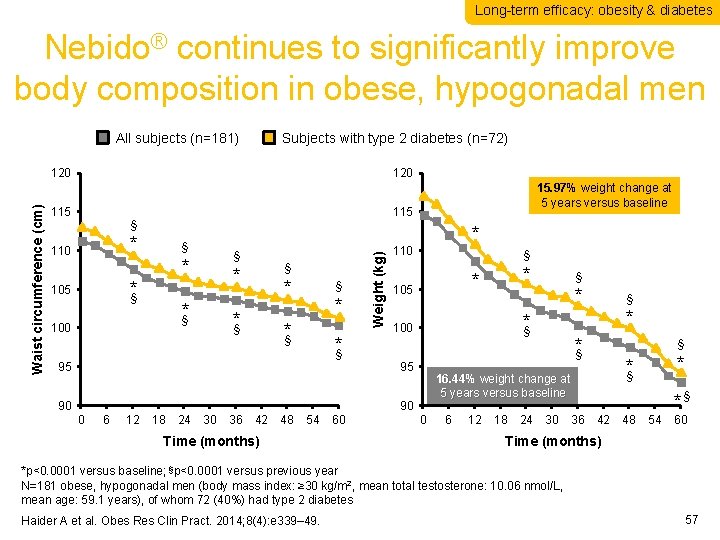
Long-term efficacy: obesity & diabetes Nebido® continues to significantly improve body composition in obese, hypogonadal men All subjects (n=181) Subjects with type 2 diabetes (n=72) 120 115 * 110 § § * *§ 105 § * *§ 100 * *§ § * *§ *§ 90 18 24 30 36 42 48 54 Time (months) 60 * * § * *§ 100 90 12 § 105 95 6 * 110 95 0 15. 97% weight change at 5 years versus baseline 115 § Weight (kg) Waist circumference (cm) 120 § * *§ § 0 6 12 18 24 30 * *§ 16. 44% weight change at 5 years versus baseline *§ 36 42 48 54 60 Time (months) *p<0. 0001 versus baseline; §p<0. 0001 versus previous year N=181 obese, hypogonadal men (body mass index: ≥ 30 kg/m 2, mean total testosterone: 10. 06 nmol/L, mean age: 59. 1 years), of whom 72 (40%) had type 2 diabetes Haider A et al. Obes Res Clin Pract. 2014; 8(4): e 339– 49. 57
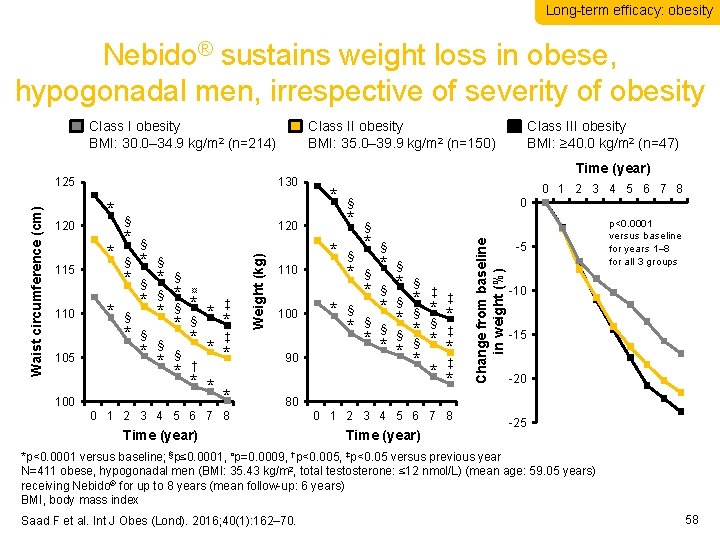
Long-term efficacy: obesity Nebido® sustains weight loss in obese, hypogonadal men, irrespective of severity of obesity 120 115 110 105 100 130 * * § * § * § § * ¤ * * ‡ § * * § * ‡ * § * * * † * * § 100 90 0 1 2 3 4 5 6 7 8 § * * 110 Class III obesity BMI: ≥ 40. 0 kg/m 2 (n=47) Time (year) 120 Weight (kg) Waist circumference (cm) 125 Class II obesity BMI: 35. 0– 39. 9 kg/m 2 (n=150) § * 0 § * § * § ‡ * * ‡ * § * § * * § § * § ‡ * * § * * ‡ * * § Change from baseline in weight (%) Class I obesity BMI: 30. 0– 34. 9 kg/m 2 (n=214) -5 p<0. 0001 versus baseline for years 1– 8 for all 3 groups -10 -15 -20 80 0 1 2 3 4 5 6 7 8 Time (year) -25 *p<0. 0001 versus baseline; §p≤ 0. 0001, ¤p=0. 0009, †p<0. 005, ‡p<0. 05 versus previous year N=411 obese, hypogonadal men (BMI: 35. 43 kg/m 2, total testosterone: ≤ 12 nmol/L) (mean age: 59. 05 years) receiving Nebido® for up to 8 years (mean follow-up: 6 years) BMI, body mass index Saad F et al. Int J Obes (Lond). 2016; 40(1): 162– 70. 58
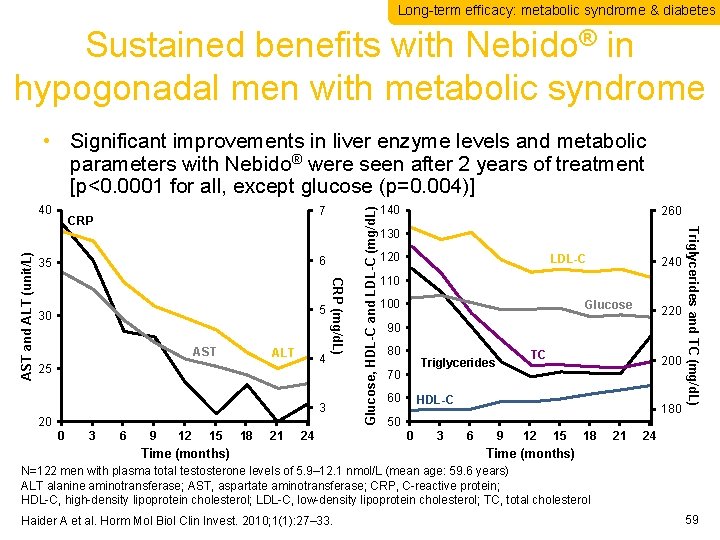
Long-term efficacy: metabolic syndrome & diabetes Sustained benefits with Nebido® in hypogonadal men with metabolic syndrome 7 6 35 5 30 AST ALT 4 25 3 20 0 3 6 9 12 15 18 21 24 Time (months) 140 260 130 120 LDL-C 240 110 100 Glucose 220 90 80 TC Triglycerides 70 60 200 HDL-C 180 50 0 3 6 9 12 15 18 21 Triglycerides and TC (mg/d. L) CRP (mg/d. L) AST and ALT (unit/L) 40 Glucose, HDL-C and LDL-C (mg/d. L) • Significant improvements in liver enzyme levels and metabolic parameters with Nebido® were seen after 2 years of treatment [p<0. 0001 for all, except glucose (p=0. 004)] 24 Time (months) N=122 men with plasma total testosterone levels of 5. 9– 12. 1 nmol/L (mean age: 59. 6 years) ALT alanine aminotransferase; AST, aspartate aminotransferase; CRP, C-reactive protein; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; TC, total cholesterol Haider A et al. Horm Mol Biol Clin Invest. 2010; 1(1): 27– 33. 59
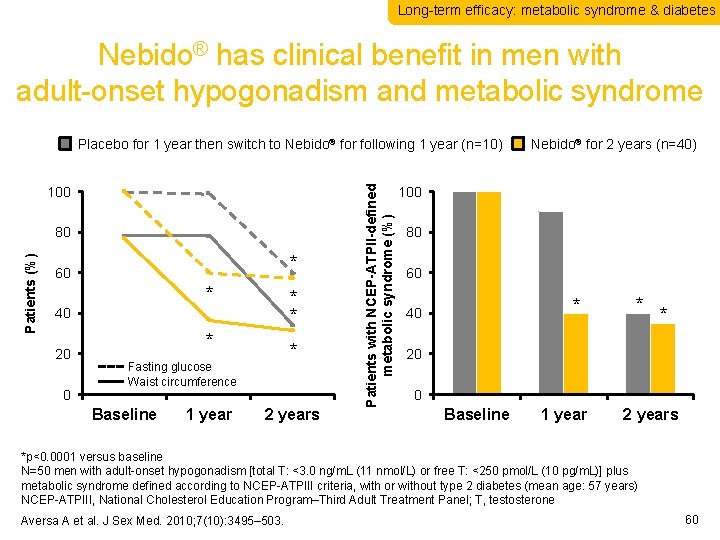
