Modern Theories of Acids Bases The Arrhenius and

Modern Theories of Acids & Bases The Arrhenius and Bronsted-Lowry Theories

Arrhenius Acids Svente Arrhenius (who first proposed theory of ionization) identified acids as substances that ionize in water to produce hydronium ion. For example: HCl + H 2 O H 3 O+ + Cl. Any substance that ionizes in water to produce hydronium ion can be classified as an acid.

Arrhenius Base Svente Arrhenius also identified bases as substances that ionize in water to produce hydroxide ion. For example: Na. OH Na+ + OHAny substance that ionizes in water to produce hydroxide ion can be classified as a base.

Salts Ionic substances that break up in solution to produce ions other than hydronium and hydroxide ions. Na. Cl (s) Na+(aq) + Cl- (aq) KNO 3(s) K+ (aq) + NO 3 - (aq) Li 2 SO 4 (s) 2 Li+ (aq) + SO 42 -(aq) Salts are made up of positive (metal) and negative (nonmetallic or polyatomic) ions. The more familiar you become with Table E, the easier it will be for you to identify salts.

Practice Identify each of the following as acids/bases/salts: 1. 2. 3. 4. 5. HC 2 H 3 O 2 K 2 SO 4 KOH Li. OH HNO 3 Acid HC 2 H 3 O 2 H+ + C 2 H 3 O 2 Salt K 2 SO 4 2 K+ + SO 42 Base KOH K+ + OHBase Li. OH Li+ + OHAcid HNO 3 H+ + NO 3 -

Bronsted-Lowry Acids & Bases Another theory of acids & bases

Not everyone was happy with Arrhenius’s definition A major problem with the Arrhenius definition of acids and bases is that it limits acids and bases to water (aqueous) solutions. Since an acid requires water to ionize and form hydronium ions, there can be no Arrhenius acids unless water is involved as the solvent.
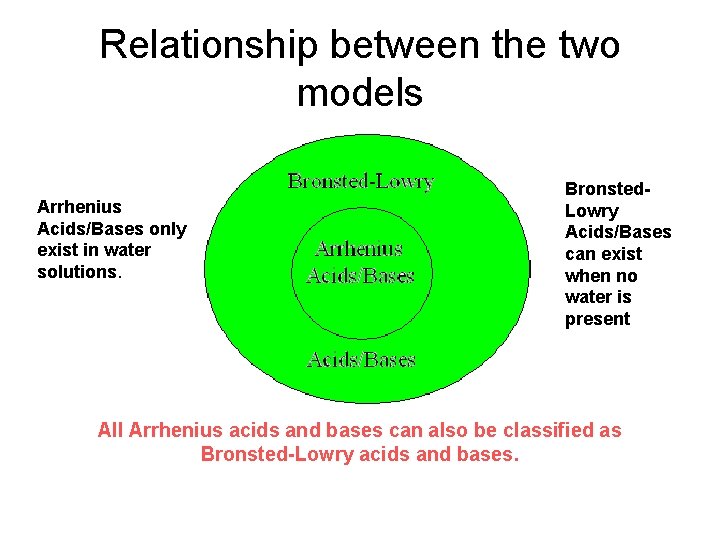
Relationship between the two models Arrhenius Acids/Bases only exist in water solutions. Bronsted. Lowry Acids/Bases can exist when no water is present All Arrhenius acids and bases can also be classified as Bronsted-Lowry acids and bases.

Bronsted-Lowry Definitions Bronsted and Lowry felt that this was too limiting, since there are many non-aqueous systems (no water is present). They came up with the following definitions for acids and bases. An acid is a proton (H+ ion) donor A base is a proton acceptor

