Module 5 Malaria Microscopy Examination and Species Identification




































- Slides: 53

Module 5 Malaria Microscopy: Examination and Species Identification

Learning Objectives At the end of this module the trainee should be able to: – Distinguish and name properly the stage and species of malaria parasite. – Recognize artifacts that may be mistaken for malaria parasites. 2

Content Outline – Microscopy – Required materials – Systematic approach to examine thick and thin blood films – Recognition of malaria parasite species and artifacts – Malaria parasite counting methods 3

Malaria Microscopy • Is the gold standard method for detecting malaria parasites in blood sample. • Requires trained lab personnel and equipment. • Examination made using Stained thick and thin blood films. – (Compliance with the SOPs, quality of rgt. , technical ability and type of technique used) 4

Examining blood film • Malaria parasites are identified by microscopic examination of thick and thin blood films stained with Giemsa stain. • Both thick and thin films should initially be examined with 10 x and 40 x to avoid missing large parasites such as microfilariae and trypanosomes. 5

Required materials, reagents and equipments Materials and Equipments – Patient Register – Pen – Lens paper – Immersion oil – Lens cleaning solution – Tissue paper – Binocular microscope – Tally counter – Stained blood film – Slide boxes 6

Approach to examination of thick film • Thick films are performed to – To detect parasites – To measure parasite density • A slide can be pronounced negative only after examination of at least 100 fields in thick film. • If malaria parasites is found a further 100 fields should be examined to confirm the presence of a mixed infection. 7

Determination of "No Parasites Found" (NPF): • At least 100 fields should be examined before a thick film is reported as negative. – In nonimmune patients, symptomatic malaria can occur at lower parasite densities, and screening more fields (e. g. , 200, 300, or even the whole film) is recommended. 8

Method 1. Place the stained slide on the mechanical stage. 2. Scan the entire film at a low magnification. 3. Examine the film using the 100×. 9

Method. . 4. Select an area that is well-stained, free of stain precipitate, and well-populated WBCs (10 -20 WBCs/field). 5. Move the blood film following the pattern shown in the diagram. 6. If you see parasites, make a tentative species determination on the thick film and then examine thin film to confirm the species present. 10

Examining the thin film • Thin films are performed – Species identification – Quantification e. g. Percentage of infected red cell – Useful in high parasitaemia. – Difficulty in examination of thick film. • Since it takes more time( ~10 x) as compared to examination of a thin film, routine examination of thin films is not recommended. 11

Method 1. Place a drop of immersion oil on the edge of the middle of the film. 2. Scan using low magnification. if this has not been done on the thick films. 3. Carefully examine the film using the 100×. – If doubtful diagnosis examine more fields 12

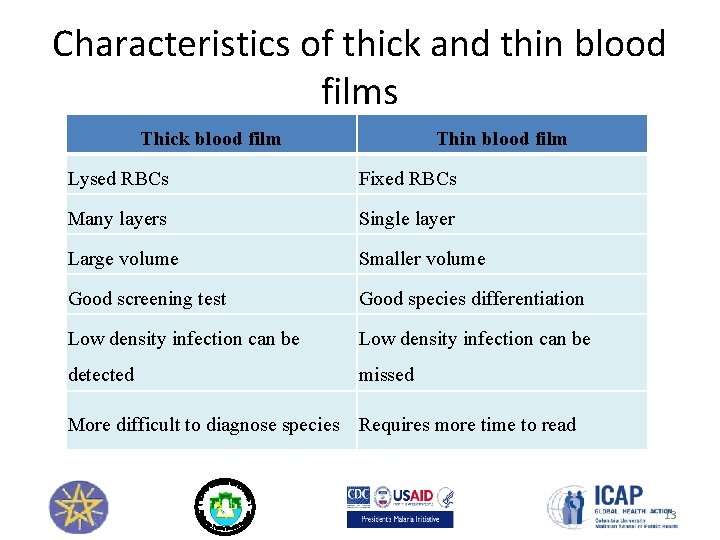
Characteristics of thick and thin blood films Thick blood film Thin blood film Lysed RBCs Fixed RBCs Many layers Single layer Large volume Smaller volume Good screening test Good species differentiation Low density infection can be detected missed More difficult to diagnose species Requires more time to read 13