Long-term efficacy: metabolic syndrome & diabetes Nebido® has clinical benefit in men with adult-onset hypogonadism and metabolic syndrome 100 Patients (%) 80 * 60 * * * 40 20 0 * * Fasting glucose Waist circumference Baseline 1 year 2 years Patients with NCEP-ATPII-defined metabolic syndrome (%) Placebo for 1 year then switch to Nebido® for following 1 year (n=10) Nebido® for 2 years (n=40) 100 80 60 * 40 * * 20 0 Baseline 1 year 2 years *p<0. 0001 versus baseline N=50 men with adult-onset hypogonadism [total T: <3. 0 ng/m. L (11 nmol/L) or free T: <250 pmol/L (10 pg/m. L)] plus metabolic syndrome defined according to NCEP-ATPIII criteria, with or without type 2 diabetes (mean age: 57 years) NCEP-ATPIII, National Cholesterol Education Program–Third Adult Treatment Panel; T, testosterone Aversa A et al. J Sex Med. 2010; 7(10): 3495– 503. 60
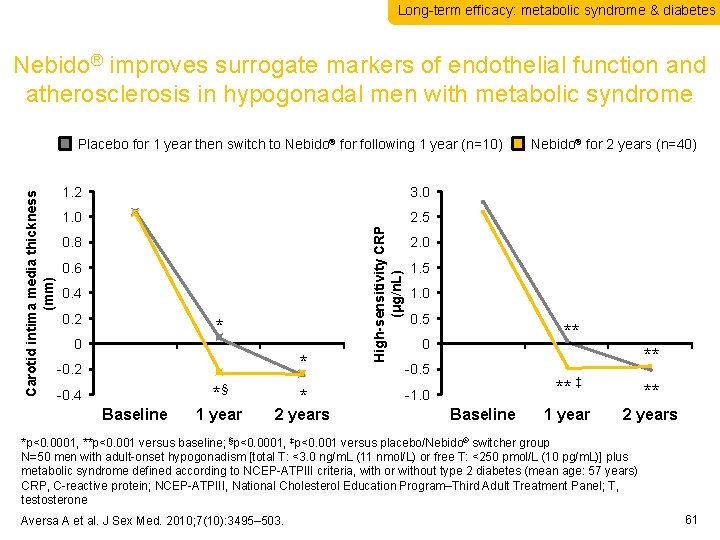
Long-term efficacy: metabolic syndrome & diabetes Nebido® improves surrogate markers of endothelial function and atherosclerosis in hypogonadal men with metabolic syndrome 1. 2 3. 0 1. 0 2. 5 0. 8 0. 6 0. 4 0. 2 * 0. 0 * -0. 2 -0. 4 Baseline * § 1 year * 2 years High-sensitivity CRP (μg/n. L) Carotid intima media thickness (mm) Placebo for 1 year then switch to Nebido® for following 1 year (n=10) Nebido® for 2 years (n=40) 2. 0 1. 5 1. 0 0. 5 ** 0. 0 ** -0. 5 -1. 0 Baseline ** ‡ ** 1 year 2 years *p<0. 0001, **p<0. 001 versus baseline; §p<0. 0001, ‡p<0. 001 versus placebo/Nebido® switcher group N=50 men with adult-onset hypogonadism [total T: <3. 0 ng/m. L (11 nmol/L) or free T: <250 pmol/L (10 pg/m. L)] plus metabolic syndrome defined according to NCEP-ATPIII criteria, with or without type 2 diabetes (mean age: 57 years) CRP, C-reactive protein; NCEP-ATPIII, National Cholesterol Education Program–Third Adult Treatment Panel; T, testosterone Aversa A et al. J Sex Med. 2010; 7(10): 3495– 503. 61
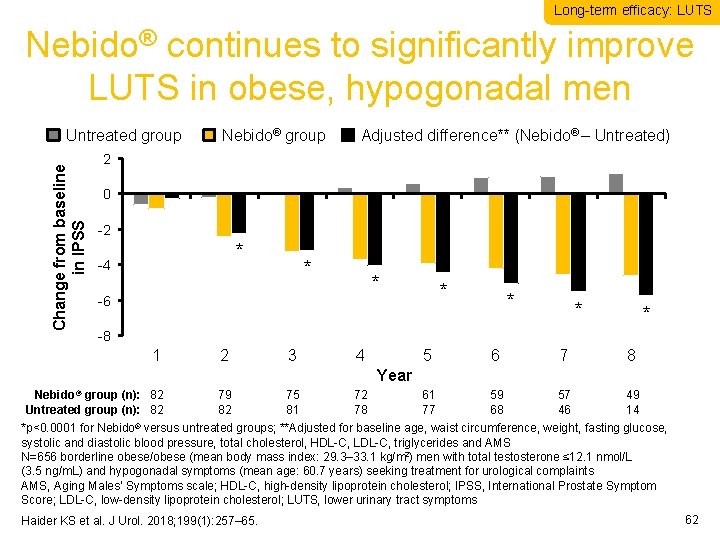
Long-term efficacy: LUTS Nebido® continues to significantly improve LUTS in obese, hypogonadal men Change from baseline in IPSS Untreated group Nebido® group Adjusted difference** (Nebido® – Untreated) 2 0 -2 * -4 * * -6 * * -8 1 2 3 4 5 6 7 8 61 77 59 68 57 46 49 14 Year Nebido® group (n): 82 Untreated group (n): 82 79 82 75 81 72 78 *p<0. 0001 for Nebido® versus untreated groups; **Adjusted for baseline age, waist circumference, weight, fasting glucose, systolic and diastolic blood pressure, total cholesterol, HDL-C, LDL-C, triglycerides and AMS N=656 borderline obese/obese (mean body mass index: 29. 3– 33. 1 kg/m 2) men with total testosterone ≤ 12. 1 nmol/L (3. 5 ng/m. L) and hypogonadal symptoms (mean age: 60. 7 years) seeking treatment for urological complaints AMS, Aging Males’ Symptoms scale; HDL-C, high-density lipoprotein cholesterol; IPSS, International Prostate Symptom Score; LDL-C, low-density lipoprotein cholesterol; LUTS, lower urinary tract symptoms Haider KS et al. J Urol. 2018; 199(1): 257– 65. 62
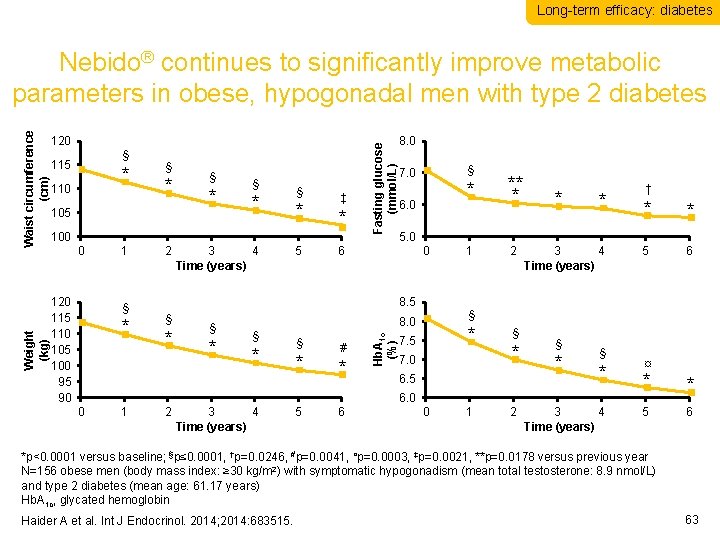
