Module 4 Nebido pharmaceutical and pharmacological data Approval

Module 4 Nebido® pharmaceutical and pharmacological data Approval Number: G. MKT. GM. MH. 04. 2018. 0513
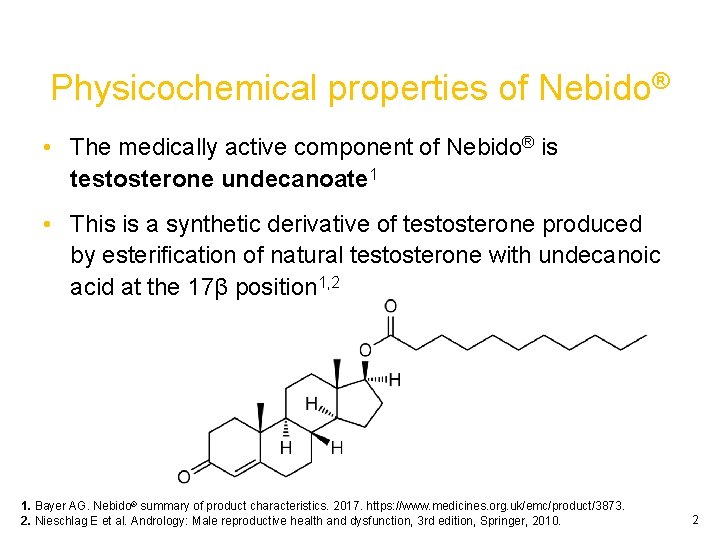
Physicochemical properties of Nebido® • The medically active component of Nebido® is testosterone undecanoate 1 • This is a synthetic derivative of testosterone produced by esterification of natural testosterone with undecanoic acid at the 17β position 1, 2 1. Bayer AG. Nebido® summary of product characteristics. 2017. https: //www. medicines. org. uk/emc/product/3873. 2. Nieschlag E et al. Andrology: Male reproductive health and dysfunction, 3 rd edition, Springer, 2010. 2

Physicochemical properties of Nebido® (2) • Nebido® is the first registered testosterone undecanoate preparation for intramuscular injection – Nebido® is available in corresponding individual packaging – One Nebido® vial contains 1, 000 mg of testosterone undecanoate in 4 m. L of solution for injection – Nebido® also contains benzyl benzoate and refined castor oil • Nebido® has a shelf life of 5 years and is recommended to be stored at room temperature; it is stable at 30 °C for at least 24 months and 40 °C for at least 6 months https: //www. nebido. com/en/hcp/product-information/nebido/; Bayer AG. Nebido® summary of product characteristics. 2017. https: //www. medicines. org. uk/emc/product/3873 3
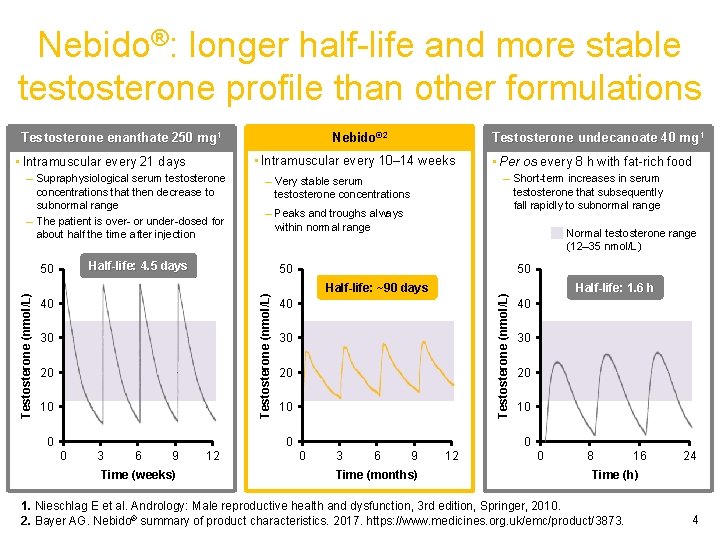
Nebido®: longer half-life and more stable testosterone profile than other formulations Testosterone enanthate 250 mg 1 – Supraphysiological serum testosterone concentrations that then decrease to subnormal range – The patient is over- or under-dosed for about half the time after injection – Peaks and troughs always within normal range Normal testosterone range (12– 35 nmol/L) 30 20 10 0 50 Half-life: ~90 days Testosterone (nmol/L) 50 40 40 30 20 10 0 0 3 6 9 Time (weeks) • Per os every 8 h with fat-rich food – Short-term increases in serum testosterone that subsequently fall rapidly to subnormal range – Very stable serum testosterone concentrations Half-life: 4. 