Module 2 Organic Chemistry Section 1 Saturated Hydrocarbons

Module 2 Organic Chemistry Section 1: Saturated Hydrocarbons

Goals: Recognition of organic families. Naming of organic molecules. Synthesis of organic molecules. Reactions of organic compounds.

Organic Chemistry Organic chemistry is the study of carbon containing compounds. Over 85% of the 18 million known compounds are carbon compounds.

The Nature of Organic Molecules • Most organic compounds are poorly soluble in water. • Those that are soluble generally don’t conduct electricity. • Only small polar organic molecules or large molecules with many polar groups dissolve in water. • Lack of water solubility for organic compounds has important biological consequences. • A living cell is a water solution that contains many hundreds of different compounds. Cells use membranes composed of water-insoluble organic molecules to enclose their interiors and regulate flow of substances across the cell boundary.
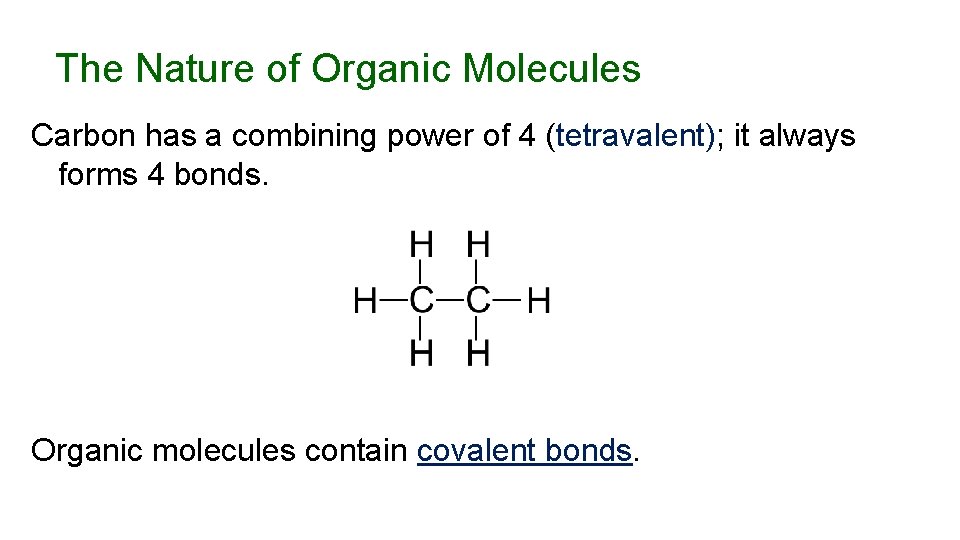
The Nature of Organic Molecules Carbon has a combining power of 4 (tetravalent); it always forms 4 bonds. Organic molecules contain covalent bonds.
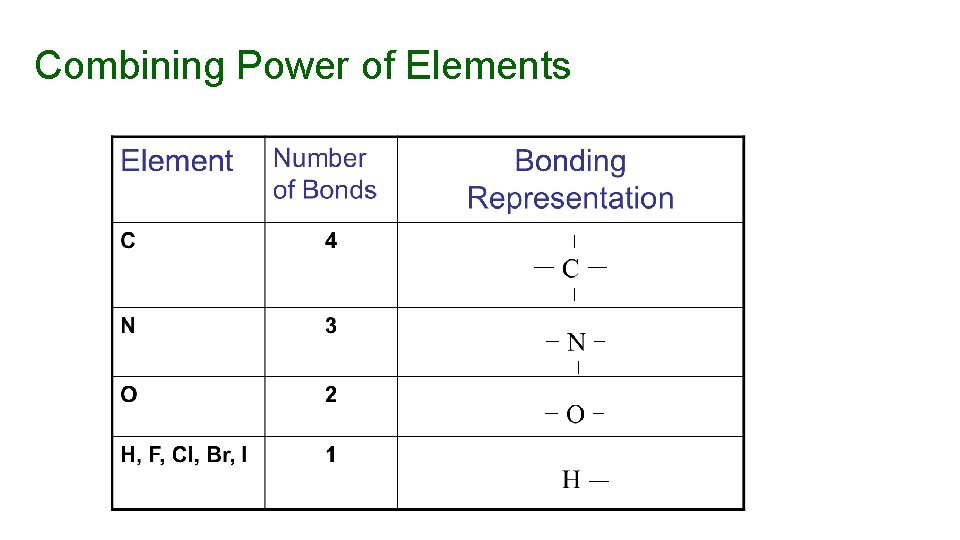
Combining Power of Elements
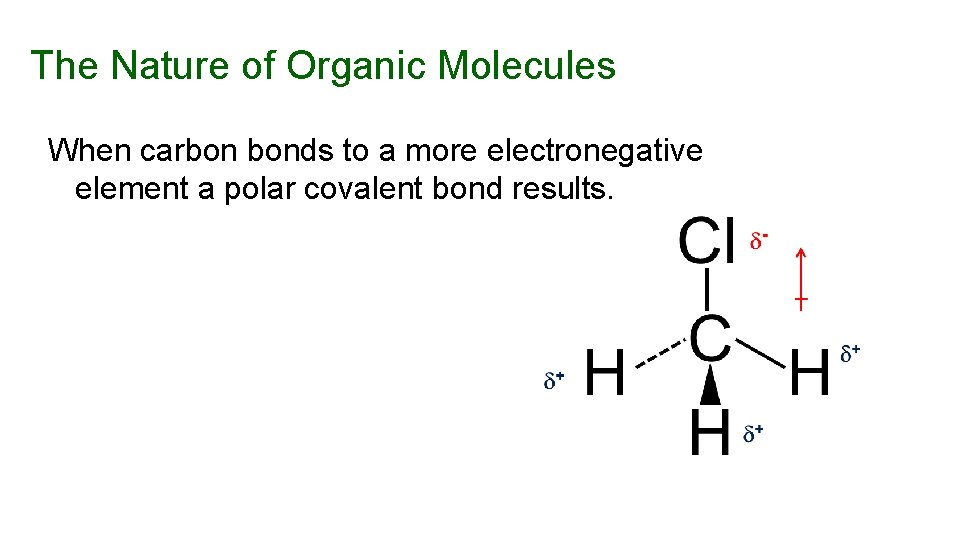
The Nature of Organic Molecules When carbon bonds to a more electronegative element a polar covalent bond results.
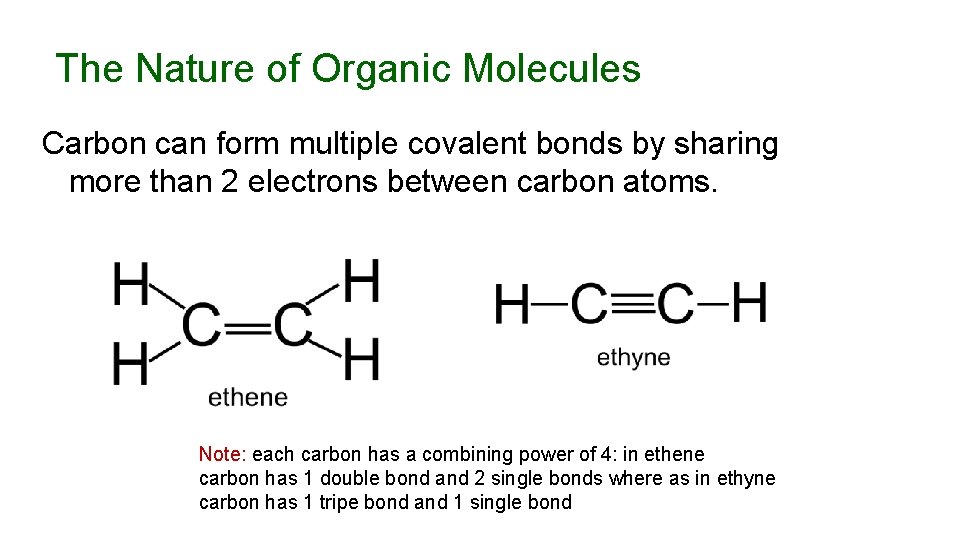
The Nature of Organic Molecules Carbon can form multiple covalent bonds by sharing more than 2 electrons between carbon atoms. Note: each carbon has a combining power of 4: in ethene carbon has 1 double bond and 2 single bonds where as in ethyne carbon has 1 tripe bond and 1 single bond

