MODULE 2 Special kinds of combustion Flash fire

MODULE 2

Special kinds of combustion • Flash fire üA flash fire is a sudden, intense fire caused by ignition of a mixture of air and a dispersed flammable substance such as a solid, flammable or combustible liquid or flammable gas. It is characterized by high temperature, short duration, a rapidly moving flame front. ü Flash fires may occur in environment where fuel, typically flammable gas or dust is mixed with air in concentrations suitable for combustion.

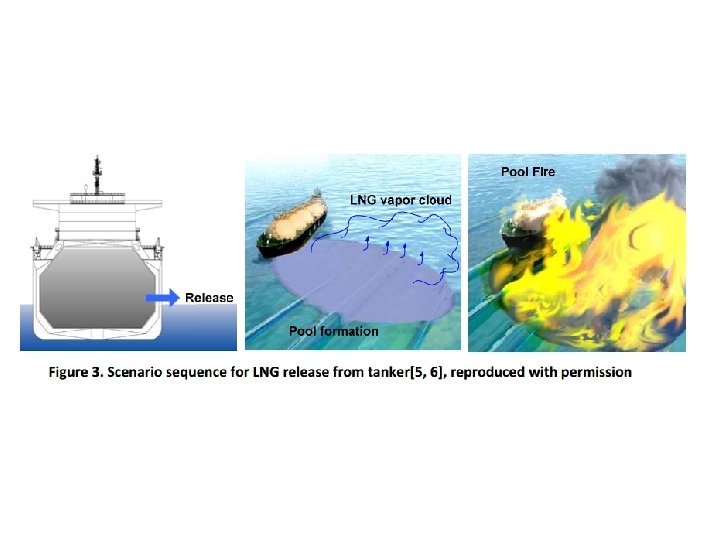
Pool fire • A pool fire is a turbulent diffusion fire burning above a horizontal pool of vaporising hydrocarbon fuel where the fuel has zero or low initial momentum. • Fires in the open will be well ventilated (fuelcontrolled), but fires within enclosures may become under-ventilated (ventilationcontrolled). Pool fires may be static (e. g. where the pool is contained) or 'running' fires.


Boil over • A boilover (or boil-over) type of fire refers to an extremely hazardous situation where an attempt is made to extinguish semienclosed oil or petrochemical fueled fire with water. The hazard results due to the difference in density between oil and water. • As water is poured onto the fuel, it quickly sinks to the bottom of the container due to the water's higher density, and has little effect on extinguishing the flames on the surface. Under certain conditions, after some time, the water on the bottom rapidly vaporises into steam, causing it to expand more than 1700 times in volume.
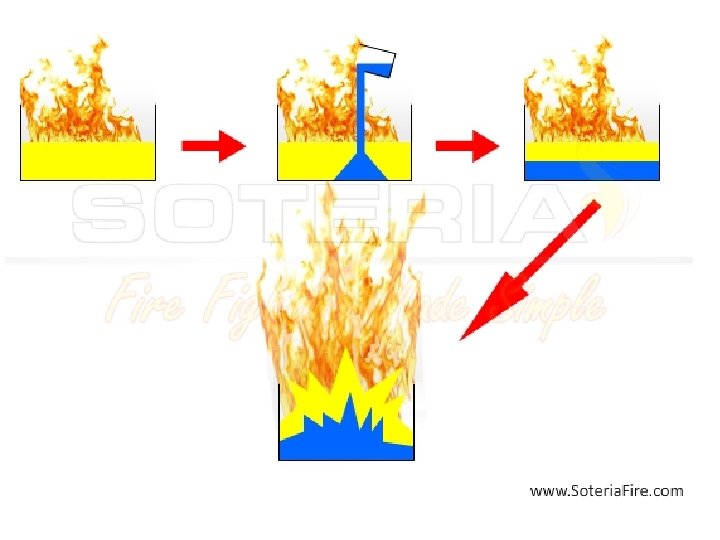

• The rapidly expanding steam (possibly superheated) expels the oil or fuel above upward and out of the tank, resulting in the discharging of burning oil onto a large and uncontrolled area outside of the container. • Boilover occures when three condidions exist simultaneously • open – tank fire, • water layer at the bottom of a tank, • over heating heat waves in a fuel.