5 days 50 Testosterone undecanoate 40 mg 1 • Intramuscular every 10– 14 weeks • Intramuscular every 21 days Testosterone (nmol/L) Nebido® 2 12 Half-life: 1. 6 h 40 30 20 10 0 0 3 6 9 Time (months) 12 0 8 16 24 Time (h) 1. Nieschlag E et al. Andrology: Male reproductive health and dysfunction, 3 rd edition, Springer, 2010. 2. Bayer AG. Nebido® summary of product characteristics. 2017. https: //www. medicines. org. uk/emc/product/3873. 4

Nebido® pharmacokinetic properties: absorption • Following intramuscular injection of Nebido® as an oily solution, testosterone undecanoate is gradually released from the depot into the circulation 1 • It is almost completely cleaved by serum esterases into testosterone and undecanoic acid 1 • An increase of serum testosterone above basal values can already be measured 1 day after administration and maximum concentrations are reached within 1– 2 weeks 1 • The intramuscular route of administration of Nebido® and the esterification at position 17β avoids hepatic first-pass metabolism 1, 2 1. Bayer AG. Nebido® summary of product characteristics. 2017. https: //www. medicines. org. uk/emc/product/3873. 2. Nieschlag E et al. Andrology: Male reproductive health and dysfunction, 3 rd edition, Springer, 2010. 5

Nebido® pharmacokinetic properties: distribution and metabolism • The majority (~98%) of circulating testosterone released by cleavage of testosterone undecanoate is bound to transport proteins and is not considered biologically active 1 Bound with higher affinity to SHBG • Testosterone is metabolized primarily in the liver but also in other tissues, including adipose tissue and skin 2 54% • Undecanoic acid is metabolized by β-oxidation like other aliphatic carboxylic acids 3 • The major active metabolites of testosterone are DHT, produced by the action of 5α-reductase and estradiol, generated by aromatization 1, 3 Bound with lower affinity to albumin 44% Free 2% DHT, dihydrotestosterone; SHBG, sex hormone-binding globulin 1. Nieschlag E et al. Andrology: Male reproductive health and dysfunction, 3 rd edition, Springer, 2010. 2. Melmed S et al. Williams Textbook of Endocrinology, 13 th edition, Elsevier, 2016. 3. Bayer AG. Nebido® summary of product characteristics. 2017. https: //www. medicines. org. uk/emc/product/3873 6

