Modern Theory of the Atom Quantum Mechanical Model
















- Slides: 24

Modern Theory of the Atom: Quantum Mechanical Model


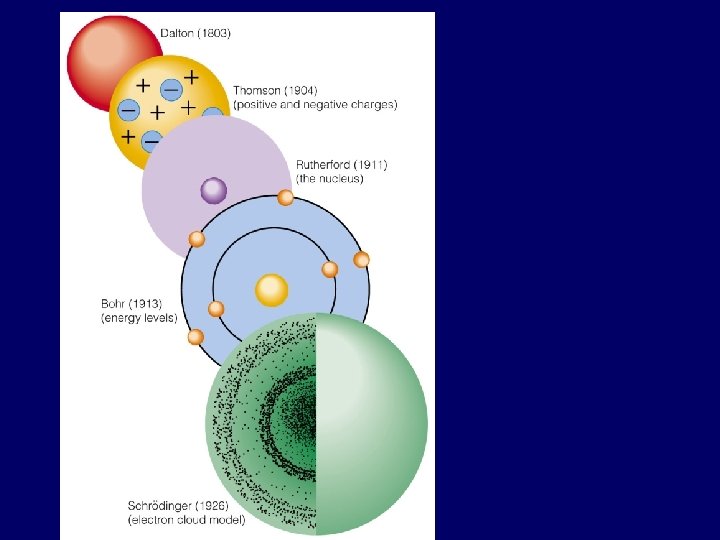
Recap of Bohr Model electrons: particles moving in circular orbits with specific speed, position, & energy levels possess specific quantum of energy electrons can move between energy levels higher energy levels farther from nucleus • e- moving up to higher E level: electron absorbs energy • e- moving down to lower E level: electron emits light energy ground state: electrons located in lowest possible energy levels, closest can be to nucleus

Bohr Model vs. Modern Theory • electron = particle • e- path is orbit – holds 2 n 2 electrons • circular path • each energy level has specific energy • can find exact position/ speed of e- • electron = wave • e– path is orbital – holds 2 electrons • not necessarily circular • each energy level has specific energy • probable location of e-

Orbital – Modern Theory • orbital: term describes region where e- might be found • each orbital: – specific energy & specific shape – described by 4 parameters of wave function (like an address) • quantum numbers = n, l, m, s • structure of orbitals explain: – bonding, magnetism, atom size, crystal structure

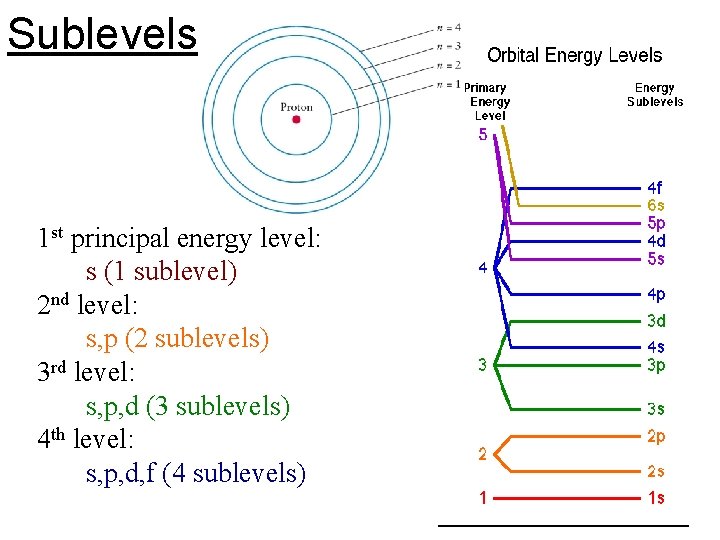
n: principal quantum number • specifies atom’s principal energy levels • whole number values: 1, 2, 3, 4, … • 2 n 2 = maximum # e- in any principal energy level