Hydrocarbons are a class of compounds containing hydrogen and carbon only. Four families of hydrocarbons – Alkanes, Alkenes, Alkynes and Aromatics
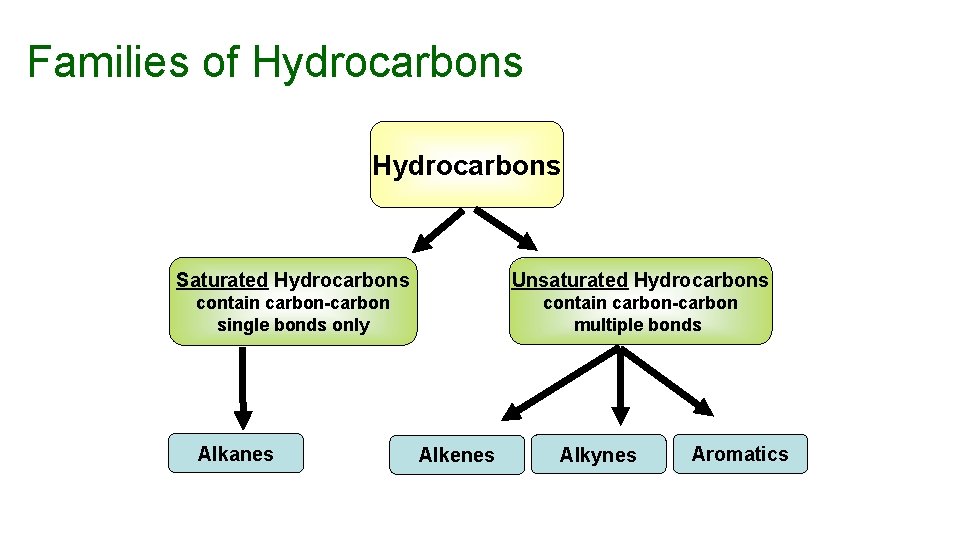
Families of Hydrocarbons Saturated Hydrocarbons Unsaturated Hydrocarbons contain carbon-carbon single bonds only contain carbon-carbon multiple bonds Alkanes Alkenes Alkynes Aromatics
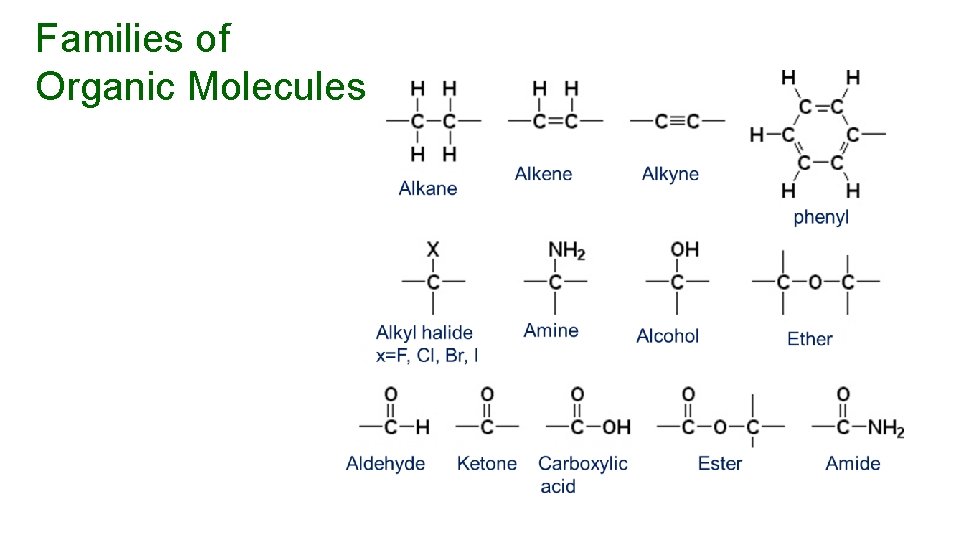
Families of Organic Molecules

Structural Formula – representation of a molecule that shows connectivity of atoms A structural formula cannot be correct (cannot represent a real compound) unless the combining power of each atom is represented correctly. Molecular formula – shows the ratio of atoms in a molecule A molecular formula is correct only if it can be translated into a correct structural formula.