Long-term efficacy: diabetes § 115 * 110 § * 105 § * § * ‡ * 100 0 120 115 110 105 100 95 90 1 § * 2 3 4 Time (years) 5 Fasting glucose (mmol/L) 120 8. 0 § 7. 0 6. 0 0 8. 5 § * * § * # * ** * 1 2 * * 3 4 Time (years) * 7. 5 7. 0 § * 6. 5 § * 6. 0 0 1 2 3 4 Time (years) 5 6 † * * 5 6 § 8. 0 § * 5. 0 6 Hb. A 1 c (%) Weight (kg) Waist circumference (cm) Nebido® continues to significantly improve metabolic parameters in obese, hypogonadal men with type 2 diabetes 0 1 2 3 4 Time (years) ¤ * * 5 6 *p<0. 0001 versus baseline; §p≤ 0. 0001, †p=0. 0246, #p=0. 0041, ¤p=0. 0003, ‡p=0. 0021, **p=0. 0178 versus previous year N=156 obese men (body mass index: ≥ 30 kg/m 2) with symptomatic hypogonadism (mean total testosterone: 8. 9 nmol/L) and type 2 diabetes (mean age: 61. 17 years) Hb. A 1 c, glycated hemoglobin Haider A et al. Int J Endocrinol. 2014; 2014: 683515. 63
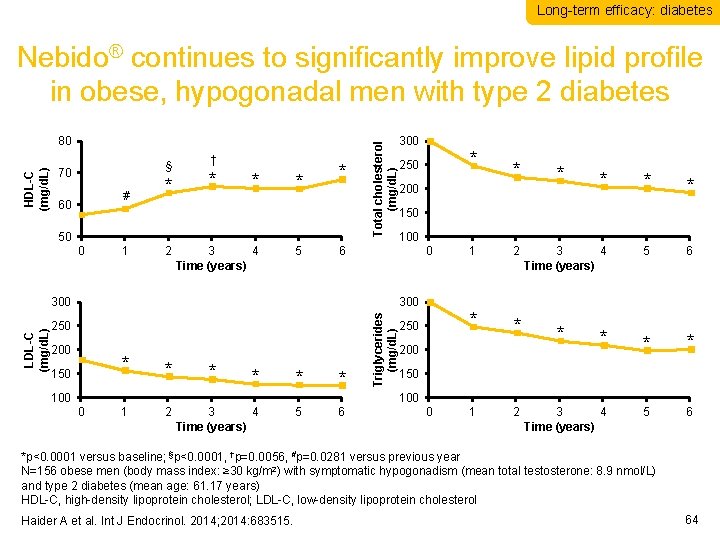
Long-term efficacy: diabetes HDL-C (mg/d. L) 80 § 70 # 60 * † * * 50 0 1 2 3 4 Time (years) 5 Total cholesterol (mg/d. L) Nebido® continues to significantly improve lipid profile in obese, hypogonadal men with type 2 diabetes * 250 * 150 * * * 100 * * * 3 4 Time (years) 5 6 100 0 Triglycerides (mg/d. L) 200 * 150 300 250 * 200 6 300 LDL-C (mg/d. L) 300 1 * 250 2 * 200 * 150 100 0 1 2 3 4 Time (years) 5 6 0 1 2 *p<0. 0001 versus baseline; §p<0. 0001, †p=0. 0056, #p=0. 0281 versus previous year N=156 obese men (body mass index: ≥ 30 kg/m 2) with symptomatic hypogonadism (mean total testosterone: 8. 9 nmol/L) and type 2 diabetes (mean age: 61. 17 years) HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol Haider A et al. Int J Endocrinol. 2014; 2014: 683515. 64
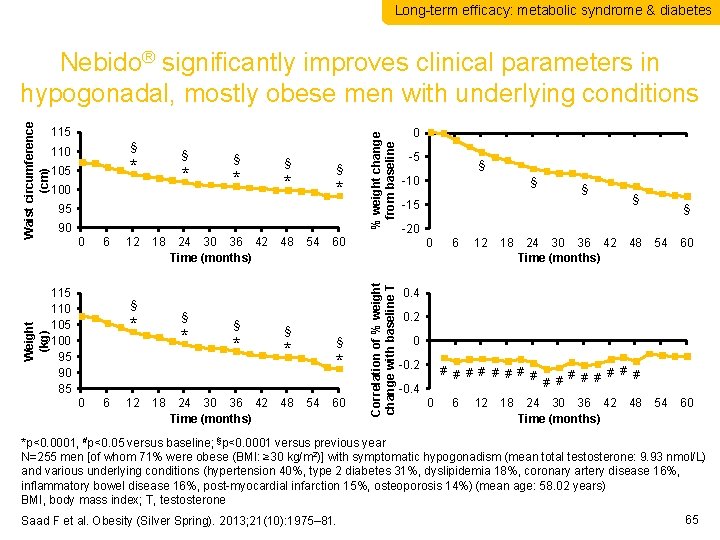
Long-term efficacy: metabolic syndrome & diabetes § 110 § * 105 * 100 § * § § * * 95 90 0 6 115 110 105 100 95 90 85 12 18 § § * 0 6 12 24 30 36 42 Time (months) * 18 § * 24 30 36 42 Time (months) 48 54 60 § § * 48 * 54 % weight change from baseline 115 60 0 -5 § -10 § § -15 § § -20 0 Correlation of % weight change with baseline T Weight (kg) Waist circumference (cm) Nebido® significantly improves clinical parameters in hypogonadal, mostly obese men with underlying conditions 6 12 18 24 30 36 42 Time (months) 48 54 60 0. 4 0. 2 0 -0. 2 # # # # -0. 4 0 6 12 18 # # # # 24 30 36 42 Time (months) 48 *p<0. 0001, #p<0. 05 versus baseline; §p<0. 0001 versus previous year N=255 men [of whom 71% were obese (BMI: ≥ 30 kg/m 2)] with symptomatic hypogonadism (mean total testosterone: 9. 93 nmol/L) and various underlying conditions (hypertension 40%, type 2 diabetes 31%, dyslipidemia 18%, coronary artery disease 16%, inflammatory bowel disease 16%, post-myocardial infarction 15%, osteoporosis 14%) (mean age: 58. 02 years) BMI, body mass index; T, testosterone Saad F et al. Obesity (Silver Spring). 2013; 21(10): 1975– 81. 65
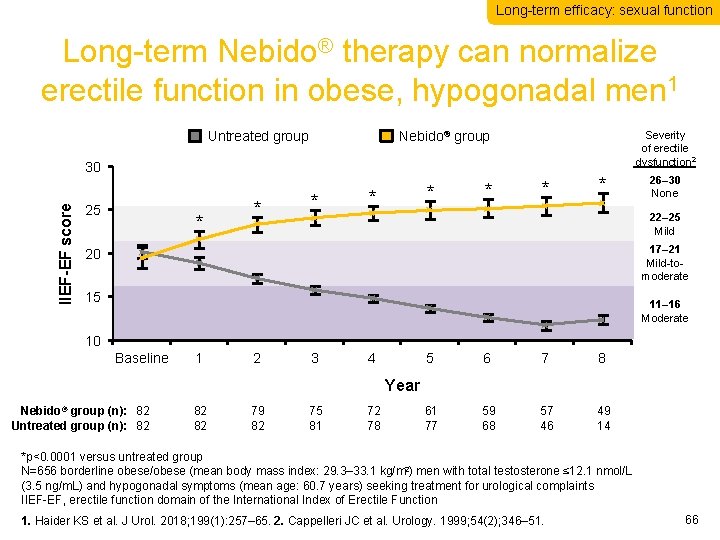
Long-term efficacy: sexual function Long-term Nebido® therapy can normalize erectile function in obese, hypogonadal men 1 Untreated group Nebido® group Severity of erectile dysfunction 2 IIEF-EF score 30 25 * * * * 26– 30 None 22– 25 Mild 17– 21 Mild-tomoderate 20 15 11– 16 Moderate 10 Baseline 1 2 3 4 5 6 7 8 61 77 59 68 57 46 49 14 Year Nebido® group (n): 82 Untreated group (n): 82 82 82 79 82 75 81 72 78 *p<0. 0001 versus untreated group N=656 borderline obese/obese (mean body mass index: 29. 3– 33. 1 kg/m 2) men with total testosterone ≤ 12. 1 nmol/L (3. 5 ng/m. L) and hypogonadal symptoms (mean age: 60. 7 years) seeking treatment for urological complaints IIEF-EF, erectile function domain of the International Index of Erectile Function 1. Haider KS et al. J Urol. 2018; 199(1): 257– 65. 2. Cappelleri JC et al. Urology. 1999; 54(2); 346– 51. 66