l= describes sublevels • sublevels are labelled by shape: –s, p, d, f

s orbitals: spherical

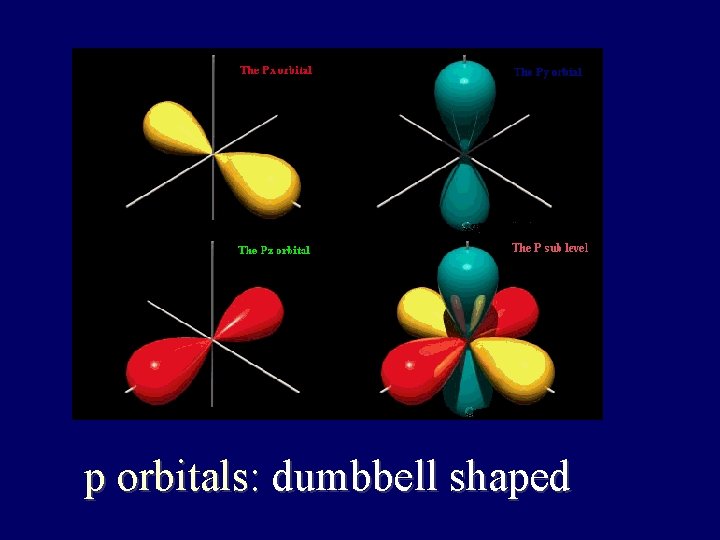
p orbitals: dumbbell shaped

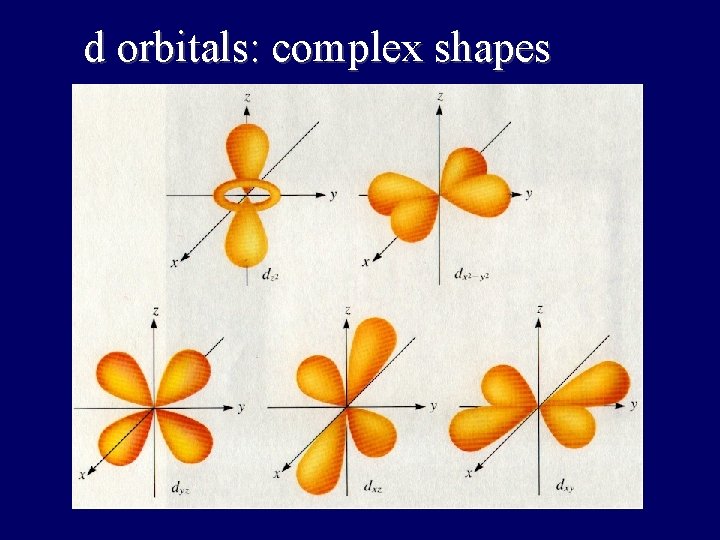
d orbitals: complex shapes

f orbitals: complex shapes too

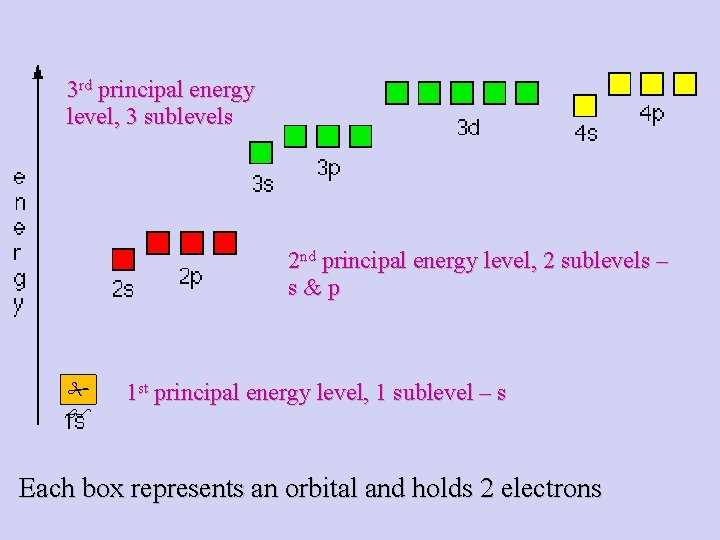
Sublevels 1 st principal energy level: s (1 sublevel) 2 nd level: s, p (2 sublevels) 3 rd level: s, p, d (3 sublevels) 4 th level: s, p, d, f (4 sublevels)

m = 3 rd quantum number (orbitals) • each sublevel contains 1 or more orbitals • each orbital holds a max of 2 electrons • • s has 1 orbital p has 3 orbitals d has 5 orbitals f has 7 orbitals 2 electrons) 1 st PEL =s (1 sublevel) = 1 orbital (__ 2 nd PEL =s, p (2 sublevels) = 4 orbitals (__8 e-) 3 rd PEL = s, p, d (3 sublevels) = 9 orbitals (___ 18 e-) 32 e-) 4 th PEL s, p, d, f (4 sublevels) = 16 orbitals (___

th 4 quantum number = s • e- spin: 2 possible values – clockwise and counter clockwise – Illustrated by arrows with opposite directions

address for each electron • • 4 quantum numbers no 2 e- can – occupy same space in atom – have same 4 quantum numbers max 2 electrons per orbital (Pauli exclusion principle)

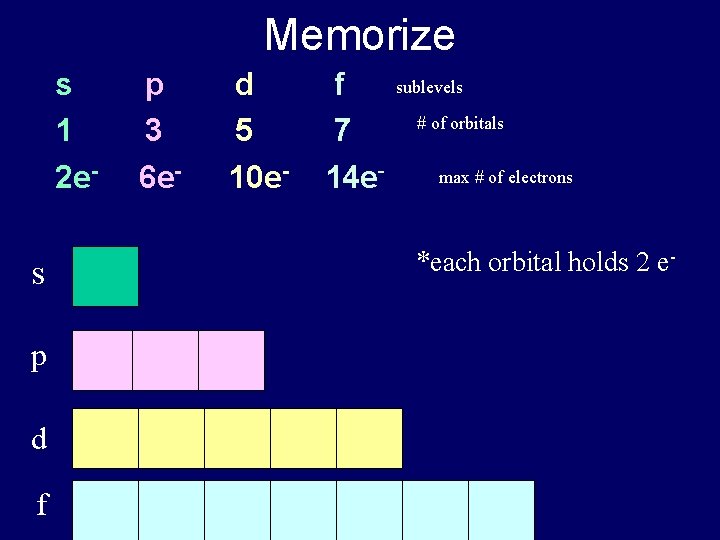
Memorize s 1 2 e- s p d f p 3 6 e- d 5 10 e- f 7 14 e- sublevels # of orbitals max # of electrons *each orbital holds 2 e-

electron configurations • add e- to atoms starting with lowest energy levels (most stable) – start with 1 s, then work upward increasing in energy • follow Aufbau Principle.