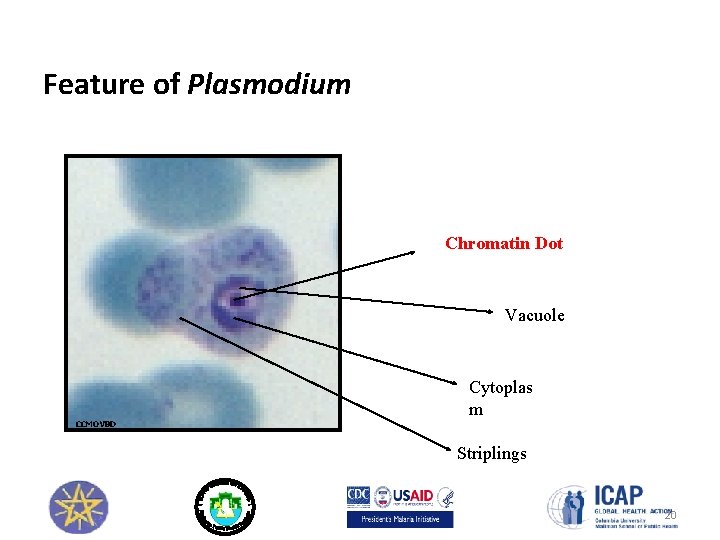
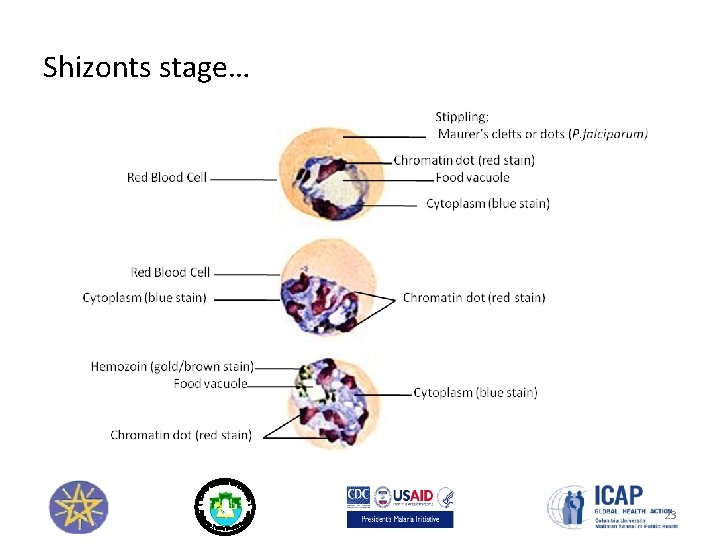
Identification of malaria parasite species and stages using Romanowsky stains – Chromatin: round in shape and stains red. – Cytoplasm : from a ring shape to a totally irregular shape and stained blue. – Malaria pigment: stain in various shades from yellow – gold through brown to black – Stippling: stains in shades of pink which vary among different species 14

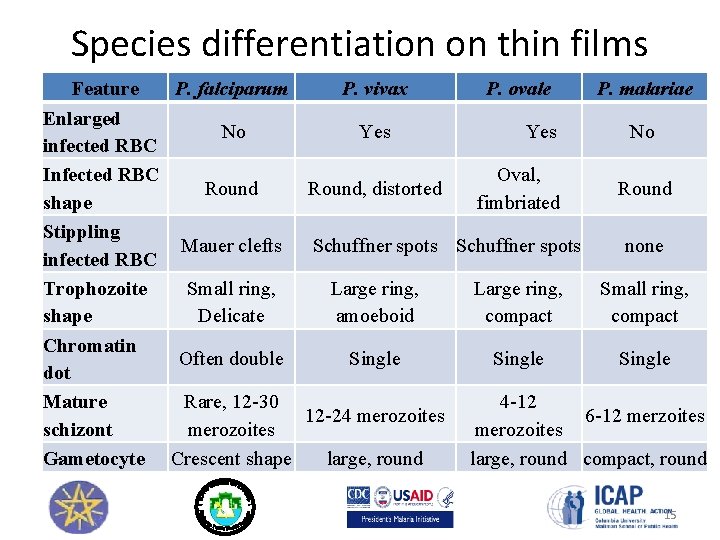
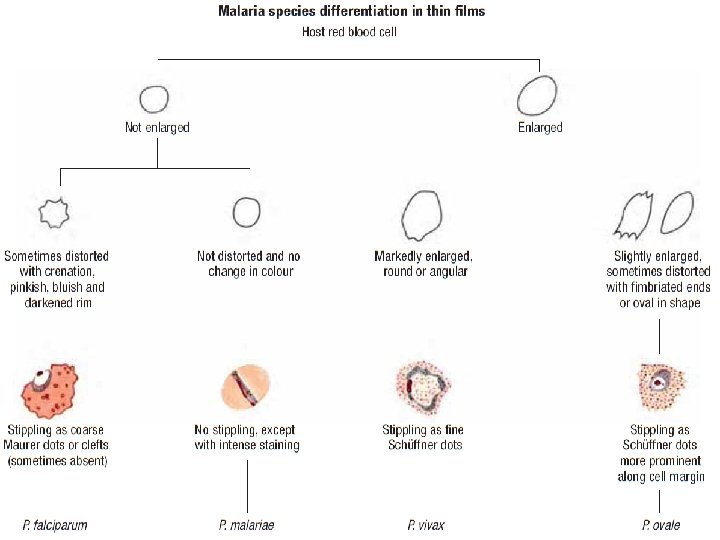
Species differentiation on thin films Feature P. falciparum P. vivax P. ovale P. malariae Enlarged No Yes No infected RBC Infected RBC Oval, Round, distorted Round shape fimbriated Stippling Mauer clefts Schuffner spots none infected RBC Trophozoite Small ring, Large ring, Small ring, shape Delicate amoeboid compact Chromatin Often double Single dot Mature Rare, 12 -30 4 -12 12 -24 merozoites 6 -12 merzoites schizont merozoites Gametocyte Crescent shape large, round compact, round 15