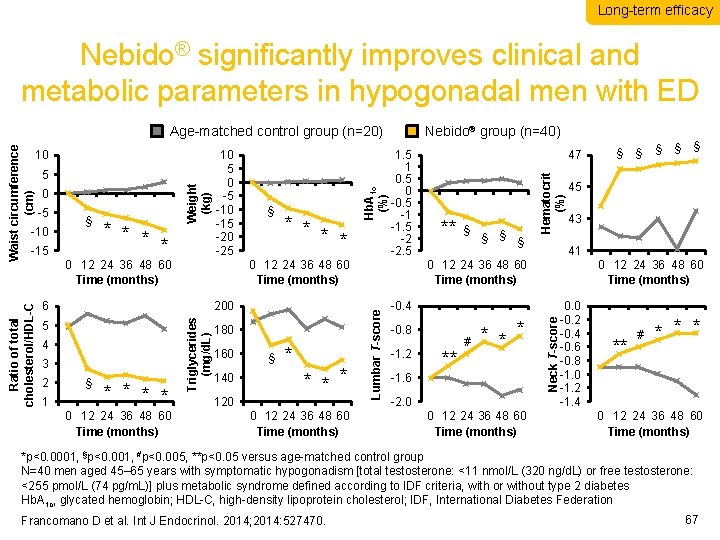
Long-term efficacy Nebido® significantly improves clinical and metabolic parameters in hypogonadal men with ED -15 * * 0 12 24 36 48 60 Time (months) 3 § * * 0 12 24 36 48 60 Time (months) Triglycerides (mg/d. L) 4 1 * * 200 5 2 § 1. 5 1 0. 5 0 -0. 5 -1 -1. 5 -2 -2. 5 180 160 140 120 § * * 0 12 24 36 48 60 Time (months) 47 ** § § Hematocrit (%) -10 § Nebido® group (n=40) -1. 2 ** * -1. 6 -2. 0 0 12 24 36 48 60 Time (months) § § § 43 0 12 24 36 48 60 Time (months) -0. 4 # § 45 0 12 24 36 48 60 Time (months) -0. 8 § 41 Neck T-score -5 Weight (kg) 0 Hb. A 1 c (%) 6 5 10 5 0 -5 -10 -15 -20 -25 Lumbar T-score Waist circumference (cm) 10 Ratio of total cholesterol/HDL-C Age-matched control group (n=20) 0. 0 -0. 2 -0. 4 -0. 6 -0. 8 -1. 0 -1. 2 -1. 4 ** # * * * 0 12 24 36 48 60 Time (months) *p<0. 0001, §p<0. 001, #p<0. 005, **p<0. 05 versus age-matched control group N=40 men aged 45– 65 years with symptomatic hypogonadism [total testosterone: <11 nmol/L (320 ng/d. L) or free testosterone: <255 pmol/L (74 pg/m. L)] plus metabolic syndrome defined according to IDF criteria, with or without type 2 diabetes Hb. A 1 c, glycated hemoglobin; HDL-C, high-density lipoprotein cholesterol; IDF, International Diabetes Federation Francomano D et al. Int J Endocrinol. 2014; 2014: 527470. 67
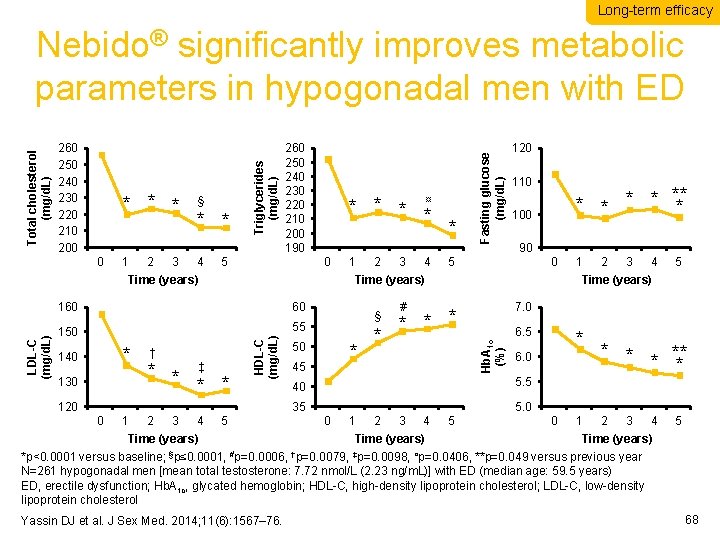
Long-term efficacy 0 1 2 3 § * * 4 5 * *¤ 0 160 60 150 55 * 140 130 † * * ‡ 120 0 1 2 3 4 120 110 * * ** * 100 90 5 0 Time (years) HDL-C (mg/d. L) LDL-C (mg/d. L) Time (years) 1 * Fasting glucose (mg/d. L) * * * 260 250 240 230 220 210 200 190 5 § 50 * 45 * # * * * 6. 5 35 5. 0 2 3 4 5 * 6. 0 5. 5 1 2 3 4 5 7. 0 40 0 1 Time (years) Hb. A 1 c (%) 260 250 240 230 220 210 200 Triglycerides (mg/d. L) Total cholesterol (mg/d. L) Nebido® significantly improves metabolic parameters in hypogonadal men with ED 0 1 * ** * 2 3 4 5 Time (years) *p<0. 0001 versus baseline; §p≤ 0. 0001, #p=0. 0006, †p=0. 0079, ‡p=0. 0098, ¤p=0. 0406, **p=0. 049 versus previous year N=261 hypogonadal men [mean total testosterone: 7. 72 nmol/L (2. 23 ng/m. L)] with ED (median age: 59. 5 years) ED, erectile dysfunction; Hb. A 1 c, glycated hemoglobin; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol Yassin DJ et al. J Sex Med. 2014; 11(6): 1567– 76. 68
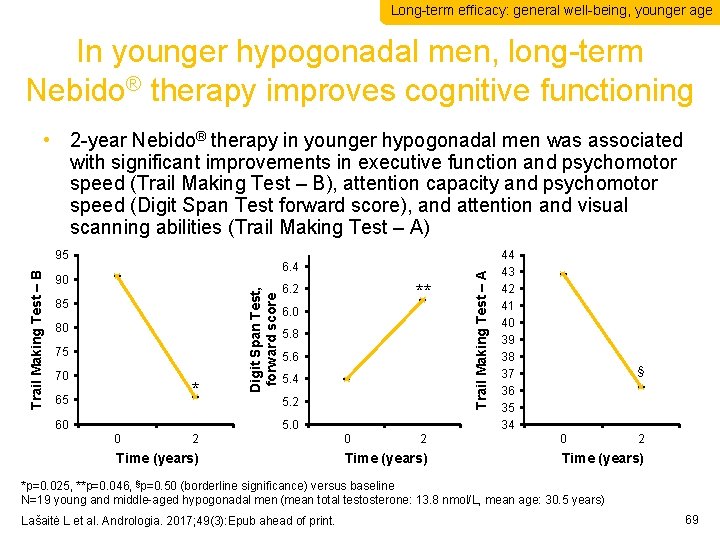