Nebido® pharmacokinetic properties: elimination • In elderly men, the elimination half-life of testosterone is ~1 h after intravenous infusion • Following administration of radiolabelled testosterone: • ~90% of radioactivity is found in urine as glucuronic and sulphuric acid conjugates • ~6% of radioactivity is found in feces after undergoing enterohepatic circulation • Metabolites excreted in the urine include androsterone and etiocholanolone Bayer AG. Nebido® summary of product characteristics. 2017. https: //www. medicines. org. uk/emc/product/3873. 7
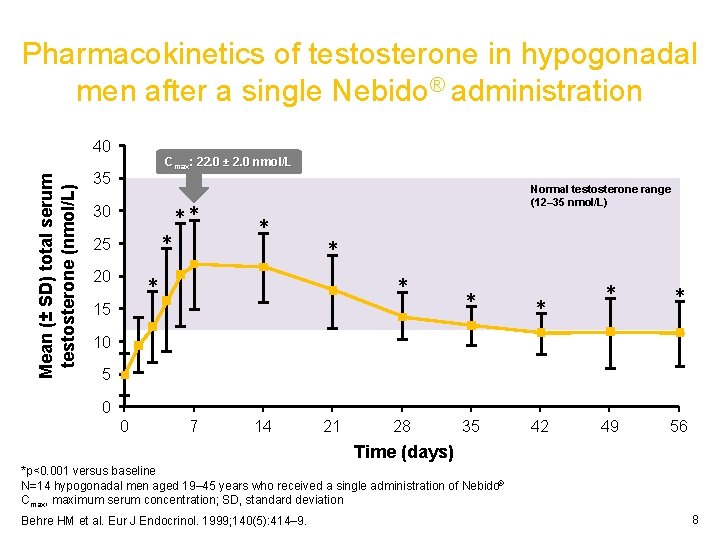
Pharmacokinetics of testosterone in hypogonadal men after a single Nebido® administration Mean (± SD) total serum testosterone (nmol/L) 40 Cmax: 22. 0 ± 2. 0 nmol/L 35 30 * 25 20 ** Normal testosterone range (12– 35 nmol/L) * * * 15 * * * 35 42 * * 49 56 10 5 0 0 7 14 21 28 Time (days) *p<0. 001 versus baseline N=14 hypogonadal men aged 19– 45 years who received a single administration of Nebido® Cmax, maximum serum concentration; SD, standard deviation Behre HM et al. Eur J Endocrinol. 1999; 140(5): 414– 9. 8
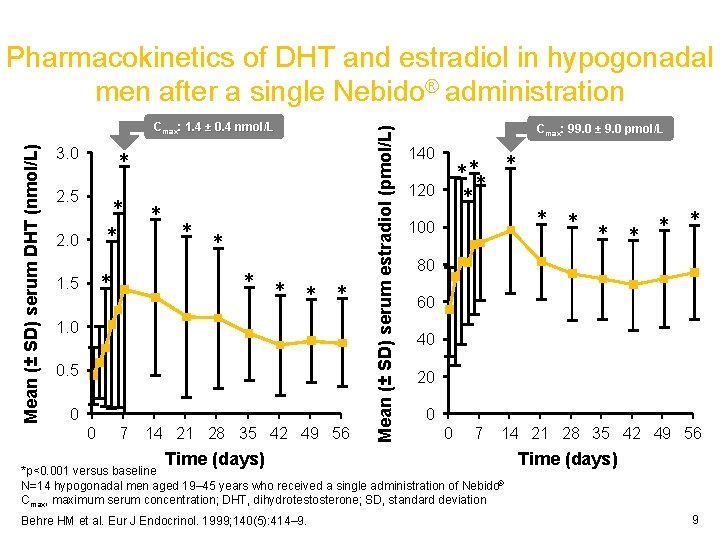
Mean (± SD) serum DHT (nmol/L) Cmax: 1. 4 ± 0. 4 nmol/L 3. 0 * 2. 5 * * 2. 0 * * * * 1. 5 * 1. 0 0. 5 0. 0 0 7 14 21 28 35 42 49 56 Time (days) Mean (± SD) serum estradiol (pmol/L) Pharmacokinetics of DHT and estradiol in hypogonadal men after a single Nebido® administration Cmax: 99. 0 ± 9. 0 pmol/L 140 *** * * 120 100 * * 80 60 40 20 0 0 7 14 21 28 35 42 49 56 *p<0. 001 versus baseline N=14 hypogonadal men aged 19– 45 years who received a single administration of Nebido® Cmax, maximum serum concentration; DHT, dihydrotestosterone; SD, standard deviation Behre HM et al. Eur J Endocrinol. 1999; 140(5): 414– 9. * * Time (days) 9
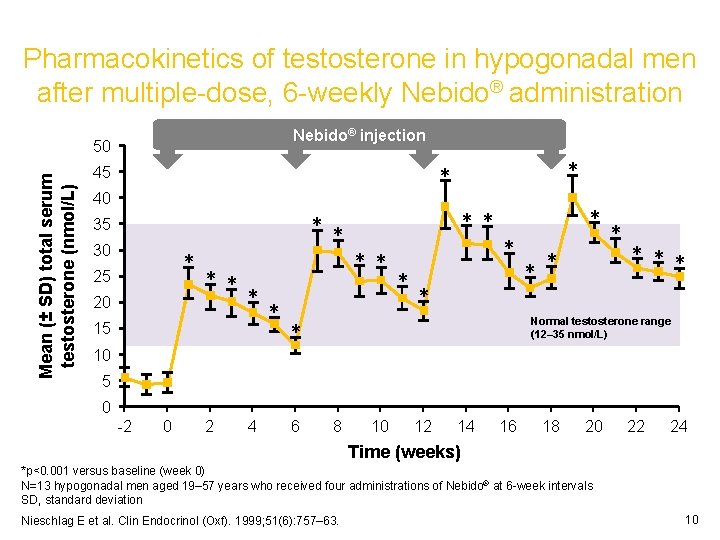