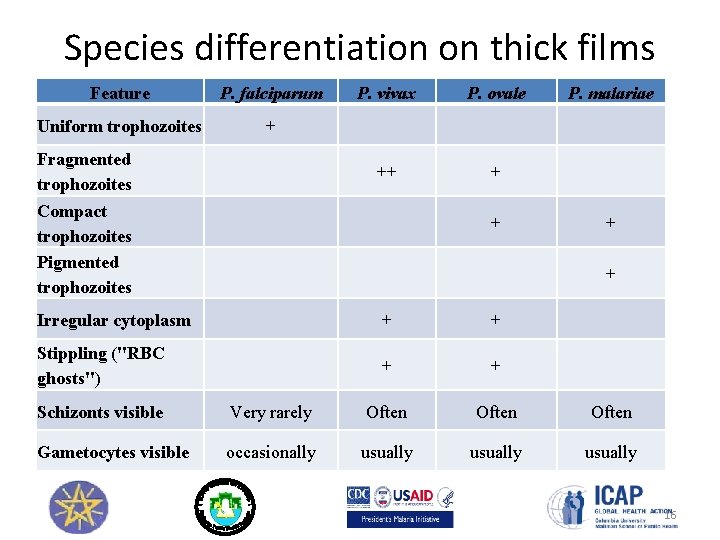
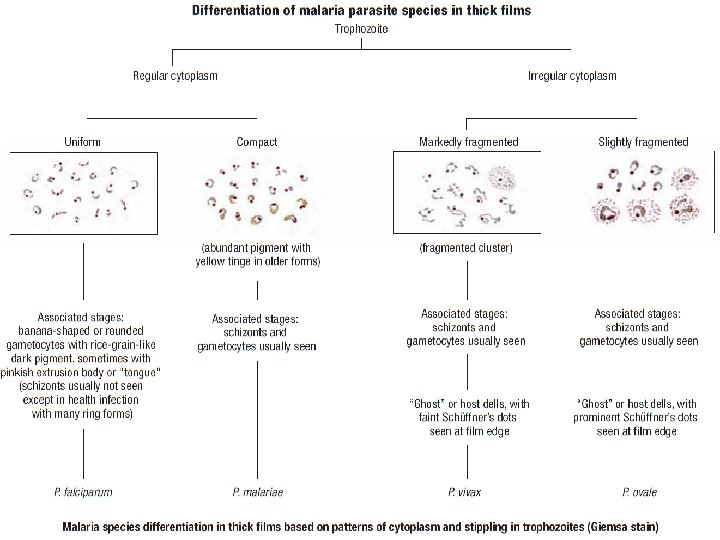
Species differentiation on thick films Feature P. falciparum Uniform trophozoites + Fragmented trophozoites Compact trophozoites Pigmented trophozoites P. vivax P. ovale ++ + + P. malariae + + Irregular cytoplasm + + Stippling ("RBC ghosts") + + Schizonts visible Very rarely Often Gametocytes visible occasionally usually 16