Long-term efficacy: general well-being, younger age In younger hypogonadal men, long-term Nebido® therapy improves cognitive functioning 6. 4 90 85 80 75 70 * 65 60 Digit Span Test, forward score Trail Making Test – B 95 ** 6. 2 6. 0 5. 8 5. 6 5. 4 5. 2 5. 0 0 2 Time (years) Trail Making Test – A • 2 -year Nebido® therapy in younger hypogonadal men was associated with significant improvements in executive function and psychomotor speed (Trail Making Test – B), attention capacity and psychomotor speed (Digit Span Test forward score), and attention and visual scanning abilities (Trail Making Test – A) 44 43 42 41 40 39 38 37 36 35 34 § 0 2 Time (years) *p=0. 025, **p=0. 046, §p=0. 50 (borderline significance) versus baseline N=19 young and middle-aged hypogonadal men (mean total testosterone: 13. 8 nmol/L, mean age: 30. 5 years) Lašaitė L et al. Andrologia. 2017; 49(3): Epub ahead of print. 69
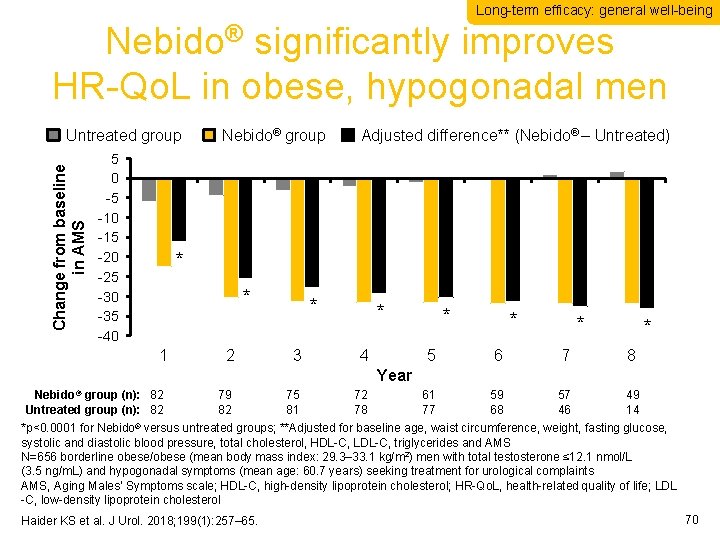
Long-term efficacy: general well-being Nebido® significantly improves HR-Qo. L in obese, hypogonadal men Change from baseline in AMS Untreated group 5 0 -5 -10 -15 -20 -25 -30 -35 -40 Nebido® group Adjusted difference** (Nebido® – Untreated) * * 1 2 * 3 * 4 * * 5 6 7 8 61 77 59 68 57 46 49 14 Year Nebido® group (n): 82 Untreated group (n): 82 79 82 75 81 72 78 *p<0. 0001 for Nebido® versus untreated groups; **Adjusted for baseline age, waist circumference, weight, fasting glucose, systolic and diastolic blood pressure, total cholesterol, HDL-C, LDL-C, triglycerides and AMS N=656 borderline obese/obese (mean body mass index: 29. 3– 33. 1 kg/m 2) men with total testosterone ≤ 12. 1 nmol/L (3. 5 ng/m. L) and hypogonadal symptoms (mean age: 60. 7 years) seeking treatment for urological complaints AMS, Aging Males’ Symptoms scale; HDL-C, high-density lipoprotein cholesterol; HR-Qo. L, health-related quality of life; LDL -C, low-density lipoprotein cholesterol Haider KS et al. J Urol. 2018; 199(1): 257– 65. 70
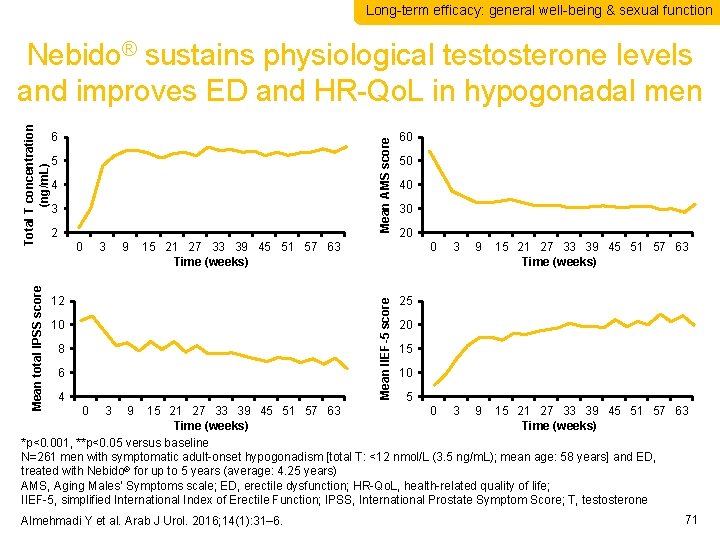
Long-term efficacy: general well-being & sexual function Mean AMS score 6 5 4 3 2 0 3 9 60 50 40 30 20 15 21 27 33 39 45 51 57 63 Time (weeks) 12 0 Mean IIEF-5 score Mean total IPSS score Total T concentration (ng/m. L) Nebido® sustains physiological testosterone levels and improves ED and HR-Qo. L in hypogonadal men 10 8 6 4 3 9 15 21 27 33 39 45 51 57 63 Time (weeks) 25 20 15 10 5 15 21 27 33 39 45 51 57 63 0 3 9 15 21 27 33 39 45 51 57 63 Time (weeks) *p<0. 001, **p<0. 05 versus baseline N=261 men with symptomatic adult-onset hypogonadism [total T: <12 nmol/L (3. 5 ng/m. L); mean age: 58 years] and ED, treated with Nebido® for up to 5 years (average: 4. 25 years) AMS, Aging Males’ Symptoms scale; ED, erectile dysfunction; HR-Qo. L, health-related quality of life; IIEF-5, simplified International Index of Erectile Function; IPSS, International Prostate Symptom Score; T, testosterone 0 3 9 Almehmadi Y et al. Arab J Urol. 2016; 14(1): 31– 6. 71
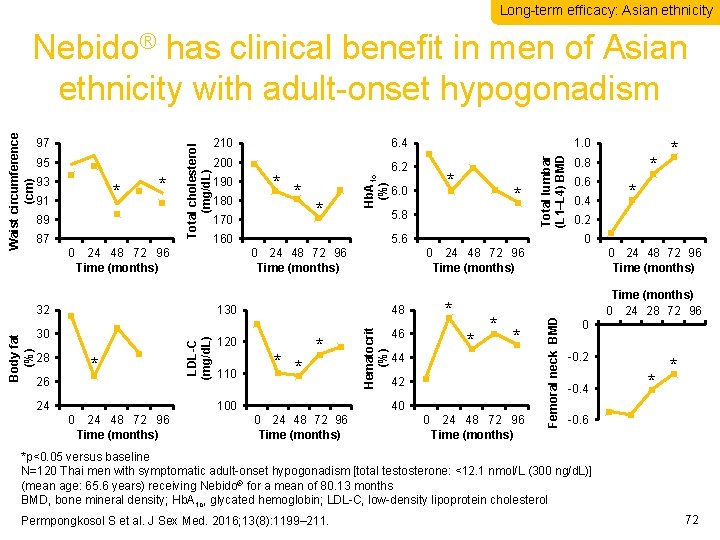
