Modern version of Mendeleevs Periodic table of the
Modern version of Mendeleev’s Periodic table of the elements
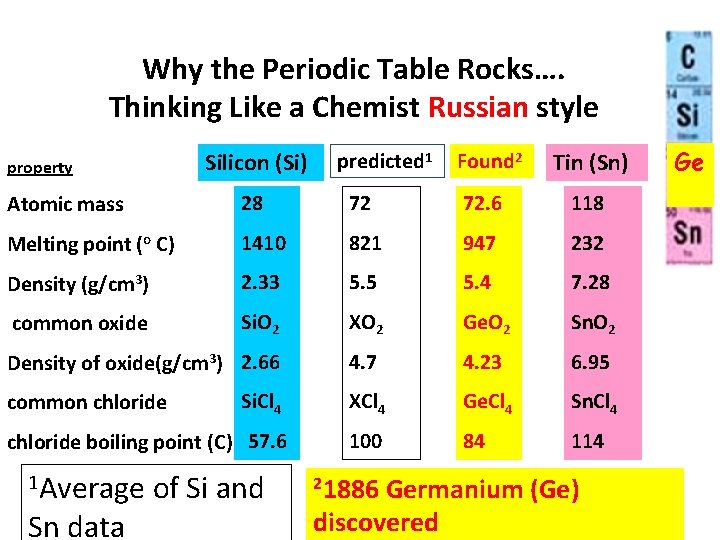
Why the Periodic Table Rocks…. Thinking Like a Chemist Russian style Silicon (Si) property predicted 1 Found 2 Tin (Sn) Atomic mass 28 72 72. 6 118 Melting point (o C) 1410 821 947 232 Density (g/cm 3) 2. 33 5. 5 5. 4 7. 28 common oxide Si. O 2 XO 2 Ge. O 2 Sn. O 2 Density of oxide(g/cm 3) 2. 66 4. 7 4. 23 6. 95 Si. Cl 4 XCl 4 Ge. Cl 4 Sn. Cl 4 chloride boiling point (C) 57. 6 100 84 114 common chloride 1 Average Sn data of Si and 21886 Germanium (Ge) discovered Ge ? ?

Another example… property Predicted for `eka-Aluminum Gallium 1 68 amu 69. 7 Low 30. 1 O C HIGH 1983 o. C Density (g/cm 3) 5. 91 common oxide X 20 3 Ga 2 O 3 Atomic mass Melting point C o Boiling point 1 o. C discovered in 1874 by Paul-Emile Lecoq de Boisbaudran
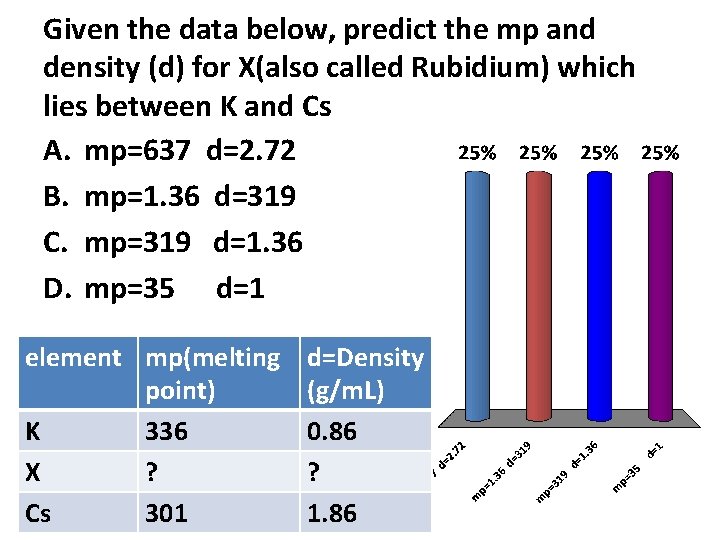
Given the data below, predict the mp and density (d) for X(also called Rubidium) which lies between K and Cs A. mp=637 d=2. 72 B. mp=1. 36 d=319 C. mp=319 d=1. 36 D. mp=35 d=1 element mp(melting point) K 336 X ? Cs 301 d=Density (g/m. L) 0. 86 ? 1. 86
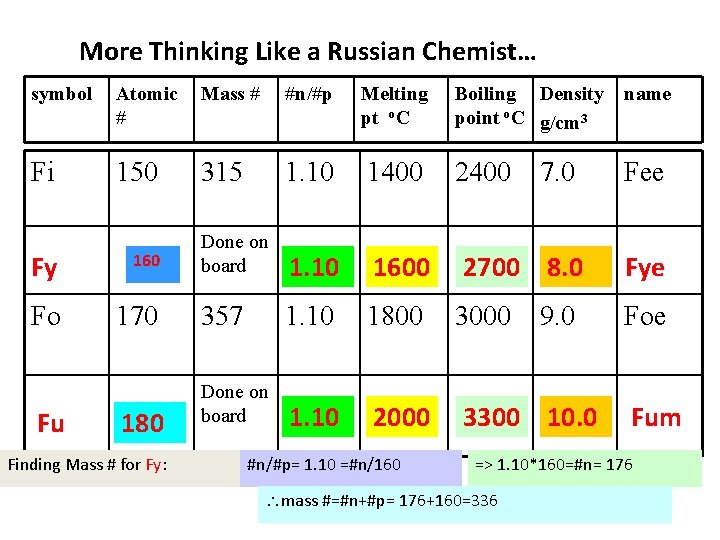
More Thinking Like a Russian Chemist… symbol Atomic # Mass # #n/#p Melting pt o. C Boiling Density point o. C g/cm 3 name Fi 150 315 1. 10 1400 2400 7. 0 Fee Fy 160 Done on board 1. 10 1600 2700 8. 0 Fye Fo 170 357 1. 10 1800 3000 Foe Done on board 1. 10 2000 3300 10. 0 Fu 180 Finding Mass # for Fy: #n/#p= 1. 10 =#n/160 9. 0 Fum => 1. 10*160=#n= 176 mass #=#n+#p= 176+160=336

