Module 5 Communication homeostasis energy Block 2 C

Module 5 Communication, homeostasis & energy Block 2 C – 5. 5 Animal responses 5. 5. 10 Contraction of the skeletal muscle 2 LESSONS

Spec

Learning Objectives To understand the sliding filament model Success Criteria Outline the role of ATP in muscle contraction (Grade E - D) Outline how the supply of ATP is maintained in the muscle (Grade C –B) Explain using diagrams the sliding filament model of muscular contraction (Grade B – A)

Starter 1. Blood vessels such as arterioles contain circular smooth muscle. Contraction of this muscle constricts the vessel. Why do blood vessels not need longitudinal muscle to act against the circular muscle in order to cause dilation? 2. Suggest the advantage of the electrical activity of the heart being able to pass from atria walls to ventricle walls only at the AV node
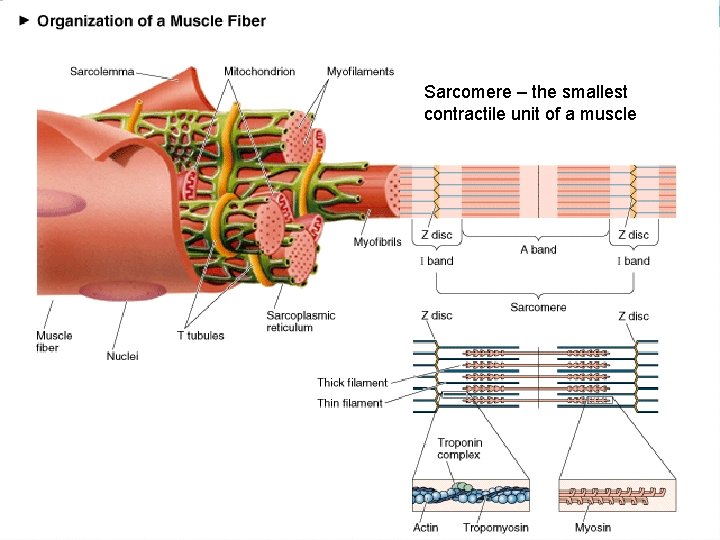
Sarcomere – the smallest contractile unit of a muscle
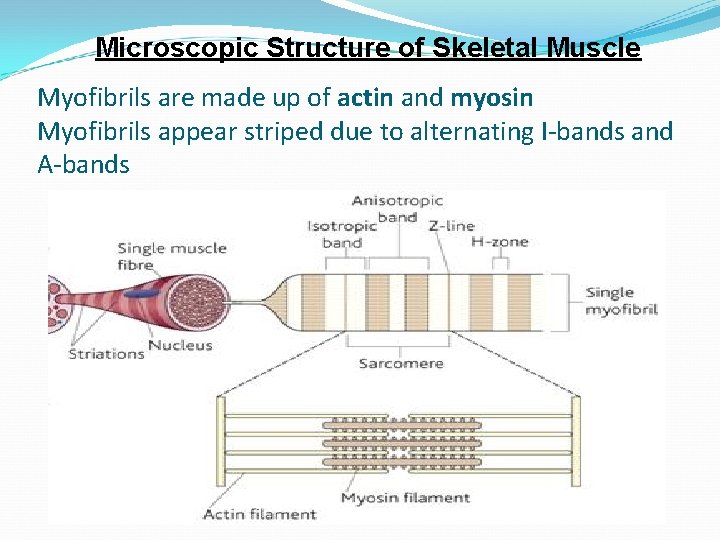
Microscopic Structure of Skeletal Muscle Myofibrils are made up of actin and myosin Myofibrils appear striped due to alternating I-bands and A-bands

Microscopic Structure of Skeletal Muscle Light bands are isotropic bands (Ibands) only actin is found in these bands Dark bands are anisotropic bands (Abands) actin and myosin overlap in these bands
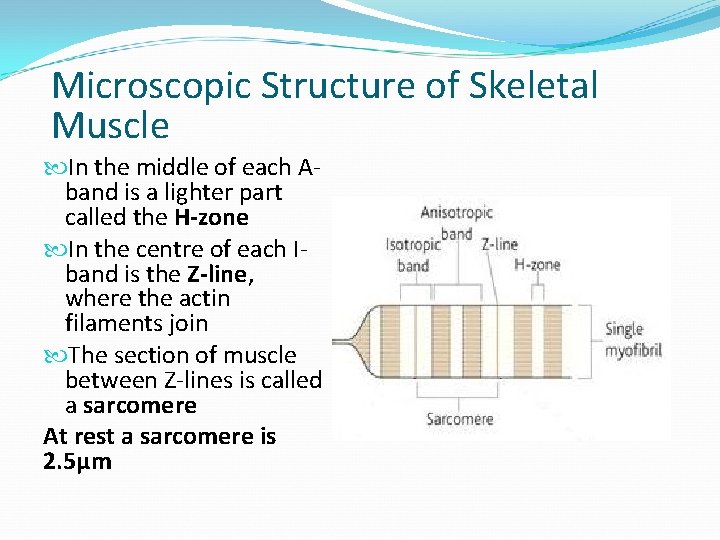
Microscopic Structure of Skeletal Muscle In the middle of each Aband is a lighter part called the H-zone In the centre of each Iband is the Z-line, where the actin filaments join The section of muscle between Z-lines is called a sarcomere At rest a sarcomere is 2. 5µm

