Modern Theories of Acids Bases The Arrhenius and

Modern Theories of Acids & Bases The Arrhenius and Bronsted-Lowry Theories From website produced at Charles W Baker High School Baldwinsville, Onondaga County New York

Acids & Bases • Acids and bases are special kinds of electrolytes. Like all electrolytes they break up into charged particles. • What sets them apart from each other, and other electrolytes is the way that they break up.

Arrhenius Acids Svente Arrhenius (who first proposed theory of ionization) identified acids as substances that ionize in water to produce hydronium ion. For example: HCl + H 2 O H 3 O+ + Cl. Any substance that ionizes in water to produce hydronium ion can be classified as an acid.

H 3 O+ (aka H+) • The hydronium ion is also known as a hydrogen ion. • This allows us to shorten the ionization reactions for acids. The following equations represent the same chemical change: HCl + H 2 O H 3 O+ + Cland HCl H+ + Cl- However, it should be understood that H+ is an abbreviation for the hydronium ion. H+ ions DO NOT exist in water solution but are snatched up by water molecules to form hydronium ions.

Arrhenius Base Svente Arrhenius also identified bases as substances that ionize in water to produce hydroxide ion. For example: Na. OH Na+ + OHAny substance that ionizes in water to produce hydroxide ion can be classified as a base.

Properties of Acids All acids have the following properties: Neutralize bases to form a salt and water Have a sour taste (example: citric acid, vinegar) React with metals to produce a salt plus hydrogen gas. Have p. H’s less than 7 / Affect indicators Why? Because all acids have H 3 O+ ions present!

Properties of Bases All bases have the following properties: Neutralize acids to form a salt and water Have a bitter taste (example: unsweetened chocolate, heroin) Are slippery. React with fats/oils to form soap (saponification) Have p. H’s greater than 7 / Affect indicators Why? Because all bases have OH- ions present!

Salts Ionic substances that break up in solution to produce ions other than hydronium and hydroxide ions. Na. Cl (s) Na+(aq) + Cl- (aq) KNO 3(s) K+ (aq) + NO 3 - (aq) Li 2 SO 4 (s) 2 Li+ (aq) + SO 42 -(aq) Salts are made up of positive (metal) and negative (nonmetallic or polyatomic) ions. The more familiar you become with Table E, the easier it will be for you to identify salts.

Strong/Weak Acids • Acids can be either strong electrolytes or weak electrolytes. • Strong acids (such as HCl) completely break up into their ions: HCl (aq) H+(aq) + Cl-(aq) • Weak acids (such as HC 2 H 3 O 2) only partially break up into their ions: HC 2 H 3 O 2 H+ (aq) + C 2 H 3 O 2 -(aq) Weak acids don’t completely break up because they go to equilibrium!

Strong/Weak Bases • Bases can be either strong electrolytes or weak electrolytes. • Strong bases (such as Na. OH) completely break up into their ions: Na. OH (aq) Na+(aq) + OH-(aq) • Weak bases (such as NH 3) only partially break up into their ions: NH 3 (aq) + H 2 O NH 4+ (aq) + OH-(aq) Weak bases don’t completely break up because they go to equilibrium!