Pharmacokinetics of testosterone in hypogonadal men after multiple-dose, 6 -weekly Nebido® administration Nebido® injection Mean (± SD) total serum testosterone (nmol/L) 50 * * 45 40 ** 35 30 * 25 20 ** * 15 10 * ** * * * *** Normal testosterone range (12– 35 nmol/L) * 5 0 -2 0 2 4 6 8 10 12 14 16 18 20 22 24 Time (weeks) *p<0. 001 versus baseline (week 0) N=13 hypogonadal men aged 19– 57 years who received four administrations of Nebido® at 6 -week intervals SD, standard deviation Nieschlag E et al. Clin Endocrinol (Oxf). 1999; 51(6): 757– 63. 10
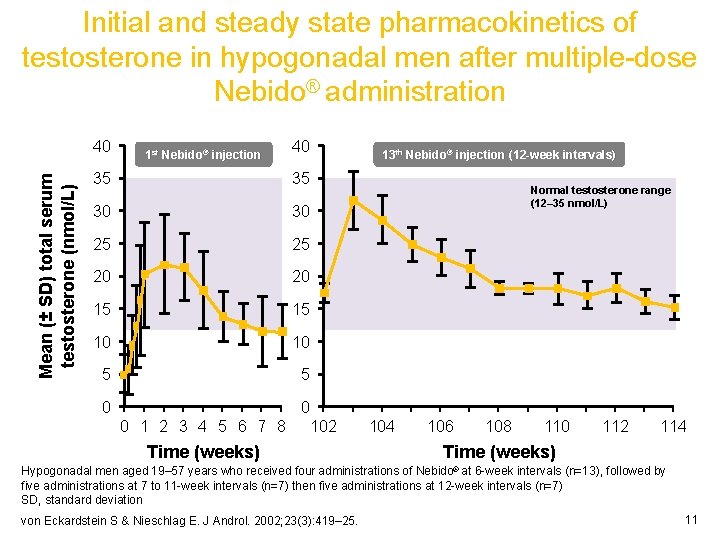
Initial and steady state pharmacokinetics of testosterone in hypogonadal men after multiple-dose Nebido® administration Mean (± SD) total serum testosterone (nmol/L) 40 1 st Nebido® injection 40 35 35 30 30 25 25 20 20 15 15 10 10 5 5 0 0 13 th Nebido® injection (12 -week intervals) Normal testosterone range (12– 35 nmol/L) 0 1 2 3 4 5 6 7 8 102 103 104 105 106 107 108 109 110 111 112 113 114 Time (weeks) Hypogonadal men aged 19– 57 years who received four administrations of Nebido® at 6 -week intervals (n=13), followed by five administrations at 7 to 11 -week intervals (n=7) then five administrations at 12 -week intervals (n=7) SD, standard deviation von Eckardstein S & Nieschlag E. J Androl. 2002; 23(3): 419– 25. 11
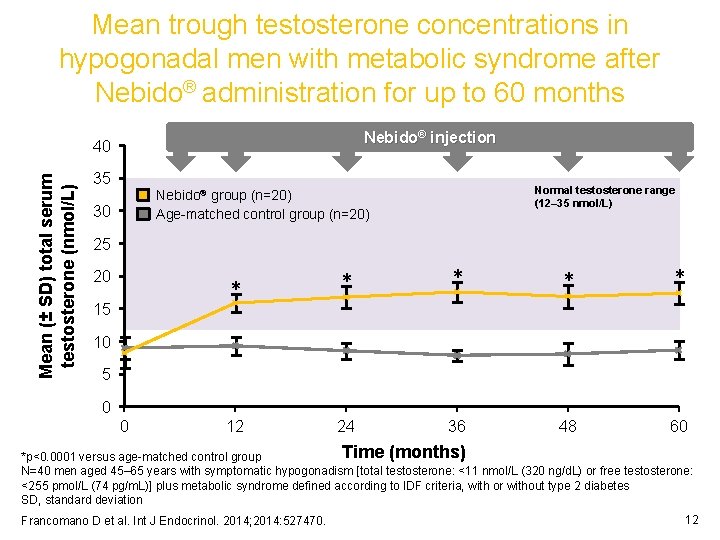
Mean trough testosterone concentrations in hypogonadal men with metabolic syndrome after Nebido® administration for up to 60 months Nebido® injection Mean (± SD) total serum testosterone (nmol/L) 40 35 Normal testosterone range (12– 35 nmol/L) Nebido® group (n=20) Age-matched control group (n=20) 30 25 20 15 * * * 12 24 36 48 60 10 5 0 0 Time (months) *p<0. 0001 versus age-matched control group N=40 men aged 45– 65 years with symptomatic hypogonadism [total testosterone: <11 nmol/L (320 ng/d. L) or free testosterone: <255 pmol/L (74 pg/m. L)] plus metabolic syndrome defined according to IDF criteria, with or without type 2 diabetes SD, standard deviation Francomano D et al. Int J Endocrinol. 2014; 2014: 527470. 12

Conclusions for Nebido® dosing derived from pharmacokinetics studies Measurement of serum testosterone for individualization of Nebido® therapy 6 -week interval 0 12 -week interval 6 12 -week interval 18 30 weeks • Measurement of serum trough testosterone concentrations and assessment of clinical symptoms should be considered