Another example… property Predicted for `eka-Aluminum Gallium 1 68 amu 69. 7 Low 30. 1 O C HIGH 1983 o. C Density (g/cm 3) 5. 91 common oxide X 20 3 Ga 2 O 3 Atomic mass Melting point C o Boiling point 1 o. C discovered in 1874 by Paul-Emile Lecoq de Boisbaudran

Example of `periodicity’: halogens p mass X MP (OC) BP ( O C) 3 7 -220 -188 11 23 -101 -35 Halogen chemistry 19 40 -7 59 • 2 Na + X 2 2 Na. X (high melting white salts) 37 85 114 184 55 133 302 337 • X 2(colored) + ethene colorless dihalides MP and BP very different • Breathing X 2 kills you
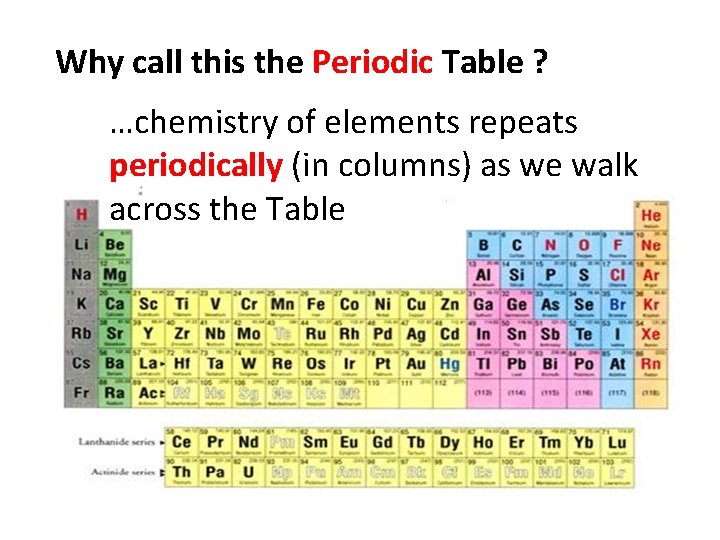
Why call this the Periodic Table ? …chemistry of elements repeats periodically (in columns) as we walk across the Table