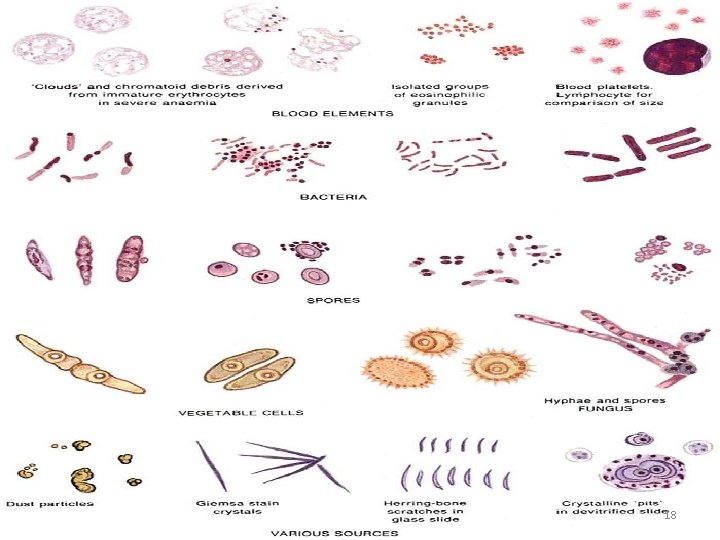
Artefacts – Vegetable spores, yeast, pollen, algae and bacteria in the stain or on the slide – Platelets – Howell-jolly, Cabot ring bodies in anaemic patients – Ghosts of immature red cells mimicking Schüffner’s dots. 17

18

Parasite morphology at specific stages Depends on: – Number and size of chromatin dots – Shape and size of cytoplasm – Degree of pigmentation within cytoplasm – Stages of parasite seen together 19

Feature of Plasmodium Chromatin Dot Vacuole Cytoplas m CCMOVBD Striplings 20

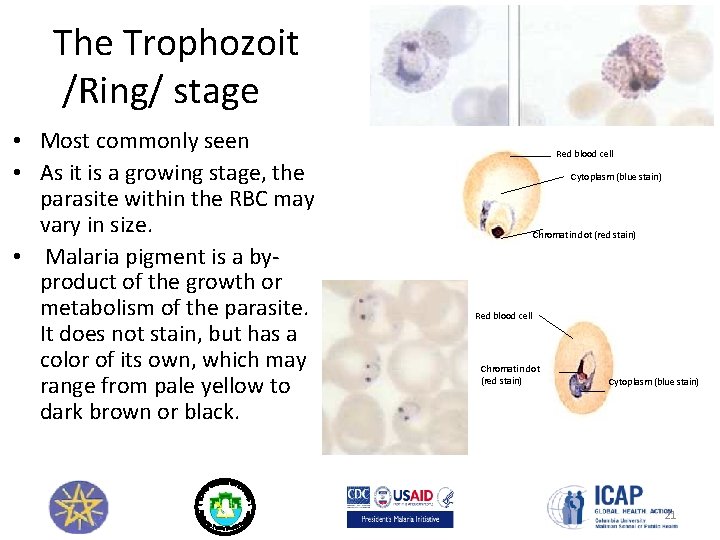
The Trophozoit /Ring/ stage • Most commonly seen • As it is a growing stage, the parasite within the RBC may vary in size. • Malaria pigment is a byproduct of the growth or metabolism of the parasite. It does not stain, but has a color of its own, which may range from pale yellow to dark brown or black. Red blood cell Cytoplasm (blue stain) Chromatin dot (red stain) Red blood cell Chromatin dot (red stain) Cytoplasm (blue stain) 21

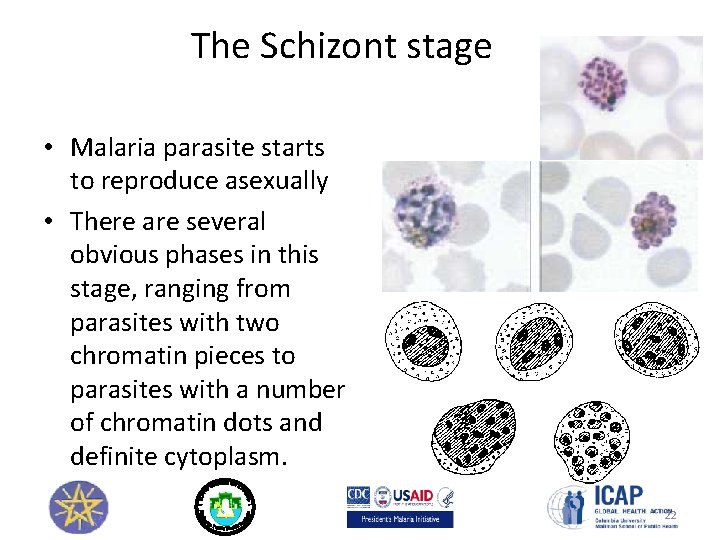
The Schizont stage • Malaria parasite starts to reproduce asexually • There are several obvious phases in this stage, ranging from parasites with two chromatin pieces to parasites with a number of chromatin dots and definite cytoplasm. 22