Examination question … Figure 1 shows a diagram of part of a muscle myofibril. (a) Name the protein present in the filaments labelled W and X. (1 mark) 9
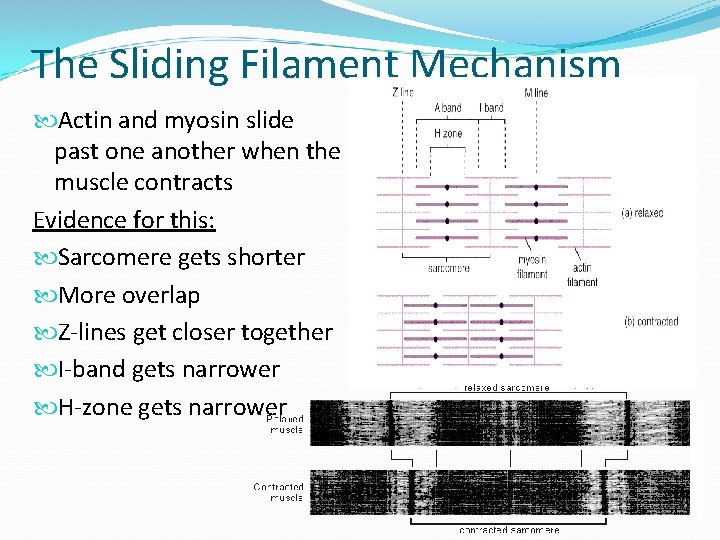
The Sliding Filament Mechanism Actin and myosin slide past one another when the muscle contracts Evidence for this: Sarcomere gets shorter More overlap Z-lines get closer together I-band gets narrower H-zone gets narrower
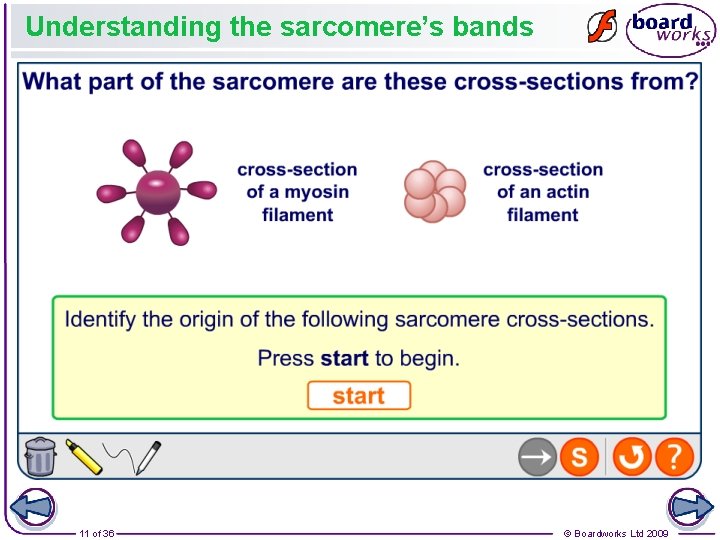
Understanding the sarcomere’s bands 11 of 36 © Boardworks Ltd 2009
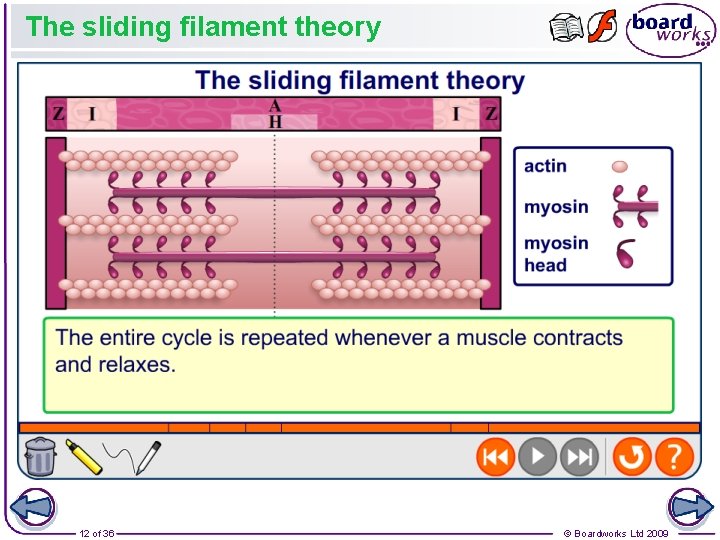
The sliding filament theory 12 of 36 © Boardworks Ltd 2009