Bronsted-Lowry Acids & Bases Another theory of acids & bases

Not everyone was happy with Arrhenius’s definition A major problem with the Arrhenius definition of acids and bases is that it limits acids and bases to water (aqueous) solutions. Since an acid requires water to ionize and form hydronium ions, there can be no Arrhenius acids unless water is involved as the solvent.
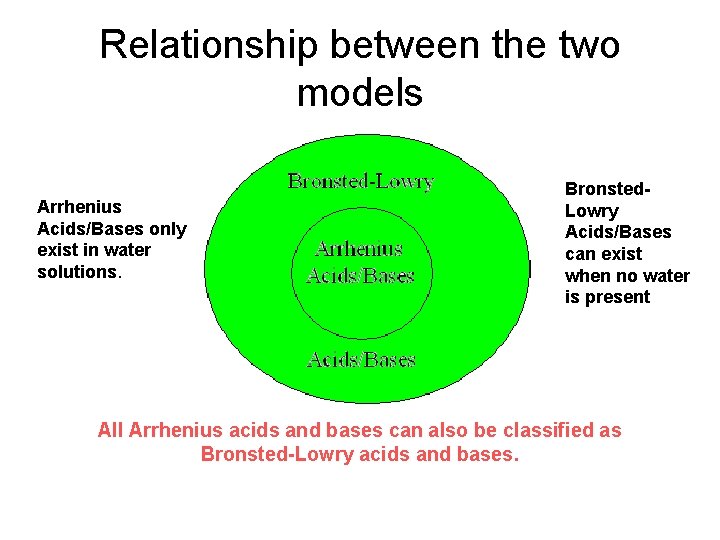
Relationship between the two models Arrhenius Acids/Bases only exist in water solutions. Bronsted. Lowry Acids/Bases can exist when no water is present All Arrhenius acids and bases can also be classified as Bronsted-Lowry acids and bases.

Bronsted-Lowry Definitions Bronsted and Lowry felt that this was too limiting, since there are many non-aqueous systems (no water is present). They came up with the following definitions for acids and bases. An acid is a proton (H+ ion) donor A base is a proton acceptor Saying goes: “FOLLOW THE BOUNCING H+ “

An example In the reaction below there are no Arrhenius acids or bases present (because no hydronium ions or hydroxide ions are formed). However, the HCl is acting as a Bronsted. Lowry acid because it is giving a H+ ion to the NH 3 (which is acting as a H+ ion acceptor - a base)