An example In the reaction below there are no Arrhenius acids or bases present (because no hydronium ions or hydroxide ions are formed). However, the HCl is acting as a Bronsted. Lowry acid because it is giving a H+ ion to the NH 3 (which is acting as a H+ ion acceptor - a base)
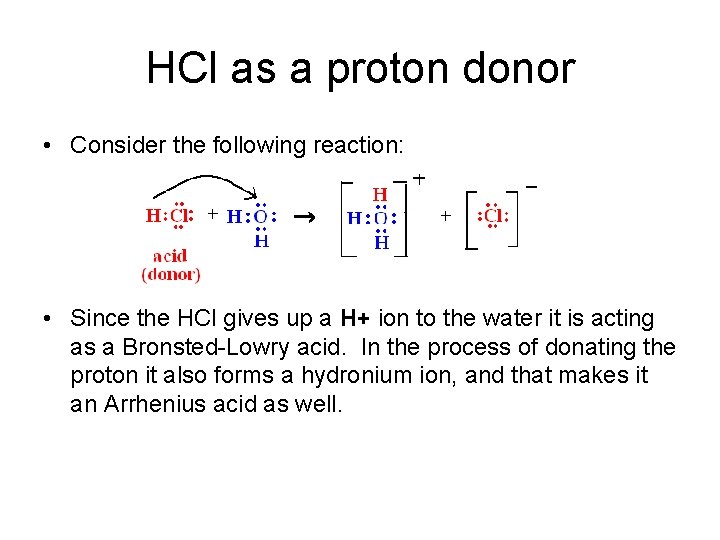
HCl as a proton donor • Consider the following reaction: • Since the HCl gives up a H+ ion to the water it is acting as a Bronsted-Lowry acid. In the process of donating the proton it also forms a hydronium ion, and that makes it an Arrhenius acid as well.
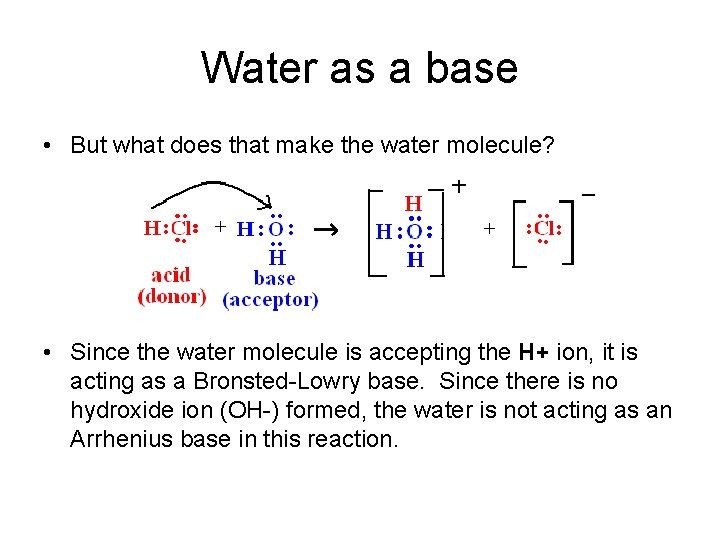
Water as a base • But what does that make the water molecule? • Since the water molecule is accepting the H+ ion, it is acting as a Bronsted-Lowry base. Since there is no hydroxide ion (OH-) formed, the water is not acting as an Arrhenius base in this reaction.

Ammonia as a base • Let’s look at another example: • Here the ammonia molecule is accepting a H+ ion and therefore is acting as an Bronsted-Lowry base. However, in the process of reacting with the water it is also forming a hydroxide ion. That makes the ammonia an Arrhenius base as well.

But what about the water? Since the water is giving up a H+ ion, it is acting as a Bronsted-Lowry acid. Since it does not form hydronium ions, it is NOT acting an Arrhenius acid.

Amphiprotic • Sometimes a molecule can donate a proton (act as an acid) and sometimes it can accept a proton (act as a base). • Molecules that have this ability to act as both an acid and a base are called amphoteric or amphiprotic. • Water is the most common example of an amphoteric substance.
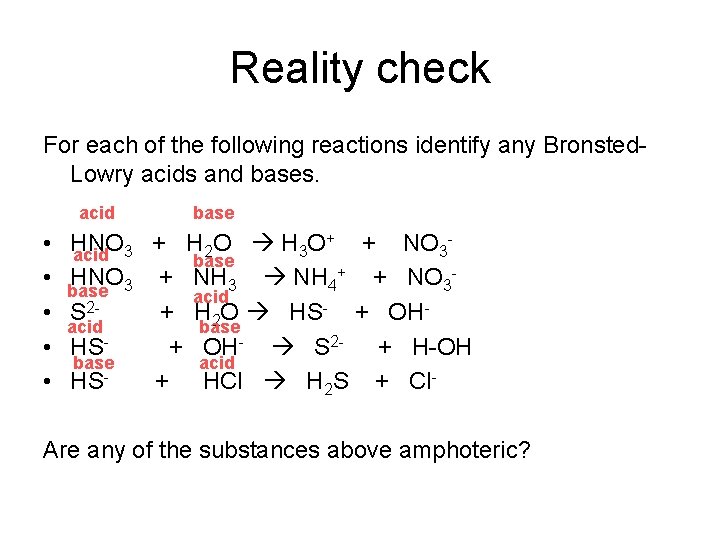
Reality check For each of the following reactions identify any Bronsted. Lowry acids and bases. acid • • • base + HNO + H O + NO 2 3 3 acid 3 base + HNO + NH + NO 3 3 4 3 base acid 2 S + H 2 O HS- + OHacid base HS + OH- S 2 - + H-OH base acid HS + HCl H 2 S + Cl- Are any of the substances above amphoteric?

Acid-base equilibrium Many acid base reactions go to equilibrium, that is they have both a forward and reverse reactions For instance, acetic acid (HC 2 H 3 O 2) reacts with water to form hydronium ion and acetate ion.

The reverse reaction However, the acetic acid only partially ionizes because a reverse reaction takes place preventing the forward reaction from reaching completion. In the reverse reaction, the Hydronium ion acts as an acid (a proton donor) while the acetate ion acts as the base.

Conjugate Acid-Bases • When a substance donates a proton, the substance that is left is its conjugative base: • Notice that the conjugative base is accepting a proton in the reverse reaction. Every acid has a conjugative base

Conjugative Acid-Base Pairs 2 • When a substance accepts a proton, the substance that is formed is its conjugative acid: • Notice that the conjugative acid is donating a proton in the reverse reaction. Every base has a conjugative acid
- Slides: 20