How does the sarcomere change? 13 of 36 © Boardworks Ltd 2009
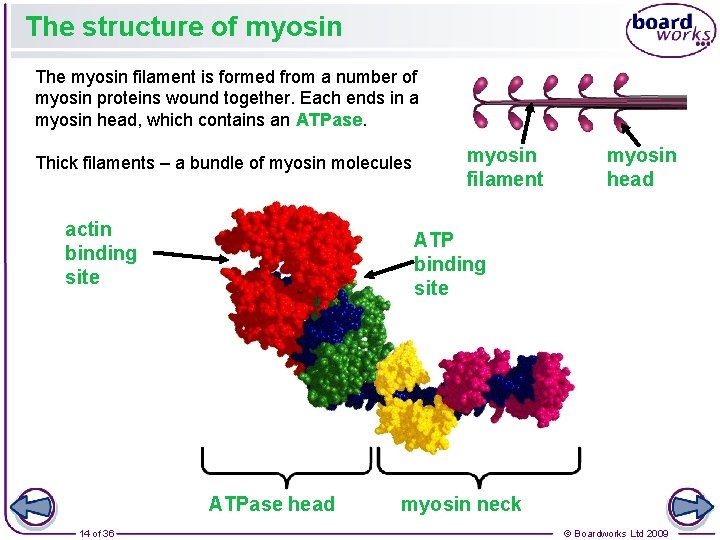
The structure of myosin The myosin filament is formed from a number of myosin proteins wound together. Each ends in a myosin head, which contains an ATPase. Thick filaments – a bundle of myosin molecules actin binding site myosin head ATP binding site ATPase head 14 of 36 myosin filament myosin neck © Boardworks Ltd 2009
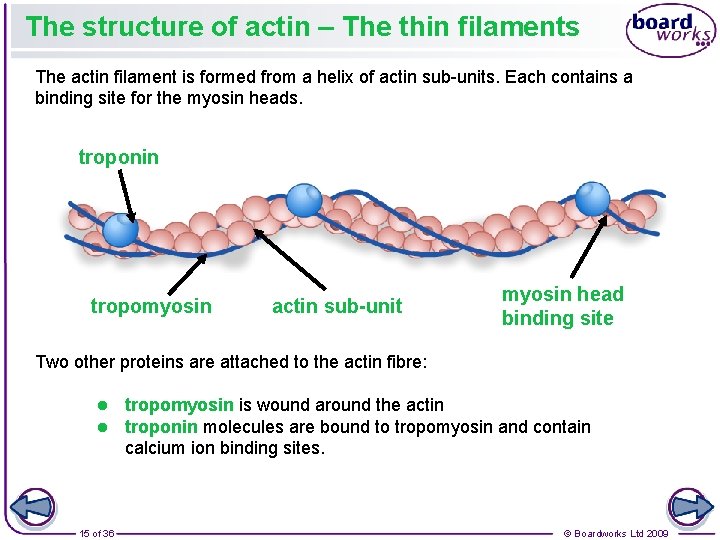
The structure of actin – The thin filaments The actin filament is formed from a helix of actin sub-units. Each contains a binding site for the myosin heads. troponin tropomyosin actin sub-unit myosin head binding site Two other proteins are attached to the actin fibre: l l 15 of 36 tropomyosin is wound around the actin troponin molecules are bound to tropomyosin and contain calcium ion binding sites. © Boardworks Ltd 2009