Another example of periodicity inert (noble) gases p mass 2 4 MP(o C) - BP (0 C ) 272 - 269 10 20 -249 -246 18 40 -189 -186 36 84 -157 -152 54 131 -112 -107 86 222 -71 -62 MP ~ BP Noble gas chemistry NONE
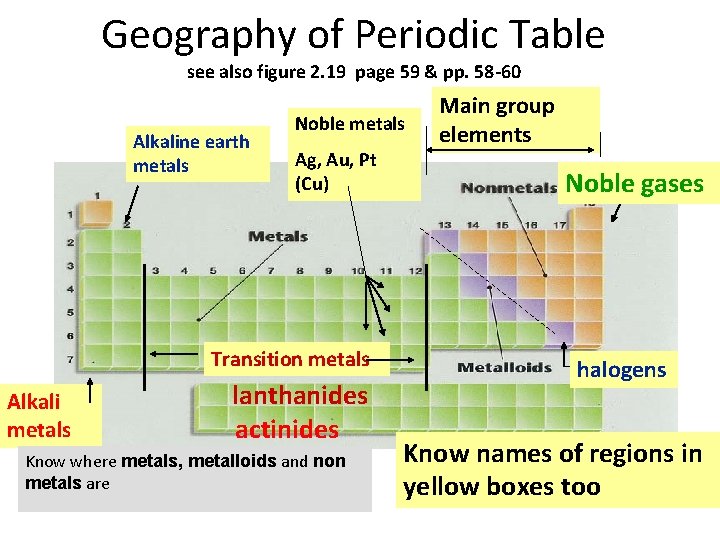
Geography of Periodic Table see also figure 2. 19 page 59 & pp. 58 -60 Alkaline earth metals Noble metals Ag, Au, Pt (Cu) Transition metals Alkali metals lanthanides actinides Know where metals, metalloids and non metals are Main group elements Noble gases halogens Know names of regions in yellow boxes too
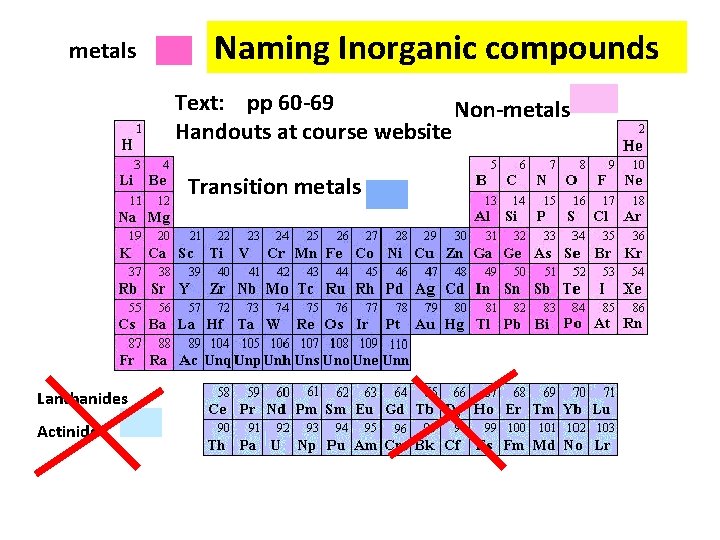
metals Naming Inorganic compounds Text: pp 60 -69 Non-metals Handouts at course website Transition metals Lanthanides Actinide

Binary compound naming 1. Non-Metal + Non-Metal = NM+NM 2. Metal + Non-Metal = M + NM 3. Transition Metal + Non-Metal = TM + NM Why three different naming patterns …. because want name pattern to reflect different chemistry for each kind of compound above
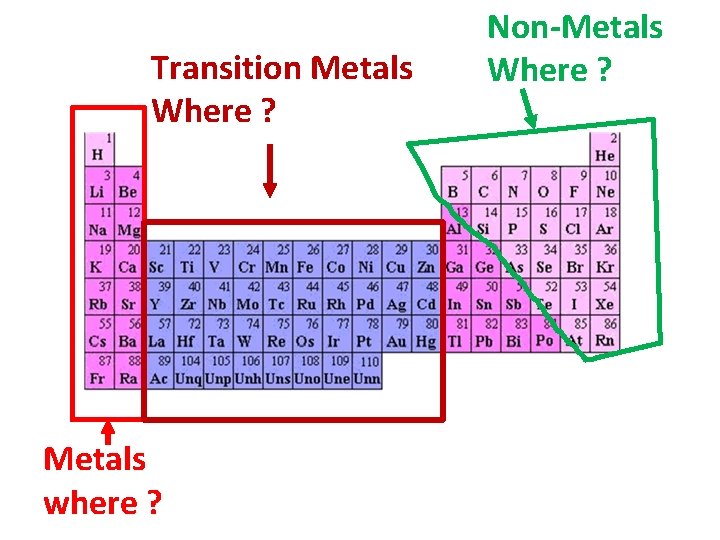
Transition Metals Where ? Metals where ? Non-Metals Where ?
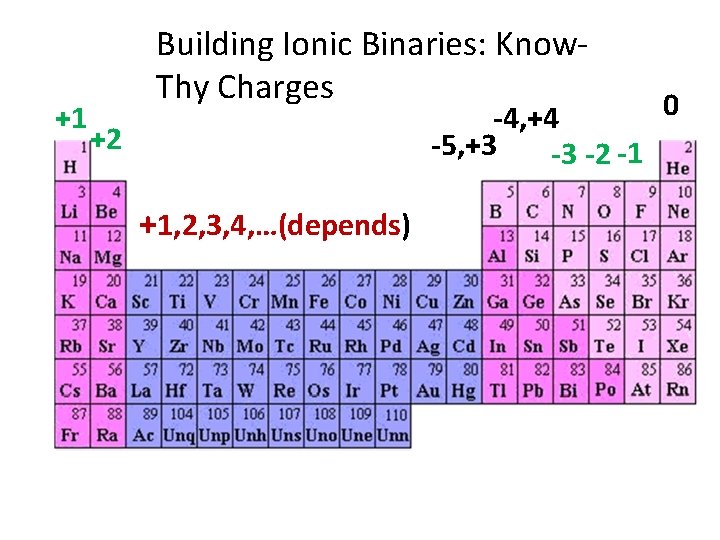
+1 Building Ionic Binaries: Know. Thy Charges 0 -4, +4 -5, +3 -3 -2 -1 +2 +1, 2, 3, 4, …(depends)
- Slides: 14