Which of the following is a correct molecular formula? CH 2 F 2 Correct combining power for carbon CH 3 F 2 To many bonds to carbon CH 2 F Not enough bonds to carbon
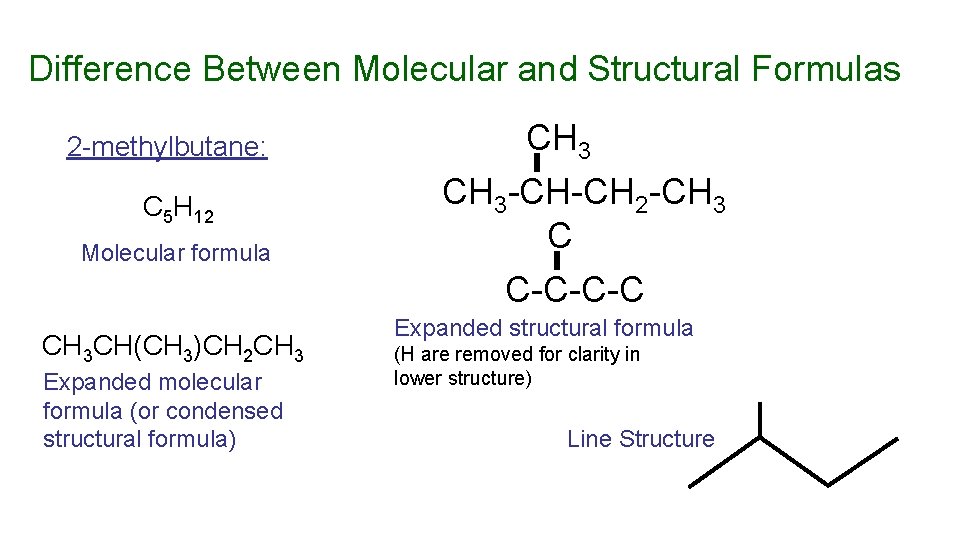
Difference Between Molecular and Structural Formulas 2 -methylbutane: C 5 H 12 Molecular formula CH 3 CH(CH 3)CH 2 CH 3 Expanded molecular formula (or condensed structural formula) CH 3 -CH-CH 2 -CH 3 C C-C-C-C Expanded structural formula (H are removed for clarity in lower structure) Line Structure
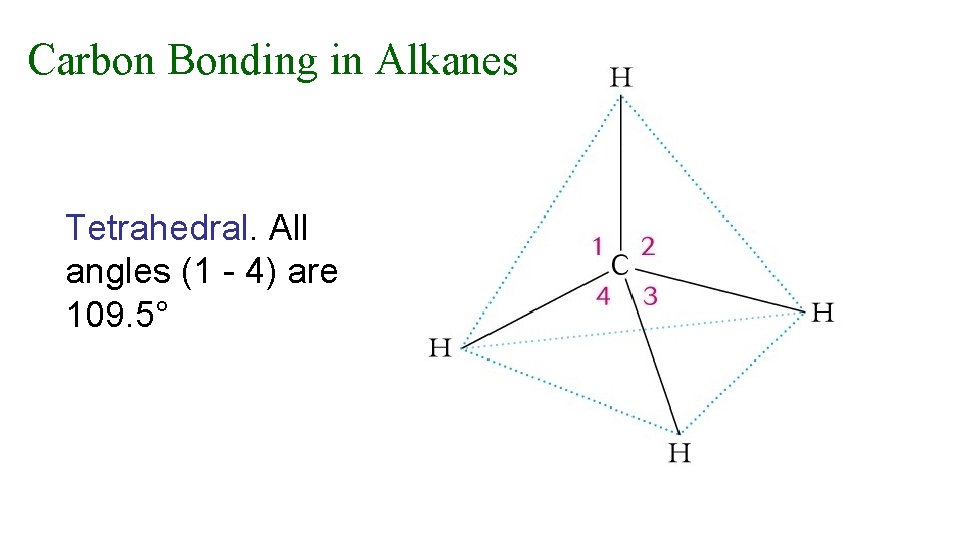
Carbon Bonding in Alkanes Tetrahedral. All angles (1 - 4) are 109. 5°

Alkanes - saturated hydrocarbons containing only carbon single bonds and have the general formula: Cn. H 2 n+2 C 2 H 6 Molecular formula Example: ethane H H H C C H H H Structural formula

First Ten Straight-Chain Alkane Hydrocarbons Know all these!