Long-term efficacy: Asian ethnicity * 91 * 89 87 0 190 180 * * 170 6. 2 * 0 24 48 72 96 Time (months) 26 24 110 * * * 100 0 24 48 72 96 Time (months) 46 * 44 * * 42 40 0 24 48 72 96 Time (months) 0. 6 0. 4 * * 0. 2 0 0 24 48 72 96 Time (months) * 48 120 * 0. 8 0. 0 0 Hematocrit (%) * LDL-C (mg/d. L) 28 * 5. 6 130 30 * 6. 0 5. 8 160 24 48 72 96 Time (months) 32 Body fat (%) 200 1. 0 Total lumbar (L 1–L 4) BMD 93 6. 4 24 48 72 96 Time (months) Femoral neck BMD 95 210 Hb. A 1 c (%) 97 Total cholesterol (mg/d. L) Waist circumference (cm) Nebido® has clinical benefit in men of Asian ethnicity with adult-onset hypogonadism Time (months) 0 24 28 72 96 0 -0. 2 -0. 4 * * -0. 6 *p<0. 05 versus baseline N=120 Thai men with symptomatic adult-onset hypogonadism [total testosterone: <12. 1 nmol/L (300 ng/d. L)] (mean age: 65. 6 years) receiving Nebido® for a mean of 80. 13 months BMD, bone mineral density; Hb. A 1 c, glycated hemoglobin; LDL-C, low-density lipoprotein cholesterol Permpongkosol S et al. J Sex Med. 2016; 13(8): 1199– 211. 72
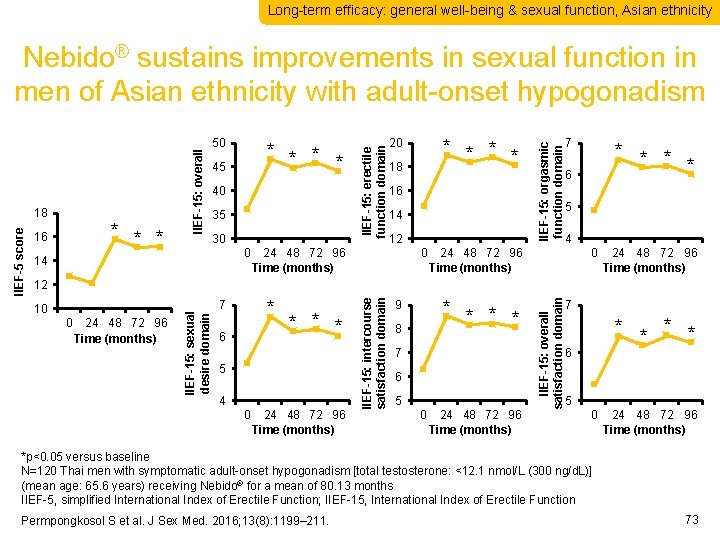
Long-term efficacy: general well-being & sexual function, Asian ethnicity * * 45 40 35 30 0 14 * * 20 18 16 14 12 24 48 72 96 Time (months) 0 IIEF-15: orgasmic function domain 50 IIEF-15: erectile function domain * * * 16 7 * * 6 5 4 24 48 72 96 Time (months) 0 24 48 72 96 Time (months) * 7 6 * * * 5 4 0 24 48 72 96 Time (months) * * 9 8 7 6 5 0 24 48 72 96 Time (months) IIEF-15: overall satisfaction domain 10 IIEF-15: intercourse satisfaction domain 12 IIEF-15: sexual desire domain IIEF-5 score 18 IIEF-15: overall Nebido® sustains improvements in sexual function in men of Asian ethnicity with adult-onset hypogonadism 7 * * 6 5 0 24 48 72 96 Time (months) *p<0. 05 versus baseline N=120 Thai men with symptomatic adult-onset hypogonadism [total testosterone: <12. 1 nmol/L (300 ng/d. L)] (mean age: 65. 6 years) receiving Nebido® for a mean of 80. 13 months IIEF-5, simplified International Index of Erectile Function; IIEF-15, International Index of Erectile Function Permpongkosol S et al. J Sex Med. 2016; 13(8): 1199– 211. 73

Module 5: Nebido® clinical efficacy Cardiovascular risk and mortality 74
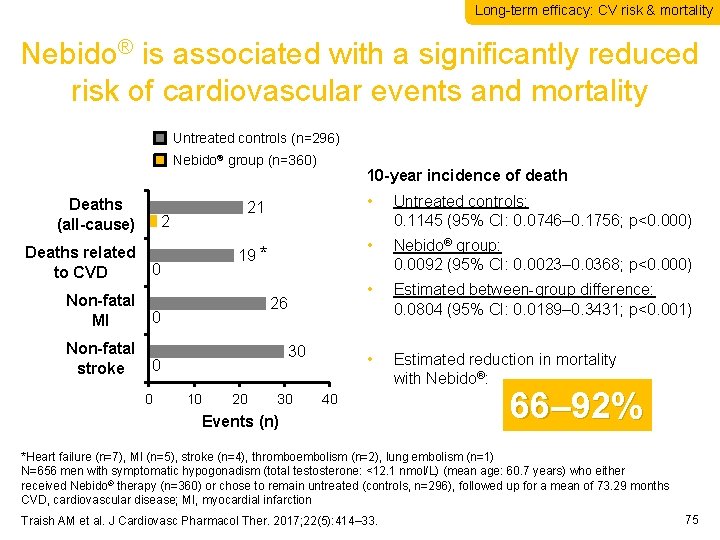
Long-term efficacy: CV risk & mortality Nebido® is associated with a significantly reduced risk of cardiovascular events and mortality Untreated controls (n=296) Nebido® group (n=360) Deaths (all-cause) 2 Deaths related to CVD 0 Non-fatal MI 0 Non-fatal stroke 0 0 10 -year incidence of death 21 • Untreated controls: 0. 1145 (95% CI: 0. 0746– 0. 1756; p<0. 000) 19 * • Nebido® group: 0. 0092 (95% CI: 0. 0023– 0. 0368; p<0. 000) • Estimated between-group difference: 0. 0804 (95% CI: 0. 0189– 0. 3431; p<0. 001) • Estimated reduction in mortality with Nebido®: 26 30 10 20 30 40 Events (n) 66– 92% *Heart failure (n=7), MI (n=5), stroke (n=4), thromboembolism (n=2), lung embolism (n=1) N=656 men with symptomatic hypogonadism (total testosterone: <12. 1 nmol/L) (mean age: 60. 7 years) who either received Nebido® therapy (n=360) or chose to remain untreated (controls, n=296), followed up for a mean of 73. 29 months CVD, cardiovascular disease; MI, myocardial infarction Traish AM et al. J Cardiovasc Pharmacol Ther. 2017; 22(5): 414– 33. 75
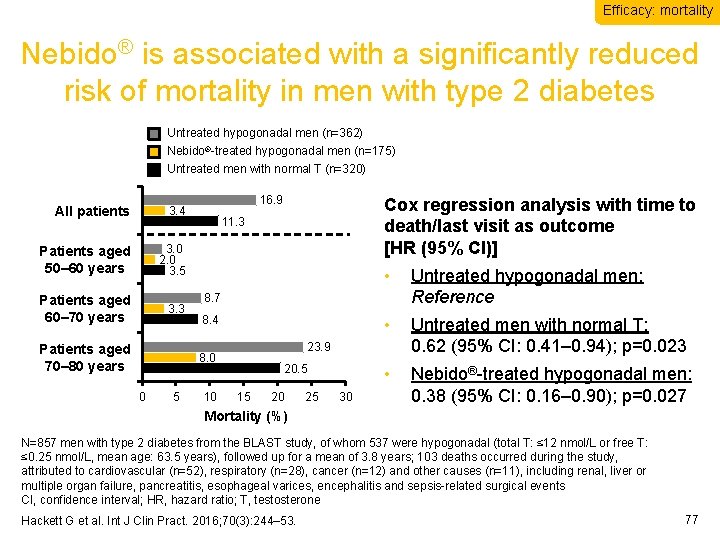