Acid & Bases are Roles In the Bronsted-Lowry definition, substances are classified as acids or bases depending on how they behave in a given situation. This means that the same substance can act as a acid in one reaction (by donating a proton) while acting as a base in another reaction
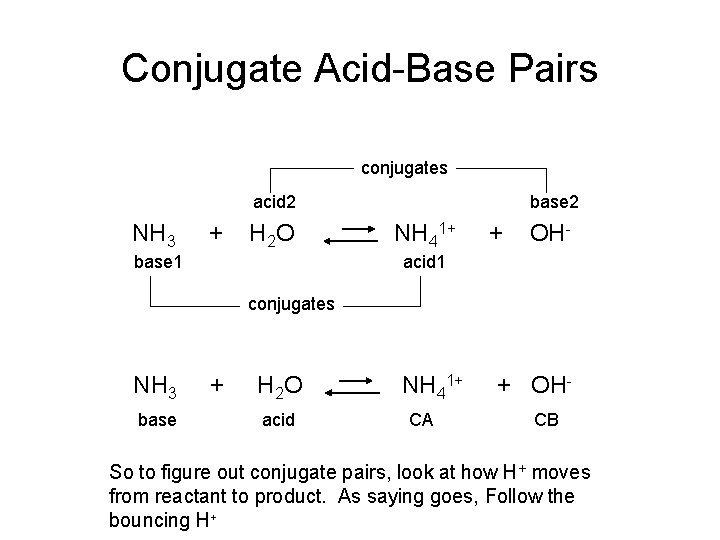
Conjugate Acid-Base Pairs conjugates acid 2 NH 3 + H 2 O base 1 base 2 NH 41+ + OH- acid 1 conjugates NH 3 base + H 2 O acid NH 41+ CA + OHCB So to figure out conjugate pairs, look at how H+ moves from reactant to product. As saying goes, Follow the bouncing H+
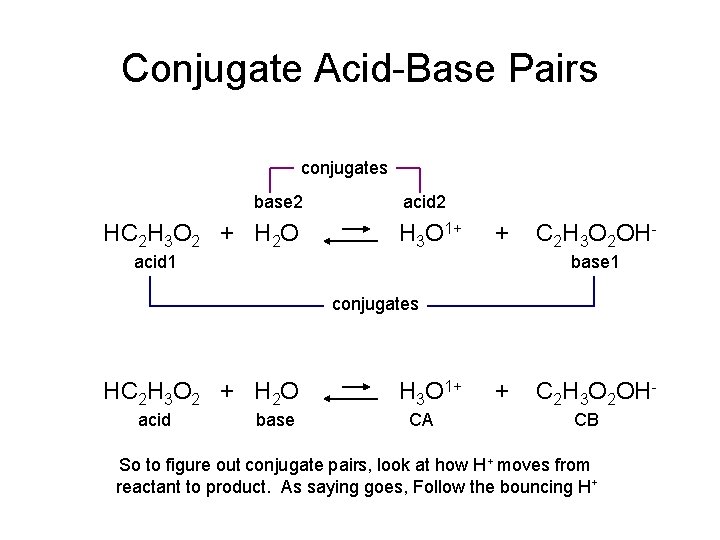
Conjugate Acid-Base Pairs conjugates base 2 HC 2 H 3 O 2 + H 2 O acid 2 H 3 O 1+ + acid 1 C 2 H 3 O 2 OHbase 1 conjugates HC 2 H 3 O 2 + H 2 O acid base H 3 O 1+ CA + C 2 H 3 O 2 OHCB So to figure out conjugate pairs, look at how H+ moves from reactant to product. As saying goes, Follow the bouncing H+
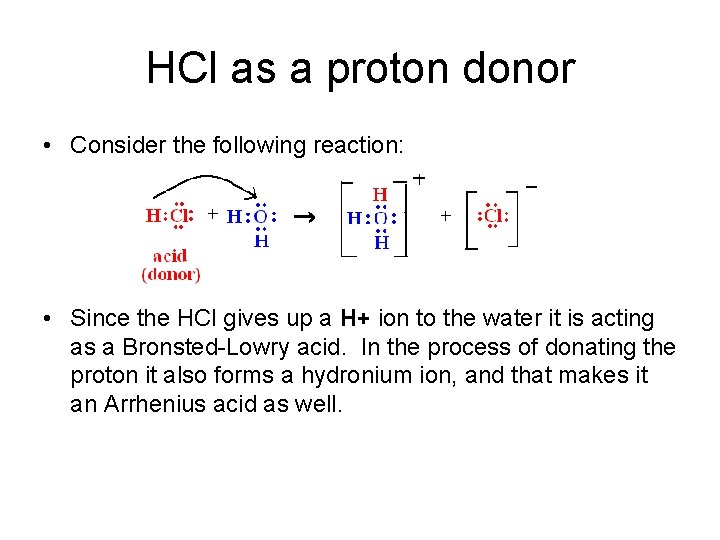
HCl as a proton donor • Consider the following reaction: • Since the HCl gives up a H+ ion to the water it is acting as a Bronsted-Lowry acid. In the process of donating the proton it also forms a hydronium ion, and that makes it an Arrhenius acid as well.
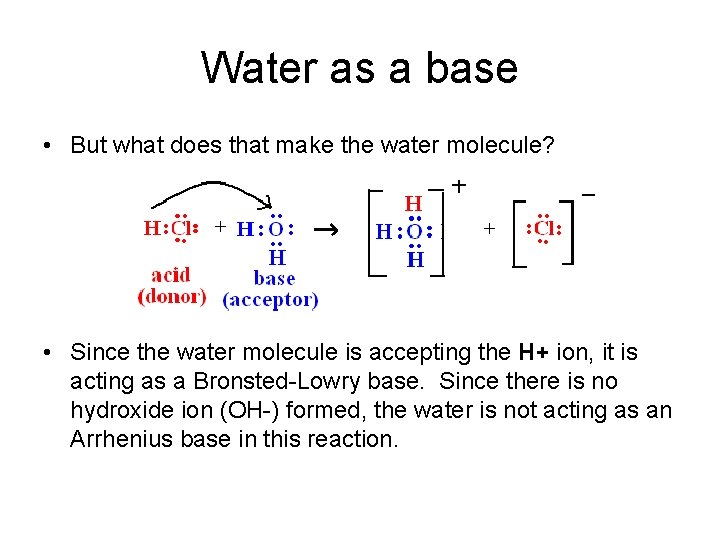
Water as a base • But what does that make the water molecule? • Since the water molecule is accepting the H+ ion, it is acting as a Bronsted-Lowry base. Since there is no hydroxide ion (OH-) formed, the water is not acting as an Arrhenius base in this reaction.