Structural Isomers of Alkanes Constitutional (Structural) Isomers- Two or more compounds with the same molecular formula but different arrangement of atoms (different bond connectivity). Isomers often differ in one or more physical and chemical properties such as boiling point, color, solubility, reactivity and density.
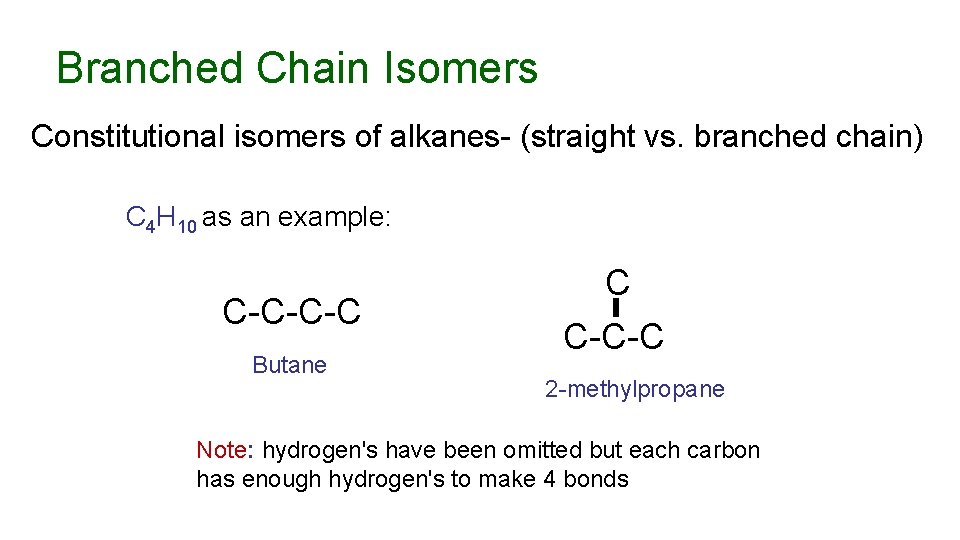
Branched Chain Isomers Constitutional isomers of alkanes- (straight vs. branched chain) C 4 H 10 as an example: C-C-C-C Butane C C-C-C 2 -methylpropane Note: hydrogen's have been omitted but each carbon has enough hydrogen's to make 4 bonds
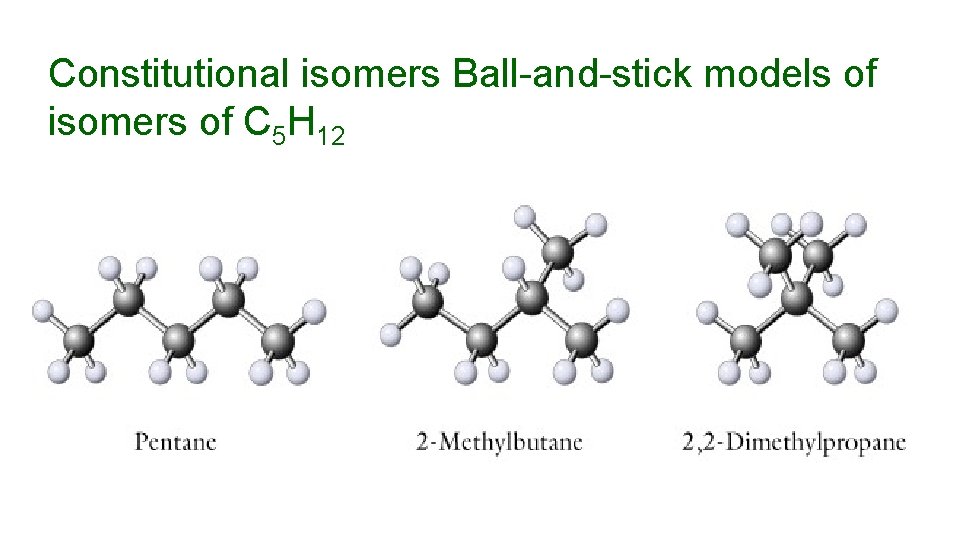
Constitutional isomers Ball-and-stick models of isomers of C 5 H 12
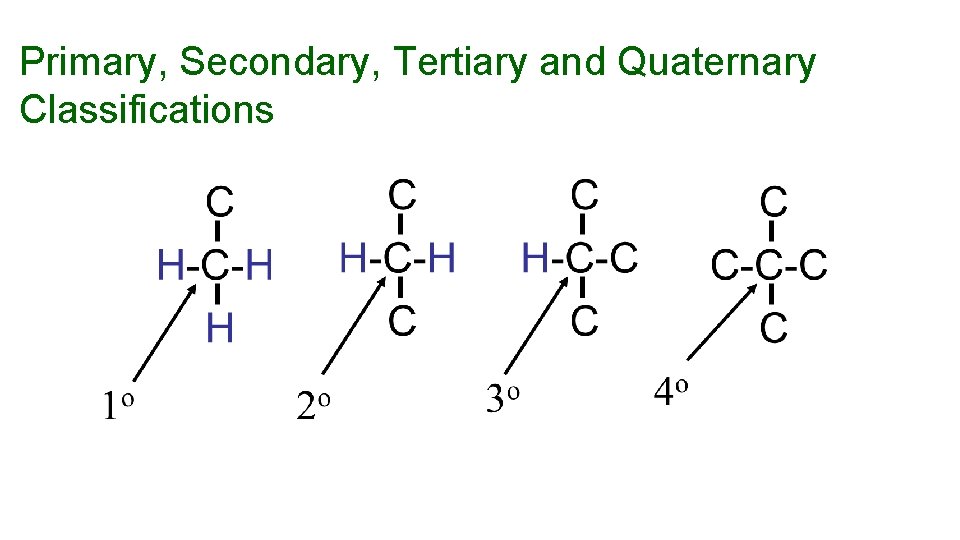
Primary, Secondary, Tertiary and Quaternary Classifications