Efficacy: mortality Nebido® is associated with a significantly reduced risk of mortality in men with type 2 diabetes Untreated hypogonadal men (n=362) Nebido®-treated hypogonadal men (n=175) Untreated men with normal T (n=320) All patients 3. 4 Patients aged 50– 60 years 3. 0 2. 0 3. 5 Patients aged 60– 70 years 3. 3 Patients aged 70– 80 years 16. 9 11. 3 5 • Untreated hypogonadal men: Reference • Untreated men with normal T: 0. 62 (95% CI: 0. 41– 0. 94); p=0. 023 • Nebido®-treated hypogonadal men: 0. 38 (95% CI: 0. 16– 0. 90); p=0. 027 8. 4 23. 9 8. 0 0 Cox regression analysis with time to death/last visit as outcome [HR (95% CI)] 10 20. 5 15 20 25 30 Mortality (%) N=857 men with type 2 diabetes from the BLAST study, of whom 537 were hypogonadal (total T: ≤ 12 nmol/L or free T: ≤ 0. 25 nmol/L, mean age: 63. 5 years), followed up for a mean of 3. 8 years; 103 deaths occurred during the study, attributed to cardiovascular (n=52), respiratory (n=28), cancer (n=12) and other causes (n=11), including renal, liver or multiple organ failure, pancreatitis, esophageal varices, encephalitis and sepsis-related surgical events CI, confidence interval; HR, hazard ratio; T, testosterone Hackett G et al. Int J Clin Pract. 2016; 70(3): 244– 53. 77

Module 5: Nebido® clinical efficacy Interruption of Nebido® therapy 78
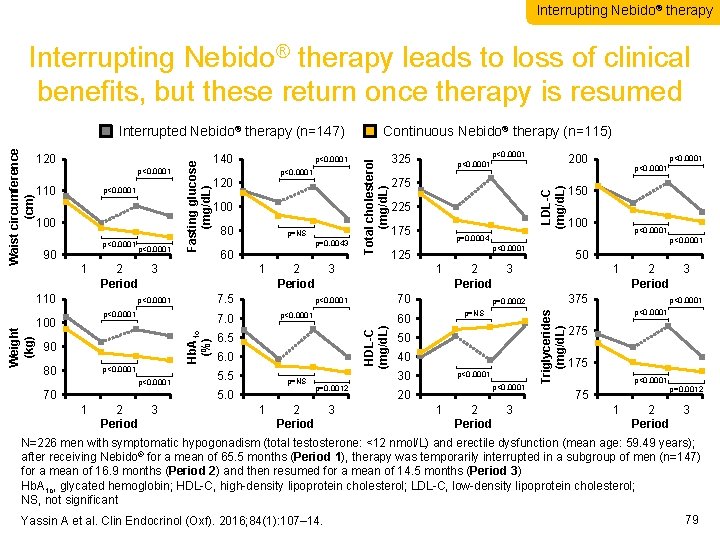
Interrupting Nebido® therapy leads to loss of clinical benefits, but these return once therapy is resumed 100 90 p<0. 0001 100 80 60 p<0. 0001 70 175 p=0. 0012 1 1 2 2 3 3 4 Period 100 p=0. 0004 125 50 1 1 2 2 3 3 4 Period 375 p=0. 0002 p=NS 60 50 40 30 p<0. 0001 20 1 1 2 2 3 Period 3 4 p<0. 0001 70 p<0. 0001 p=NS 225 p<0. 0001 150 1 1 2 2 3 3 4 Period p<0. 0001 6. 0 5. 5 275 3 4 6. 5 5. 0 1 1 2 2 3 3 4 Period 1 2 2 3 Period 7. 5 p<0. 0001 90 80 1 Hb. A 1 c (%) 100 p=NS p=0. 0043 1 1 2 2 3 3 4 Period 110 Weight (kg) p<0. 0001 120 200 p<0. 0001 LDL-C (mg/d. L) p<0. 0001 325 Triglycerides (mg/d. L) 110 p<0. 0001 Total cholesterol (mg/d. L) p<0. 0001 140 Continuous Nebido® therapy (n=115) HDL-C (mg/d. L) 120 Fasting glucose (mg/d. L) Waist circumference (cm) Interrupted Nebido® therapy (n=147) p<0. 0001 275 175 p<0. 0001 75 p=0. 0012 1 1 2 2 3 3 4 Period N=226 men with symptomatic hypogonadism (total testosterone: <12 nmol/L) and erectile dysfunction (mean age: 59. 49 years); after receiving Nebido® for a mean of 65. 5 months (Period 1), therapy was temporarily interrupted in a subgroup of men (n=147) for a mean of 16. 9 months (Period 2) and then resumed for a mean of 14. 5 months (Period 3) Hb. A 1 c, glycated hemoglobin; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; NS, not significant Yassin A et al. Clin Endocrinol (Oxf). 2016; 84(1): 107– 14. 79
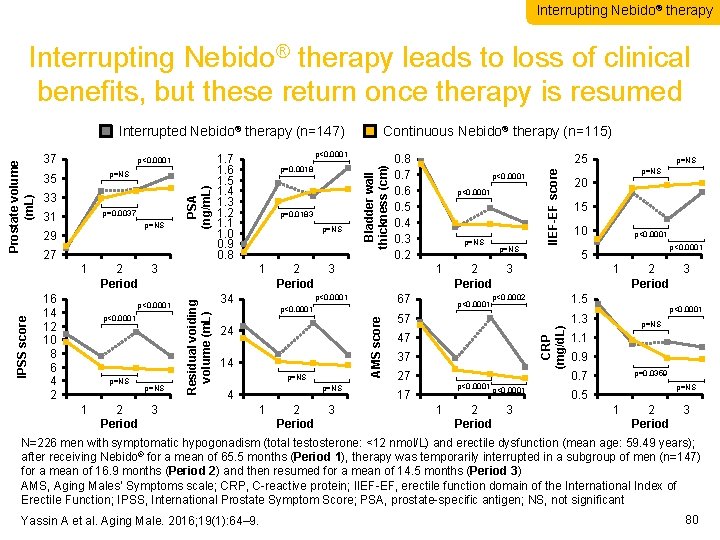
Interrupting Nebido® therapy leads to loss of clinical benefits, but these return once therapy is resumed 31 p=0. 0037 p=NS 29 27 16 14 12 10 8 6 4 2 p<0. 0001 p=NS 1 1 2 2 3 3 4 Period Residual voiding volume (m. L) IPSS score 1 1 2 2 3 3 4 Period p=0. 0183 0. 8 0. 7 0. 6 0. 5 0. 4 0. 3 0. 2 34 p=NS 4 1 1 2 2 3 3 4 Period p<0. 0001 p=NS 67 p<0. 0001 14 p<0. 0001 p=NS p<0. 0001 20 15 10 47 37 27 p<0. 0001 17 1 p<0. 0001 1 2 2 3 Period 3 4 p<0. 0001 5 1 1 2 2 3 Period 1. 