for individualization of Nebido® therapy – Serum trough testosterone concentrations should be in the lower third of the normal range (12– 18 nmol/L) • It is assumed that steady state serum testosterone concentrations are achieved after the first 6 months of treatment; therefore, it is advisable to monitor serum trough testosterone concentrations before the fourth Nebido® injection https: //www. nebido. com/en/hcp/product-information/faq-nebido/; Bayer AG. Nebido® summary of product characteristics. 2017. https: //www. medicines. org. uk/emc/product/3873 13

Additional considerations for Nebido® administration in select patients • Following the standard Nebido® dosing schedule, most hypogonadal patients will achieve eugonadal serum testosterone concentrations within 18 weeks • Occasionally, adjustment of dosing frequency may need to be considered for some patients, based on certain characteristics including: – Type and etiology of hypogonadism 1 – Age 1 – Size (body weight, body mass index, body surface area)1 – Baseline testosterone level 1, 2 – Pharmacogenetics (polymorphisms in genes affecting testosterone metabolism androgen sensitivity)2, 3 1. Moisey R et al. Clin Endocrinol (Oxf). 2008; 69(4): 642– 7. 2. Zitzmann M & Nieschlag E. J Clin Endocrinol Metab. 2007; 92(10): 3844– 53. 3. Bang AK et al. Front Endocrinol (Lausanne). 2013; 4: 94. 14

Summary • Nebido® is the first registered testosterone undecanoate preparation for intramuscular injection • It has a longer half-life and more stable testosterone profile than other oral or conventional intramuscular formulations • Stable physiological serum testosterone concentrations are achieved within 1 week after initial Nebido® administration – Serum concentrations of DHT and estradiol follow the pattern of testosterone • Physiological serum testosterone concentrations are maintained when Nebido® is given at intervals of ~12 weeks after an initial interval of 6 weeks • Occasionally, Nebido® dosing frequency adjustments may be considered in certain patients, based on their age, size, baseline testosterone level and pharmacogenetic makeup DHT, dihydrotestosterone 15
- Slides: 15