Alkyl Groups Alkyl groups- are alkanes with a hydrogen atom removed and are bonded to a straight chain hydrocarbon. C C-C-C “A branched chain” or Alkyl group Straight chain
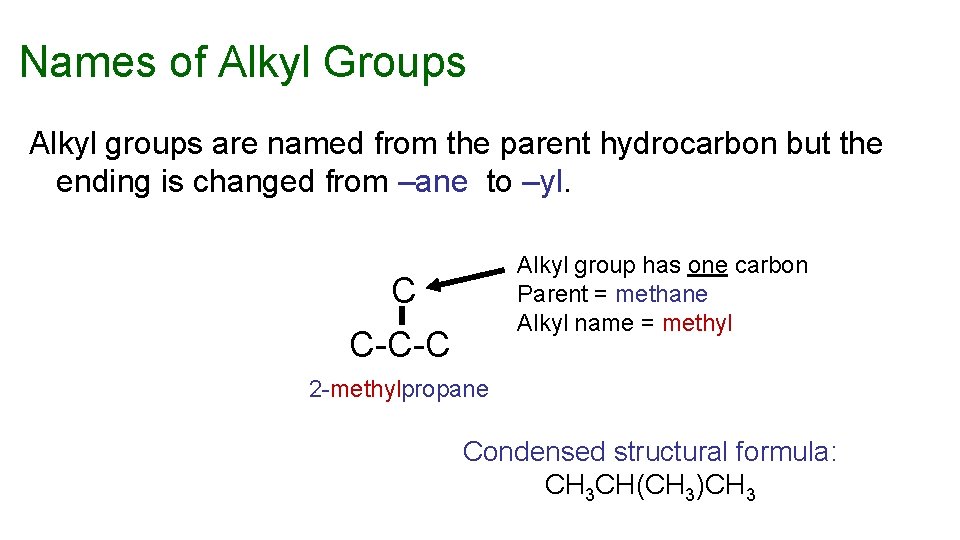
Names of Alkyl Groups Alkyl groups are named from the parent hydrocarbon but the ending is changed from –ane to –yl. Alkyl group has one carbon Parent = methane Alkyl name = methyl C C-C-C 2 -methylpropane Condensed structural formula: CH 3 CH(CH 3)CH 3


Naming Branched-Chain Alkanes – IUPAC system) 1) Identify the longest continous straight chain and name it as the parent or main chain. 2) Number the carbons in the main chain beginning at the end nearest the first branch point. 3) Name the attached alkyl groups and number them according to the point of attachment. 4) Write the final name as a single word. Hyphens separate #’s from prefixes, commas separate #’s, different substituent groups are ordered alphabetically, multiples of the same substituent identified by di- tri-, tetra-