5 p<0. 0002 57 p=NS 1 1 2 2 3 3 4 Period p<0. 0001 24 25 IIEF-EF score 33 p=0. 0018 CRP (mg/d. L) p=NS p<0. 0001 1. 7 1. 6 1. 5 1. 4 1. 3 1. 2 1. 1 1. 0 0. 9 0. 8 Bladder wall thickness (cm) 35 p<0. 0001 Continuous Nebido® therapy (n=115) AMS score 37 PSA (ng/m. L) Prostate volume (m. L) Interrupted Nebido® therapy (n=147) 1. 3 3 4 p<0. 0001 p=NS 1. 1 0. 9 0. 7 p=0. 0359 p=NS 0. 5 1 1 2 2 3 Period 3 4 N=226 men with symptomatic hypogonadism (total testosterone: <12 nmol/L) and erectile dysfunction (mean age: 59. 49 years); after receiving Nebido® for a mean of 65. 5 months (Period 1), therapy was temporarily interrupted in a subgroup of men (n=147) for a mean of 16. 9 months (Period 2) and then resumed for a mean of 14. 5 months (Period 3) AMS, Aging Males’ Symptoms scale; CRP, C-reactive protein; IIEF-EF, erectile function domain of the International Index of Erectile Function; IPSS, International Prostate Symptom Score; PSA, prostate-specific antigen; NS, not significant Yassin A et al. Aging Male. 2016; 19(1): 64– 9. 80

Summary • Nebido® is efficacious for the treatment of male hypogonadism alone or in the presence of underlying conditions, such as metabolic syndrome, type 2 diabetes, dyslipidemia and ED • In hypogonadal men, TTh with Nebido®: – Has a favorable effect on body composition by significantly increasing muscle mass and decreasing fat mass – Significantly improves: • Muscle strength • LUTS • Bone mineral density • Markers of cardiovascular disease • Dyslipidemia • Parameters of sexual function – Can attenuate anemia – Exerts a positive effect on mood, thus improving self-confidence and activity, and reduces fatigue and feelings of exhaustion ED, erectile dysfunction; LUTS, lower urinary tract symptoms; TTh, testosterone therapy 81

Summary (2) • In hypogonadal men with obesity, TTh with Nebido®: – Promotes and sustains weight loss, and reductions in waist circumference – Significantly improves clinical and metabolic parameters, glycemic control, dyslipidemia, anemia, LUTS, sexual function and HR-Qo. L • In hypogonadal men with diabetes, TTh with Nebido®: – Significantly improves glycemic control and insulin resistance, clinical and metabolic parameters, and sexual function – Is associated with a significantly reduced risk of cardiovascular events and mortality – May reduce or improve disease-related complications HR-Qo. L, health-related quality of life; LUTS, lower urinary tract symptoms; TTh, testosterone therapy 82

Summary (3) • Long-term TTh with Nebido® for hypogonadism: – Is associated with continuing, significant reductions in body weight, body mass index and waist circumference – Is associated with continuing, significant improvements in lipid profile and glycemic control – Is associated with continuing, significant improvements in LUTS – Can restore erectile function to normal – Significantly improves surrogate markers of endothelial function and atherosclerosis – Significantly improves HR-Qo. L, health-related quality of life; LUTS, lower urinary tract symptoms; TTh, testosterone therapy 83
- Slides: 82