Shizonts stage… 23

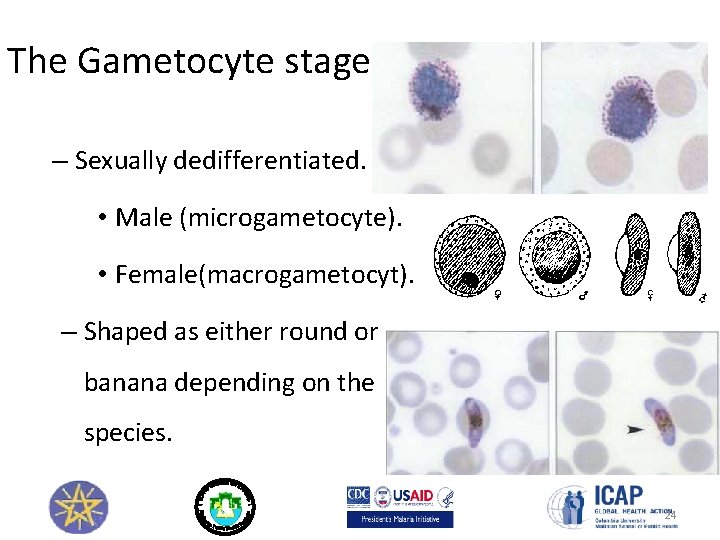
The Gametocyte stage – Sexually dedifferentiated. • Male (microgametocyte). • Female(macrogametocyt). – Shaped as either round or banana depending on the species. 24

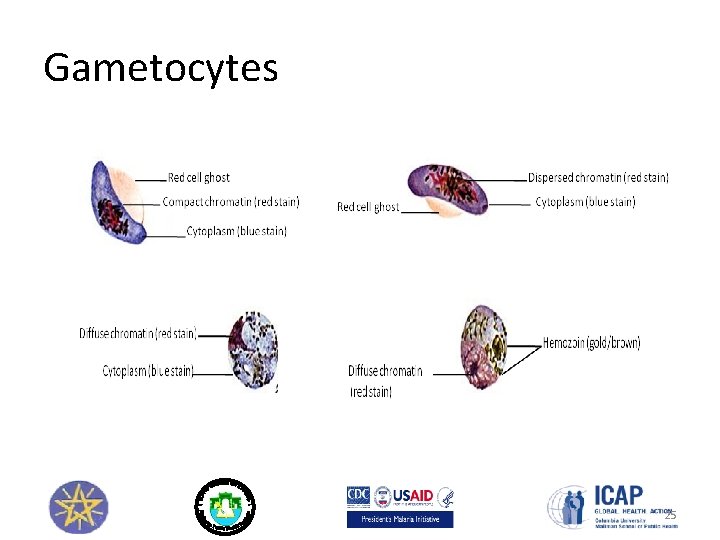
Gametocytes 25

P. falciparum – Gametocyte Stage P. vivax - Gametocyte Stage 26


Plasmodium falciparum Diagnostic points: • • • RBCs not enlarged. Maurer’ dot may be present No Schuffener’s dot Rings appear fine and delicate May be several in one cell. Some rings with 2 chromatin dots. Presence of marginal or appliqué forms/acol formation/. Gametocytes crescent/banana shape Usually trophozoites + gametocytes seen Schizont rarely seen 28

Plasmodium vivax Diagnostic points: • RBCs are usually enlarged. • Schuffner's dots are frequently present in the red cells. • The mature ring forms tend to be large and coarse. • Trophozoites schizonts & gametocytes • Developing forms are frequently present 29