Example of Naming Alkyl group 1 carbon = methyl C C-C-C 1 2 3 4 Longest chain 5 carbons = pentane 5 2 -methylpentane Carbon numbers
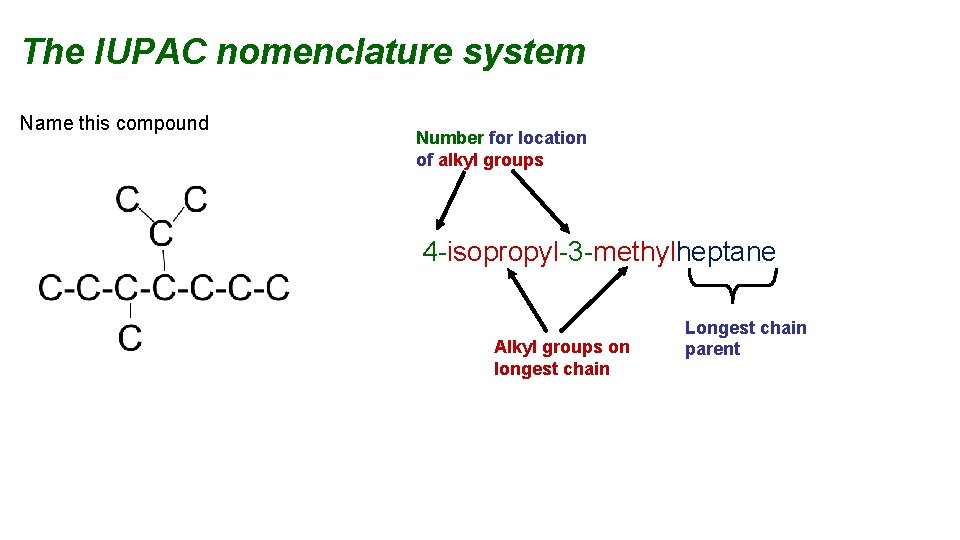
The IUPAC nomenclature system Name this compound Number for location of alkyl groups 4 -isopropyl-3 -methylheptane Alkyl groups on longest chain Longest chain parent
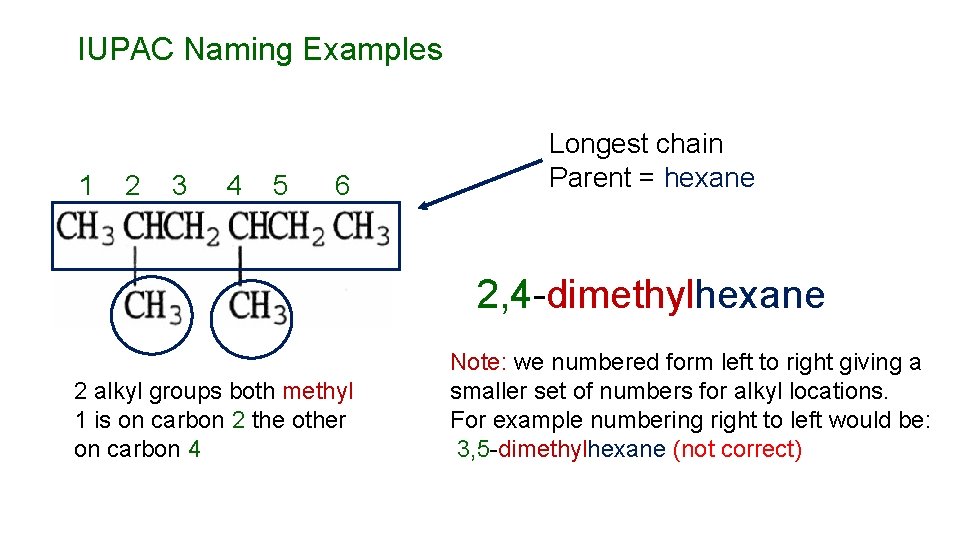
IUPAC Naming Examples 1 2 3 4 5 6 Longest chain Parent = hexane 2, 4 -dimethylhexane 2 alkyl groups both methyl 1 is on carbon 2 the other on carbon 4 Note: we numbered form left to right giving a smaller set of numbers for alkyl locations. For example numbering right to left would be: 3, 5 -dimethylhexane (not correct)
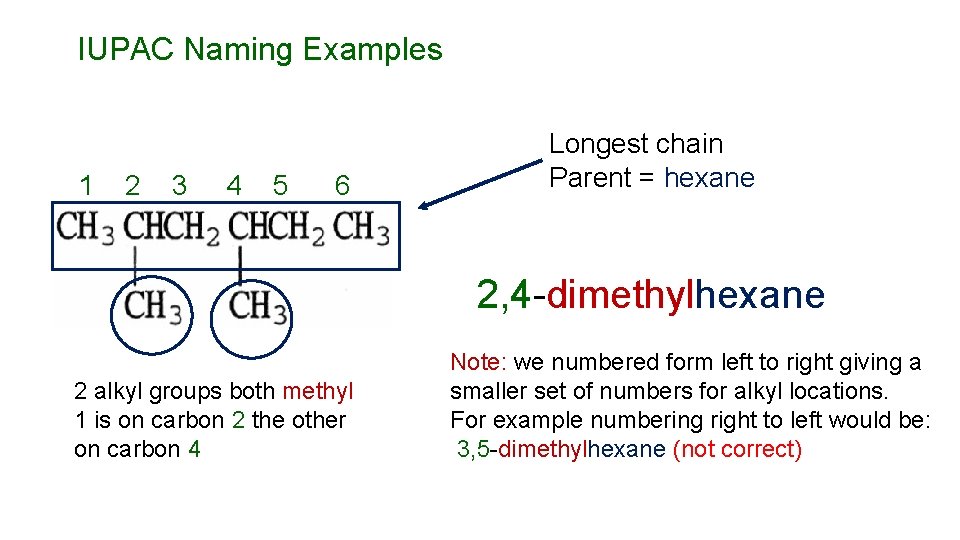
IUPAC Naming Examples 1 2 3 4 5 6 Longest chain Parent = hexane 2, 4 -dimethylhexane 2 alkyl groups both methyl 1 is on carbon 2 the other on carbon 4 Note: we numbered form left to right giving a smaller set of numbers for alkyl locations. For example numbering right to left would be: 3, 5 -dimethylhexane (not correct)
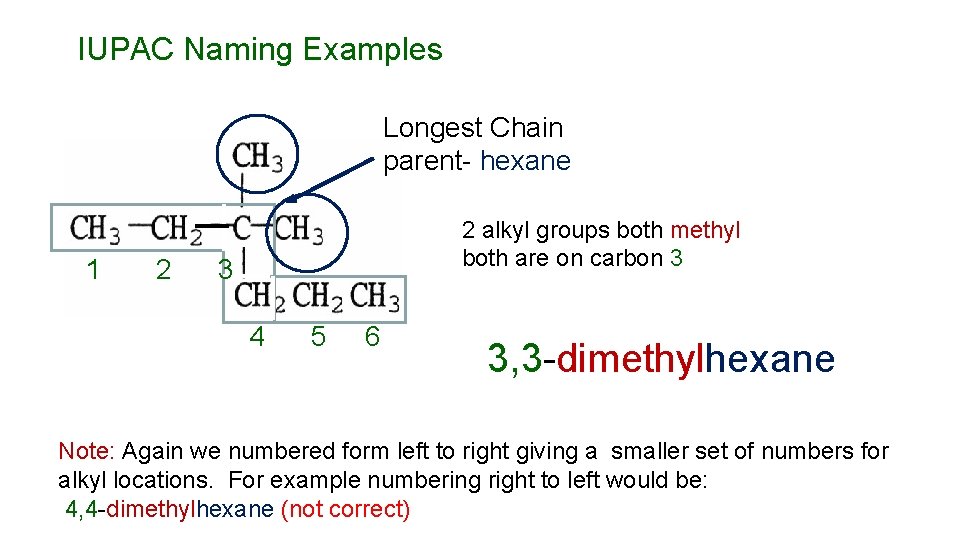
IUPAC Naming Examples Longest Chain parent- hexane 1 2 2 alkyl groups both methyl both are on carbon 3 3 4 5 6 3, 3 -dimethylhexane Note: Again we numbered form left to right giving a smaller set of numbers for alkyl locations. For example numbering right to left would be: 4, 4 -dimethylhexane (not correct)
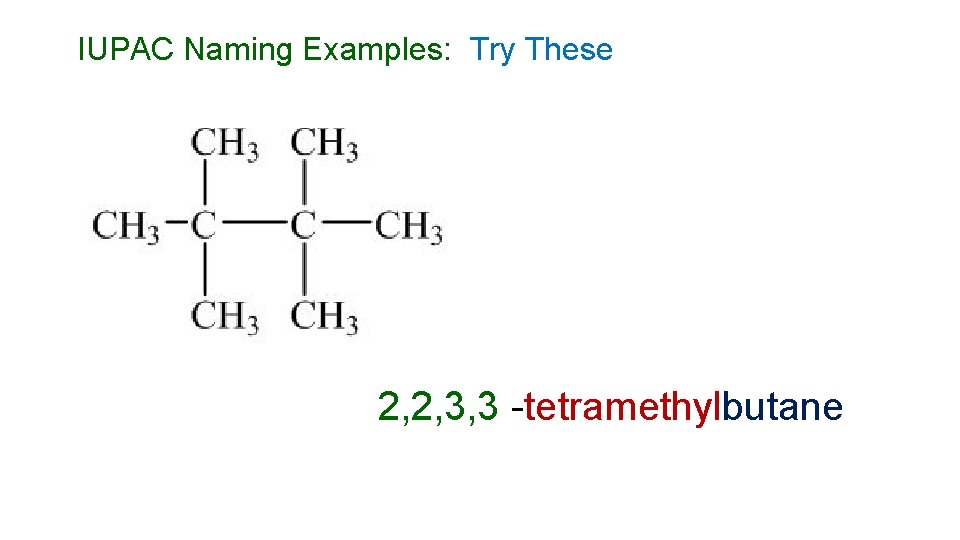
IUPAC Naming Examples: Try These 2, 2, 3, 3 -tetramethylbutane
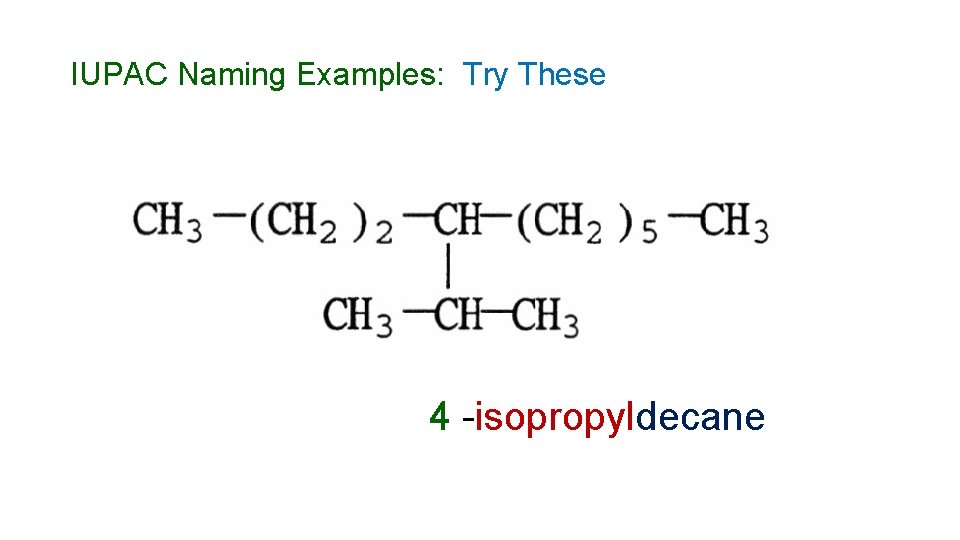
IUPAC Naming Examples: Try These 4 -isopropyldecane
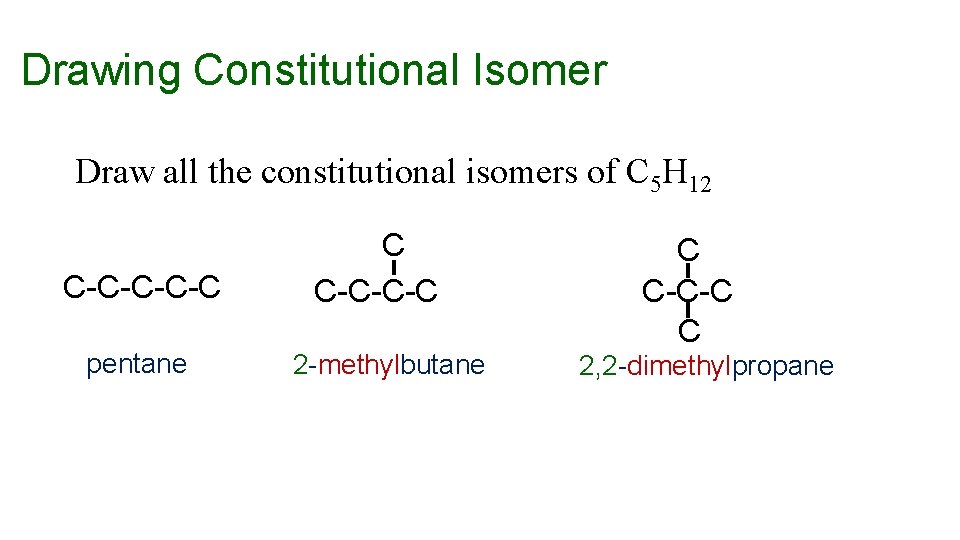
Drawing Constitutional Isomer Draw all the constitutional isomers of C 5 H 12 C-C-C pentane C C-C-C-C 2 -methylbutane C C-C-C C 2, 2 -dimethylpropane