Spillover or slop over • The slopover or spillover is a minor boil over and takes place in wet oil containing a percentage of water burning in a tank is full or nearly full and due to the sudden application of foam or spray from cooling branches falling upon the hot oil surface. • It is for this reason that when the oil level is within 3 meters of the top of the tank, great care should be taken when applying foam, specially when the oil is boiling. • Warning of the slop over is given by the lighter colour of the smoke at the top of the tank on the wind ward side owing to formation of steam, while a sizzling sound indicates wetness in oil.


Deep seated fire • It may be defined as a fire that has penetrated deep into bulk materials which is difficult to extinguish. • Fibrous materials, like wool, silk, cotton, jute etc will rapidly form deep seated fire. • A deep seated fire is one in which due to good thermal insulation the surface temperature of some of the fuel in the bulk of the material is sufficiently high for it to react with the oxygen that diffuses in, even though the oxygen is at very low concentrations.



BLEVE • There are three characteristics of liquids which are relevant to the discussion of a BLEVE • If a liquid in a sealed container is boiled, the pressure inside the container increases. As the liquid changes to a gas it expands - this expansion in a vented container would cause the gas and liquid to take up more space. In a sealed container the gas and liquid are not able to take up more space and so the pressure rises.

• Pressurized vessels containing liquids can reach an equilibrium where the liquid stops boiling and the pressure stops rising. This occurs when no more heat is being added to the system (either because it has reached ambient temperature or has had a heat source removed). • The boiling temperature of a liquid is dependent on pressure - high pressures will yield high boiling temperatures, and low pressures will yield low boiling temperatures.

• A common simple experiment is to place a cup of water in a vacuum chamber, and then reduce the pressure in the chamber until the water boils. By reducing the pressure the water will boil even at room temperature. This works both ways - if the pressure is increased beyond normal atmospheric pressures, the boiling of hot water could be suppressed far beyond normal temperatures. The cooling system of a modern internal combustion engine is a real-world example. • When a liquid boils it turns into a gas. The resulting gas takes up far more space than the liquid did.


• Typically, a BLEVE starts with a container of liquid which is held above its normal, atmosphericpressure boiling temperature. Many substances normally stored as liquids, such as. CO 2, propane, and other similar industrial gases have boiling temperatures, at atmospheric pressure, far below room temperature. • In the case of water, a BLEVE could occur if a pressurized chamber of water is heated far beyond the standard 100 °C (212 °F). That container, because the boiling water pressurizes it, is capable of holding liquid water at very high temperatures

• If the pressurized vessel, containing liquid at high temperature (which may be room temperature, depending on the substance) ruptures, the pressure which prevents the liquid from boiling is lost. If the rupture is catastrophic, where the vessel is immediately incapable of holding any pressure at all, then there suddenly exists a large mass of liquid which is at very high temperature and very low pressure. • This causes the entire volume of liquid to instantaneously boil, which in turn causes an extremely rapid expansion. Depending on temperatures, pressures and the substance involved, that expansion may be so rapid that it can be classified as an explosion, fully capable of inflicting severe damage on its surroundings


What Is Dust? • Dust is defined as “solid particles generated by handling, crushing, grinding, rapid impact, detonation, and decrepitation of organic or inorganic materials, such as rock, ore, metal, coal, wood, and grain. ” • A wide range of particle sizes is produced during a dust -generating process. Particles that are too large to remain airborne settle out, while the smallest ones remain suspended in air indefinitely. • Dusts are measured in micrometers (commonly known as microns or μm).

Dust explosion • Most finely divided combustible materials are hazardous. Finely divided dusts when suspended in air and ignited, they can cause several explosion. • Combustible dust may be classified as follows – Class I – this class comprises dust which ignite and propagate flame readily, the source of heat required for ignition being comparatively small. Eg lighted match stick, sugar, coco etc. – Class II- this class comprises dust which ignited but for the propagation of flame requires a source of heat of large size and high temperature( an electric arc) eg. Leather, saw dust, horn meal etc.

• Class III – this contains which ignite but do not appear to be capable of propagating flame under any condition likely to occur in a factory because. – They do not readily form a cloud of dust. – They Are contaminated with large quantities of non combustible materials. – The material of which they are composed, does not burn rapidly enough. Eg. tobacco. , cotton seed, charcoal etc.

Conditions required • A combustible dust • The dust is suspended in the air at a sufficiently high concentration • There is an oxidant (typically atmospheric oxygen) • There is an ignition source

Factors influencing dust explosion • • • The size of its particles Dust concentration Impurities present Oxygen concentration The strength of the source of ignition.