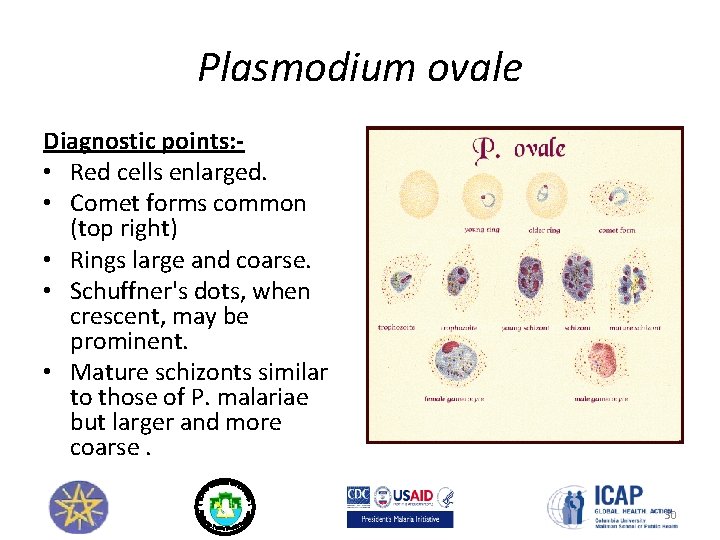
Plasmodium ovale Diagnostic points: • Red cells enlarged. • Comet forms common (top right) • Rings large and coarse. • Schuffner's dots, when crescent, may be prominent. • Mature schizonts similar to those of P. malariae but larger and more coarse. 30

Plasmodium malariae Diagnostic points : • RBC not enlarged • Ring forms may have a Squarish appearance. • Band forms are a characteristic of this species. • Mature schizonts may have a typical daisy head appearance with up to ten merozoites. 31

Estimating Parasitaemia • Important for clinical purposes – To monitor the progress of the disease and – The efficacy of therapy. • Methods 1. 2. 3. 4. Number of parasites/µL of blood (thick film) Number of parasites/µL of blood (thin film) Proportion of parasitized erythrocytes (thin film) Semi quantitative count (thick film): 32

1. Number of parasites/µL of blood (thick film): • Requires observation of at least 100 fields while you count 200 WBCs. • Number of asexual parasites and WBCs should be counted in each field until the number of WBCs reaches 200. • If number of WBCs is unknown, it can be assumed to be 8000/µL 33

Example: • Patient WBC = 8000 ul • Parasite count against 200 WBCs = 650 • Parasite count/ul = 650 x 8000/ul 200 = 26 000 parasites/ul 34

2. Number of parasites/µL of blood (thin film) • Requires the preliminary determination of RBCs number present in the average microscopic field. • The number of asexual parasites is counted in at least 25 microscopic fields. • The number of RBCs in the average microscopic field is about 200, so total RBCs counted in 25 fields is about 200 x 25 = 5000. • RBCs/µl is assumed to be 5 millions/µl for males and 4. 5 millions/µl for females. 35

Example: Parasite counted against 5000 RBCs/25 fields/= 50 # of RBCs in 25 fields= 5000 RBC count = 5 million/Male patient/ 50 x 5, 000 5000 • 50, 000/µl of blood. • • • 36

3. Proportion of parasitized erythrocytes (thin film): • Indicate the percentage of erythrocytes that are infected by malaria parasites. • The number of parasitized erythrocytes (asexual forms) present in 25 microscopic fields is counted divided by the total number of erythrocytes present in these fields (about 5000), and multiplied by 100. 37

Example – Average # of RBCs/25 fields=5000 – # Parasitized RBCs/25 fields=100 – % of parasitized RBCS= 100 X 100 5000 – 2% of RBCs are infected with asexual for of malaria parasite. 38

4. Semi quantitative count (thick film): • Very quick but less accurate. • Used only when not possible to perform more accurate methods. • Reporting: –+ 1 -10 asexual parasites / 100 thick film fields – ++ 11 -100 asexual parasites / 100 thick film fields – +++ 1 -10 asexual parasites / single thick film field – ++++ > 10 asexual parasites / single thick film field 39

Reporting of BF results If positive : • Check the presence of • Different stages ( Throphozoits, schizonts and gametocytes). • Mixed infection • Report the species, stage and density of parasites and if present malaria pigments. • If negative after examination of a minimum of 100 x thick film fields. – Report No parasite or hemoparasite found. • 40

Diagnostic quality Control: Depends upon – Compliance with standards – Availability of supplies/equipments/infrastructure – Condition of microscope – Training of laboratory personnel – Regular supervision – quality of reagents & stains – cleanliness – Work load – Technical ability & type of techniques used 41

Key to identifying malaria parasite stages in thin blood films 1. Are there one or more redstained chromatin dots and blue cytoplasm? Yes: go to 2 No: what you see is not a parasite 2. Are the size and shape right for a malaria parasite? Yes: what you see is probably a malaria parasite; go to 3 No: what you see is not a parasite 42

Key… 3. Is there malaria pigment in the cell? Yes: go to 7 No: go to 4 4. Does the parasite have one chromatin dot attached to blue cytoplasm in the form of a regular ring with a vacuole? Yes: this is a trophozoite stage No: go to 5 43