Muscle Contraction – Sliding Filament hypothesis Heads of myosin form cross-bridges with the actin filaments (attach to binding sites) Myosin heads flex together and pull the actin along the myosin They detach Return to original angle and re-attach (uses ATP) Repeats 100 times a second
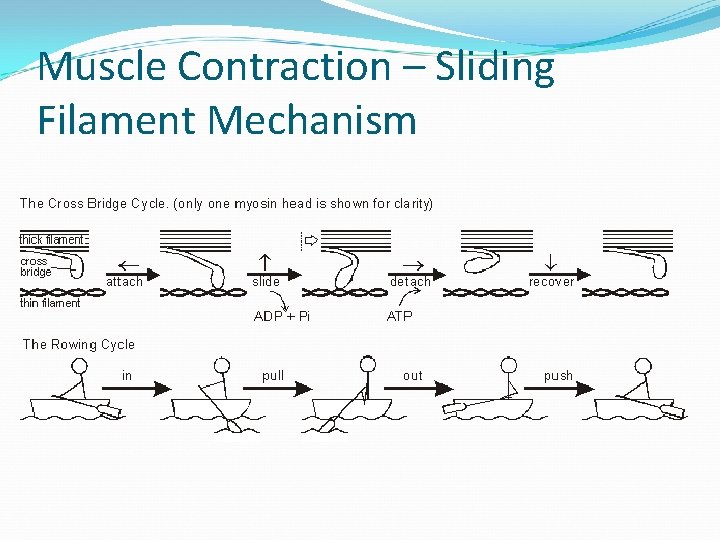
Muscle Contraction – Sliding Filament Mechanism
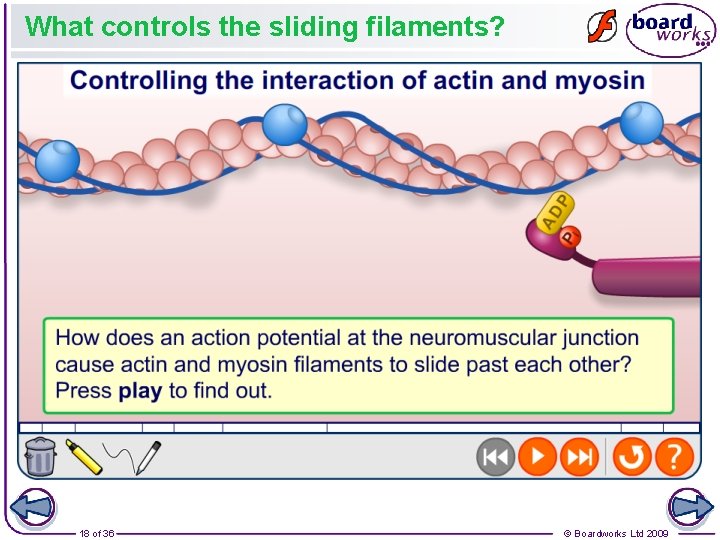
What controls the sliding filaments? 18 of 36 © Boardworks Ltd 2009

Task Using the play doh demonstrate muscle contraction Four sections 1. Arrival of an action potential – Ca 2+, troponin, tropomyosin, actin-myosin crossbridge 2. Movement of the actin filament – ATPase, ATP, power stroke 3. Breaking of the cross bridge – ATP, myosin head 4. Return to resting state – troponin, Ca 2+, sarcoplasmic reticulum Use keywords – write down a flowchart explaining each stage

Maintaining ATP supply 1. Aerobic respiration in muscle cells mitochondria Needs a supply of respiratory substrate and oxygen 21 of 36 2. Anaerobic respiration in muscle cell sarcoplasm Produces lactate and can lead to fatigue/cramp. 3. Creatinine phosphate – another chemical present in muscle cells can donate its phosphate to recharge ADP back to ATP (supports a further 2 -4 seconds © Boardworks Ltd 2009

Investigating muscle stimulation An electromyograph (EMG) can be used to investigate electrical activity of muscles Electrodes applied to the skin surface detects action potentials in the muscle fibres. The recorded trace is called an electromyogram. The amplitude of an EMG recording refelects the number and size of motor units involved in contraction – More powerful contraction = higher amplitude. Page 105 – figure 8 – describe what you see on each trace

Plenary • Complete cloze sheet – summary sliding filament theory 23 of 36 © Boardworks Ltd 2009

Sliding filament theory • • • When a nerve impulse arrives at a neuromuscular junction, calcium ions are released from the sarcoplasmic reticulum. The calcium ions diffuse through the sarcoplasm. This initiates the movement of the protein filaments as follows: Calcium ions attach to the troponin molecules causing them to move. As a result, the tropomyosin on the actin filament shifts position, exposing myosin binding sites on the actin filaments. Myosin heads bind with myosin binding sites on the actin filament, forming cross bridges. When the myosin head binds to the actin, ADP and Pi on the myosin head are released. The myosin changes shape, causing the myosin head to nod forward. This movement results in the relative movement of the filaments. The attached actin moves over the myosin. An ATP molecule binds to the myosin head. This causes the myosin head to detach. An ATPase on the myosin head hydrolyses the ATP forming ADP and Pi. This hydrolysis causes a change in the shape of the myosin head. It returns to its upright position. This enables the cycle to start again. 24 of 36 © Boardworks Ltd 2009
Crash course video • https: //www. youtube. com/watch? v=jqy 0 i 1 KXUO 4 25 of 36 © Boardworks Ltd 2009

Learning Objectives • To understand the sliding filament model Success Criteria • Outline the role of ATP in muscle contraction (Grade E - D) • Outline how the supply of ATP is maintained in the muscle (Grade C –B) • Explain using diagrams the sliding filament model of muscular contraction (Grade B – A) 26 of 36 © Boardworks Ltd 2009
- Slides: 26