Reactions of Alkanes Combustion reactions. Cn. H 2 n+2 + O 2 CO 2 + H 2 O Mono-halogenation - (halogen substitution) CH 4 + Cl 2 CH 3 Cl + HCl
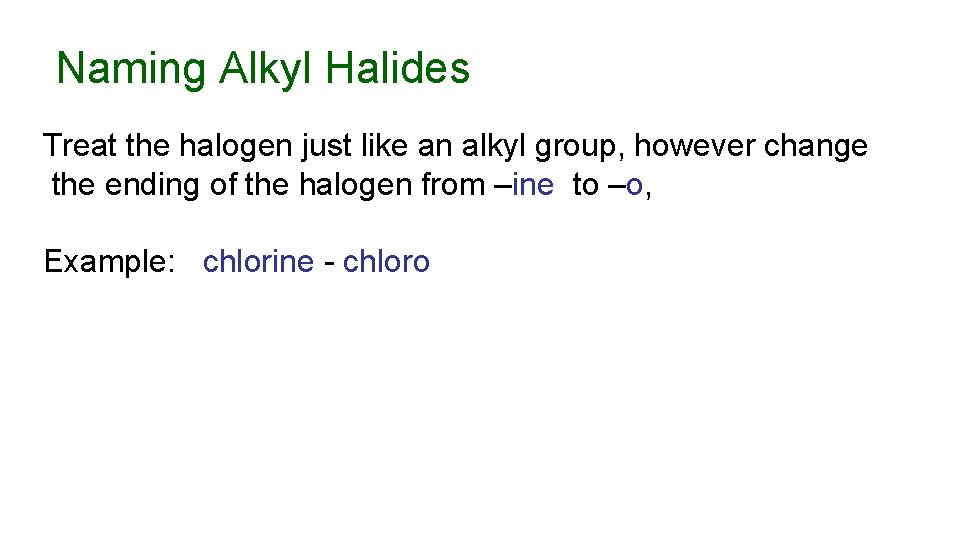
Naming Alkyl Halides Treat the halogen just like an alkyl group, however change the ending of the halogen from –ine to –o, Example: chlorine - chloro
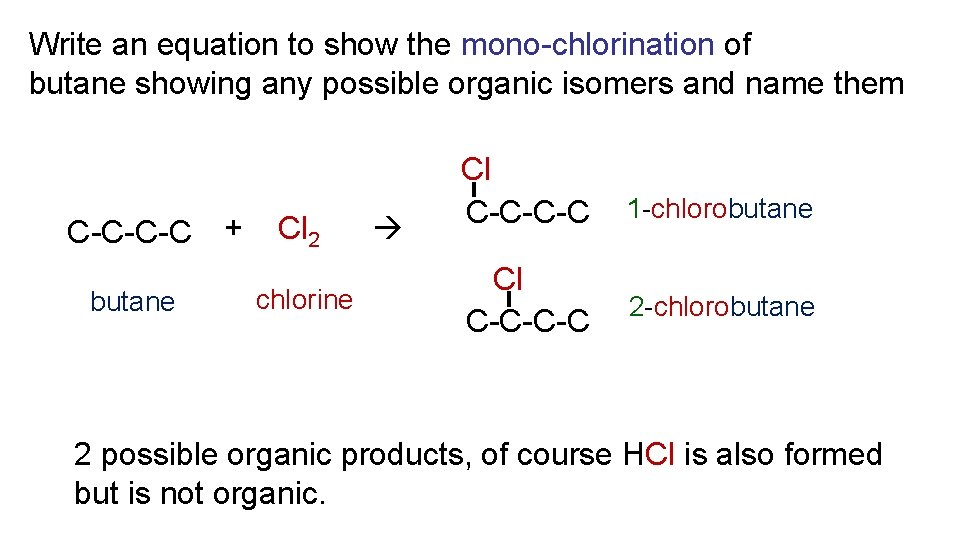
Write an equation to show the mono-chlorination of butane showing any possible organic isomers and name them C-C-C-C + butane Cl 2 chlorine Cl C-C-C-C 1 -chlorobutane 2 possible organic products, of course HCl is also formed but is not organic.
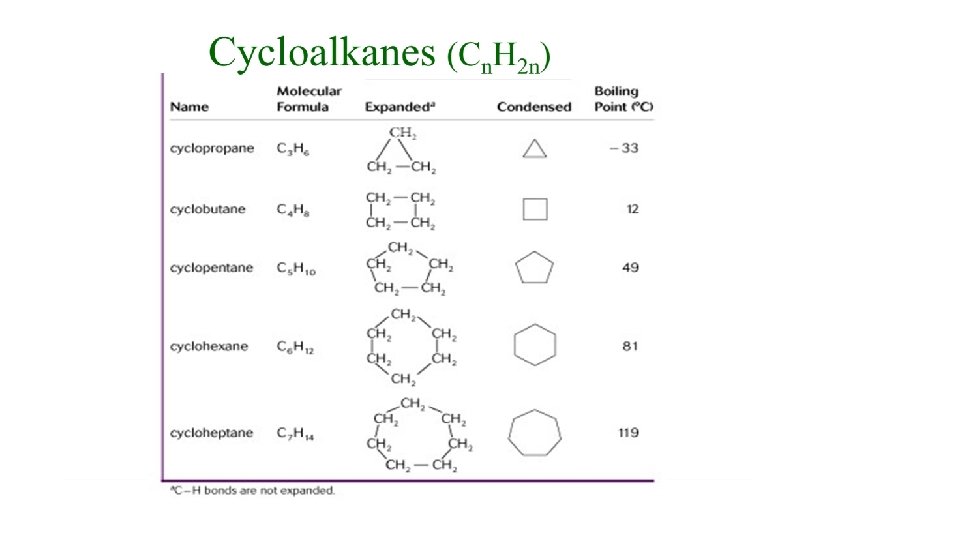