Key…. 5. Does the parasite have one chromatin dot attached to blue cytoplasm in the form of a small solid or regular ring or with a vacuole? Yes: this is a trophozoite stage No: go to 6 6. Is the parasite with one chromatin dot irregular or fragmented? Yes: this is a trophozoite stage No: go to 8 44

Key… 7. Does the parasite with malaria pigment have one chromatin dot? Yes: go to 8 No: go to 9 8. Does the parasite have a vacuole or is it still fragmented in some way? Yes: this is probably a late trophozoite stage No: go to 11 45

Key… 9. Does the parasite have two chromatin dots attached to a ring and also have a vacuole? Yes: this is a trophozoite stage No: go to 10 10. Does the parasite have between 2 and 32 chromatin dots and pigment? Yes: this is a schizont stage 46

Key… 11. Is the parasite rounded or bananashaped? Rounded: go to 12 Banana-shaped: go to 14 12. Does the rounded parasite have clearly stained chromatin and deep blue cytoplasm? Yes: this is a female gametocyte No: go to 13 47

Key… 13. Does the rounded parasite have a reddish overall colour to the staining, so that the chromatin cannot be clearly seen? Yes: this is a male gametocyte 14. Does the banana-shaped parasite have densely stained blue cytoplasm and bright red chromatin? Yes: this is a female gametocyte No: go to 15 15. Does the banana-shaped parasite have a reddish overall color to the staining, so that the chromatin is indistinct? Yes: This is a male gametocyte 48

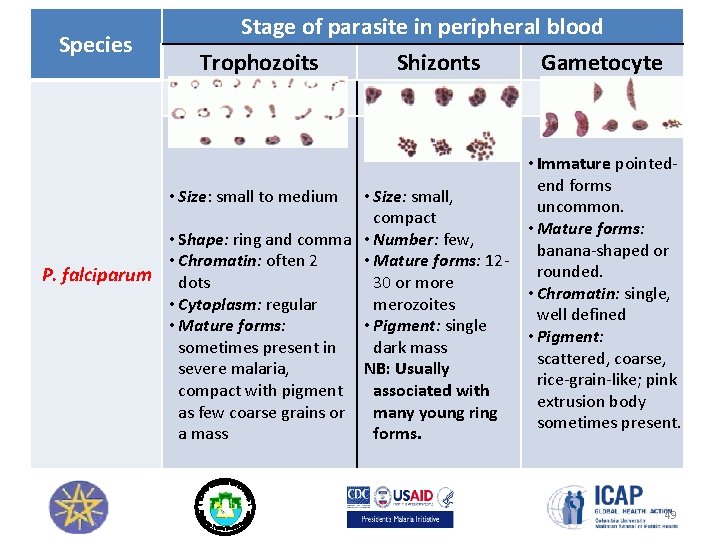
Species Stage of parasite in peripheral blood Trophozoits Shizonts • Size: small, compact • Shape: ring and comma • Number: few, • Chromatin: often 2 • Mature forms: 12 P. falciparum dots 30 or more • Cytoplasm: regular merozoites • Mature forms: • Pigment: single sometimes present in dark mass severe malaria, NB: Usually compact with pigment associated with as few coarse grains or many young ring a mass forms. • Size: small to medium Gametocyte • Immature pointedend forms uncommon. • Mature forms: banana-shaped or rounded. • Chromatin: single, well defined • Pigment: scattered, coarse, rice-grain-like; pink extrusion body sometimes present. 49

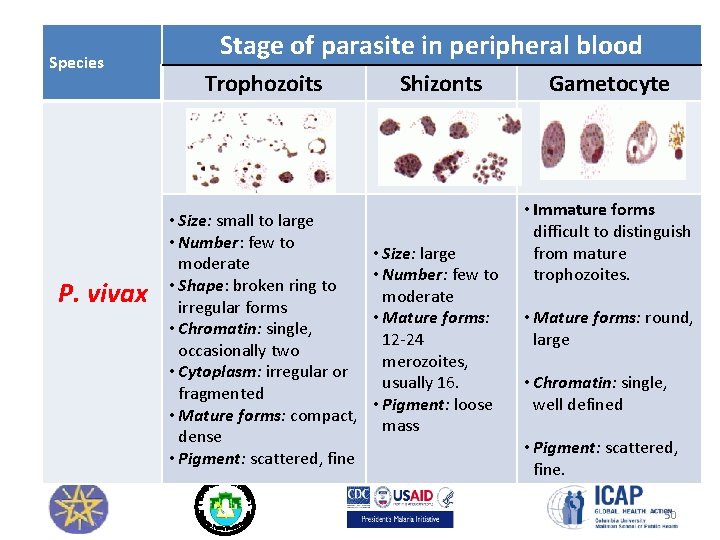
Species P. vivax Stage of parasite in peripheral blood Trophozoits • Size: small to large • Number: few to moderate • Shape: broken ring to irregular forms • Chromatin: single, occasionally two • Cytoplasm: irregular or fragmented • Mature forms: compact, dense • Pigment: scattered, fine Shizonts • Size: large • Number: few to moderate • Mature forms: 12 -24 merozoites, usually 16. • Pigment: loose mass Gametocyte • Immature forms difficult to distinguish from mature trophozoites. • Mature forms: round, large • Chromatin: single, well defined • Pigment: scattered, fine. 50