How Dust Explosions Occur • When all of these elements are in place, rapid combustion known as deflagration (a rapid burning slower than the speed of sound) can occur. If this event is confined by an enclosure, such as a building, room, vessel or process equipment, the resulting pressure rise can cause an explosion (a rapid burning faster than the speed of sound).

Secondary Explosions Are Catastrophic • An initial (primary) explosion (see Figure 2) in processing equipment or in areas where fugitive dust has accumulated may dislodge additional dust or damage a collection system (such as a duct, vessel or collector). This dust, if ignited, causes additional explosions, which can result in damage that is more severe than the original explosion due to increased concentrations and quantities of dispersed combustible dust.


Dust Control Some of the recommendations found in NFPA 654 for the control of dusts to prevent explosions include the following: • Minimize the escape of dust from process equipment or ventilation systems. • Use dust collection systems and filters. • Use surfaces that reduce dust accumulation and help with cleaning. • Provide access to all hidden areas to permit inspection.

• Conduct regular inspections for dust in open and hidden areas. • Clean dust residues at regular intervals. • Use cleaning methods that do not generate dust clouds if ignition sources are present (e. g. , do not use compressed air). • Use vacuum cleaners only if approved (e. g. , UL, FM) for combustible dust collection. • Locate relief valves away from dust hazard areas. • Develop and implement a written program for hazardous dust inspection, housekeeping and control. Ignition

Ignition Control NFPA 654 also contains guidance on the control of ignition sources to prevent dust explosions, including the following: Ø Use appropriate electrical equipment and wiring methods. Ø Control static electricity, including bonding of equipment to ground. Ø Control smoking, open flames and sparks. Ø Control friction and mechanical sparks. Ø Use separator devices to remove foreign materials capable of igniting combustibles from process materials. Ø Separate heated surfaces from dusts.

Unconfined Vapor Cloud Explosion (UCVE • In chemical industries, flammable liquids may be processed or stored at temperatures above their boiling points. A sudden uncontrolled release of such liquids in atmosphere can readily vaporize, creating an explosive mixture diffusing in air. • Fine mist from such a release remains suspended in air which readily exists in droplet sizes from 0. 5 microns to 10 microns. In variably such vapour are denser that air and drift over a wide area covering the plant, equipments etc.


• Where the releases have been massive, ignition occurs within 15 to 20 seconds from the periphery of the cloud. • Rapid flame propagation through vapour-air mixture in the form of a premixed flame travelling very rapidly through the cloud attains deflagration velocity(subsonic) with sever shockwave from blast, resulting widespread damages. • On the other hand, there are instances of such clouds becoming sufficiently diffused below flammable limits without any effect.

Human body exposure to heat and high temperature • An average man when seated in comfort zone of 25 deg and 50 percent relative humidity produces heat at the rate of 58. 2 W/m 2 by oxidation of food elements. This is equal to 100 W. • A normal healthy man has a capacity of producing 1200 watts of heat energy and dissipating it to atmosphere at the age of 20 years.

• It is observed that men feel comfortable up to 37 deg C. when body temperature rises more than 2 deg C above 37 deg C man begins to lose efficiency at normal metabolic rate itself. • Body temperature above 43 deg C may lethal if induced for more than few minutes.

Mans tolerance limits to high temperature can be related to his ability to withstand • Sensible to temperature rise • Loss of heat by regulatory sweating • Movement of heat from his body core to skin surface by blood circulation. • Sweating is the process which rescues human beings from heat strokes but sweating rates of more than 0. 0005 kg/sec cannot be produced.


eeling Ill ignite

Burn • A burn is a type of injury to skin , or other tissues, caused by heat, electricity, chemicals, friction, or radiation. Most burns are due to heat from hot liquids, solids, or fire. • Burns that affect only the superficial skin layers are known as superficial or firstdegree burns. • They appear red without blisters and pain typically lasts around three days.

• When the injury extends into some of the underlying skin layer, it is a partial-thickness or second-degree burn. Blisters are frequently present and they are often very painful. Healing can require up to eight weeks and scarring may occur. In a fullthickness or third-degree burn, the injury extends to all layers of the skin. • Often there is no pain and the burn area is stiff. Healing typically does not occur on its own. A fourth-degree burn additionally involves injury to deeper tissues, such as muscle, tendons, or bone.


- Slides: 43