Naming Cycloalkanes (IUPAC) 1) Use the cycloalkane name as the parent. 2) Identify and number the substituents. Start numbering at the group with alphabetical priority (#1) and continue around ring in the direction that gives the second substituent the lowest number. 3) Names are written using the guidelines for branched chain alkanes.
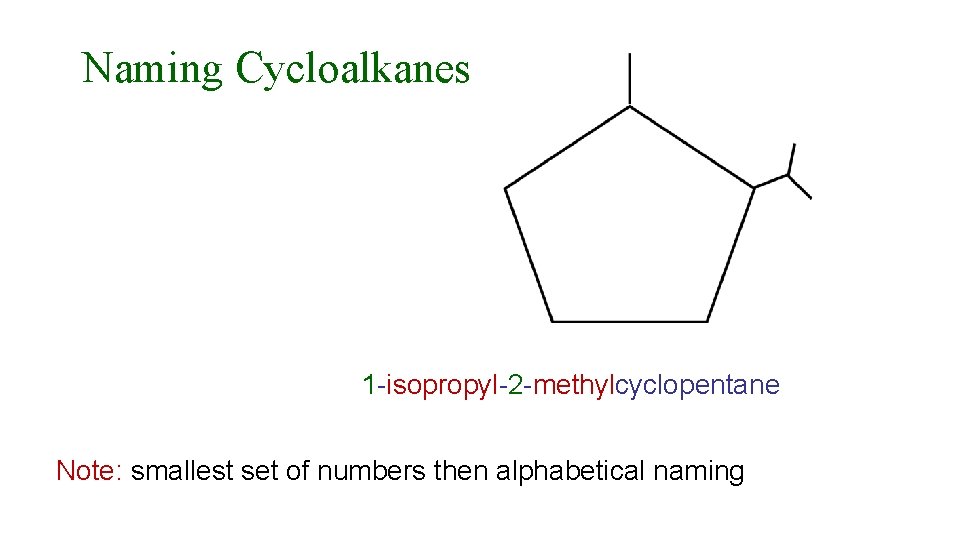
Naming Cycloalkanes 1 -isopropyl-2 -methylcyclopentane Note: smallest set of numbers then alphabetical naming

Cis-Trans Isomers of Cycloalkanes
- Slides: 40