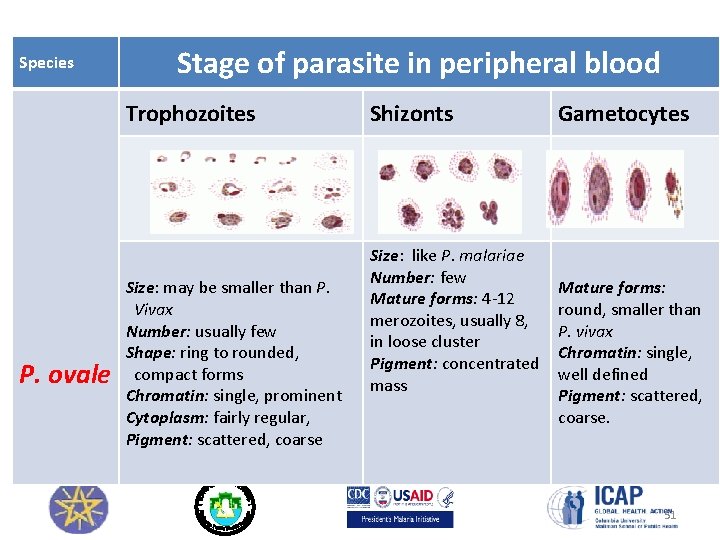
Species Stage of parasite in peripheral blood Trophozoites P. ovale Size: may be smaller than P. Vivax Number: usually few Shape: ring to rounded, compact forms Chromatin: single, prominent Cytoplasm: fairly regular, Pigment: scattered, coarse Shizonts Size: like P. malariae Number: few Mature forms: 4 -12 merozoites, usually 8, in loose cluster Pigment: concentrated mass Gametocytes Mature forms: round, smaller than P. vivax Chromatin: single, well defined Pigment: scattered, coarse. 51

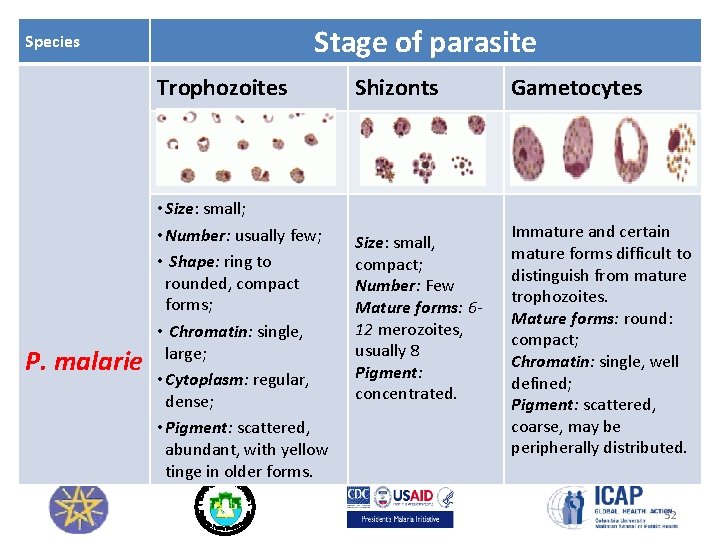
Stage of parasite Species Trophozoites P. malarie • Size: small; • Number: usually few; • Shape: ring to rounded, compact forms; • Chromatin: single, large; • Cytoplasm: regular, dense; • Pigment: scattered, abundant, with yellow tinge in older forms. Shizonts Size: small, compact; Number: Few Mature forms: 612 merozoites, usually 8 Pigment: concentrated. Gametocytes Immature and certain mature forms difficult to distinguish from mature trophozoites. Mature forms: round: compact; Chromatin: single, well defined; Pigment: scattered, coarse, may be peripherally distributed. 52

B e ta m Ameseginale hu