3 rd principal energy level, 3 sublevels 2 nd principal energy level, 2 sublevels – s&p 1 st principal energy level, 1 sublevel – s Each box represents an orbital and holds 2 electrons

Aufbau Principle 1 s 2 s 3 s 4 s 5 s 6 s 7 s follow arrows 2 p 3 p 4 p 5 p 6 p 7 p 3 d 4 d 5 d 6 d 7 d 4 f 5 f 6 f 7 f sequence of orbitals: 1 s, 2 p, 3 s, 3 p, 4 s, 3 d, 4 p, 5 s, 4 d, … exceptions do occur: - half-filled orbitals have extra stability - magic # is 8

2 1 s He 2 s 2 2 p 6 3 s 2 3 p 6 3 d 10 2 2 s 2 2 p 2 1 s C 4 s 2 4 p 6 4 d 10 4 f 14 5 s 2 5 p 6 5 d 10 5 f 14 2 2 s 2 2 p 6 3 s 2 Mg 1 s 6 s 2 6 p 6 6 d 10 7 s 2 7 p 6 7 d 10 Zn 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10

from modern configurations can figure out Bohr configurations: add e- in each energy level (shell) He 1 s C 2 1=2 2 1 s 22 p 2 1=2 2 = 2+2 2– 4 Mg Zn 1 s 22 p 63 s 2 1=2 2 = 2+6 3=2 2– 8– 2 1 s 22 p 63 s 23 p 64 s 23 d 10 1=2 2 = 2+6 3 = 2+6+10 2 – 8 – 18 – 2 4=2

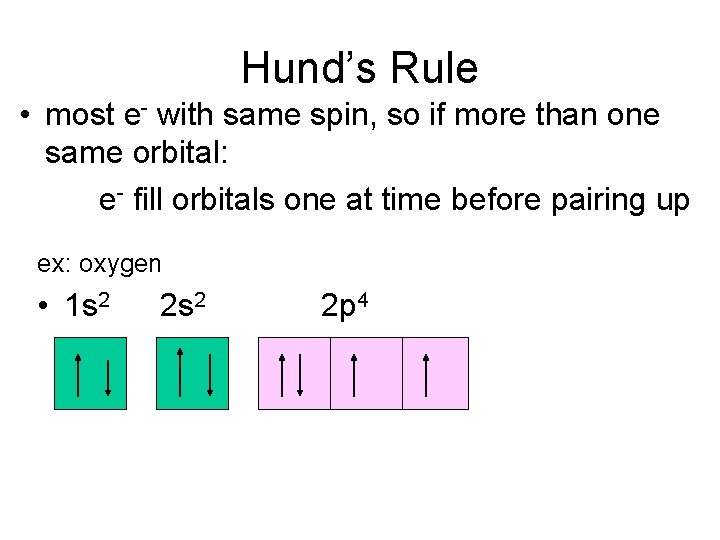
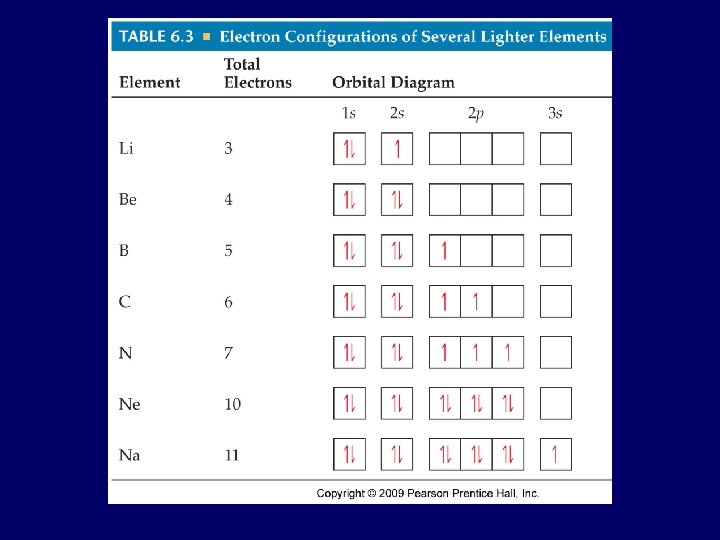
Hund’s Rule • most e- with same spin, so if more than one same orbital: e- fill orbitals one at time before pairing up ex: oxygen • 1 s 2 2 p 4


Which element? boron How many unpaired e-? 1 How many principal energy level’s occupied? How many principal energy level’s are fully occupied? 1 How many sublevels contain e-? 3 How many sublevels full? 2 How many orbitals contain e-? 3 2