Ammonia as a base • Let’s look at another example: • Here the ammonia molecule is accepting a H+ ion and therefore is acting as an Bronsted-Lowry base. However, in the process of reacting with the water it is also forming a hydroxide ion. That makes the ammonia an Arrhenius base as well.

But what about the water? Since the water is giving up a H+ ion, it is acting as a Bronsted-Lowry acid. Since it does not form hydronium ions, it is NOT acting an Arrhenius acid.

So is water an acid or a base? • In one example, we said that water was acting as a base, and in another example we said that it was acting as an acid. • Some of you may be confused by this because you are thinking of acids and bases as being like boys and girls. Boys are boys and girls are girls, and they can’t switch back and forth. However, acids and bases are NOT like this.

Amphiprotic • Sometimes a molecule can donate a proton (act as an acid) and sometimes it can accept a proton (act as a base). • Molecules that have this ability to act as both an acid and a base are called amphoteric or amphiprotic. • Water is the most common example of an amphoteric substance.

Acid-base equilibrium Many acid base reactions go to equilibrium, that is they have both a forward and reverse reactions For instance, acetic acid (HC 2 H 3 O 2) reacts with water to form hydronium ion and acetate ion.

The reverse reaction However, the acetic acid only partially ionizes because a reverse reaction takes place preventing the forward reaction from reaching completion. In the reverse reaction, the Hydronium ion acts as an acid (a proton donor) while the acetate ion acts as the base.

Conjugate Acid-Bases • When a substance donates a proton, the substance that is left is its conjugative base: • Notice that the conjugative base is accepting a proton in the reverse reaction. Every acid has a conjugative base

Conjugative Acid-Base Pairs 2 • When a substance accepts a proton, the substance that is formed is its conjugative acid: • Notice that the conjugative acid is donating a proton in the reverse reaction. Every base has a conjugative acid

Identify the amphoteric substances in this chart.


Strong and weak acids In predicting the type of salt it is helpful to remember the three strong acids: • • • HCl – hydrochloric acid HNO 3 – nitric acid H 2 SO 4 – sulfuric acid Most other acids are weak. Acetic acid (HC 2 H 3 O 2) is the most common weak acid discussed in Regents chemistry.

Strong and Weak Bases For bases, the Group I hydroxides are all strong bases. Li. OH – lithium hydroxide Na. OH – sodium hydroxide KOH – potassium hydroxide • Most other hydroxides can be considered as weak bases. Ammonia or Ammonium hydroxide (NH 3 or NH 4 OH) is the most common weak base discussed in Regents chemistry.

Acid Strength and Ionization Constants • For the ionization or dissociation equilibrium of an acid in aqueous solution, such as: HA(aq) + H 2 O H 3 O+(aq) + A-(aq); The ionization or dissociation constant is expressed as follows: The values of Ka indicate the relative strength of the acids. Strong acids have very large Ka, while weak acids have small Ka’s (Ka << 1)


Relative Strength of Acids and Their Conjugate Bases Acids Conjugate Bases Very Strong Very Weak Strong Very Weak Very Strong _______________ • Strong acids lose protons very readily weak conjugate bases; • Weak acids do not lose protons very readily strong conjugate bases.
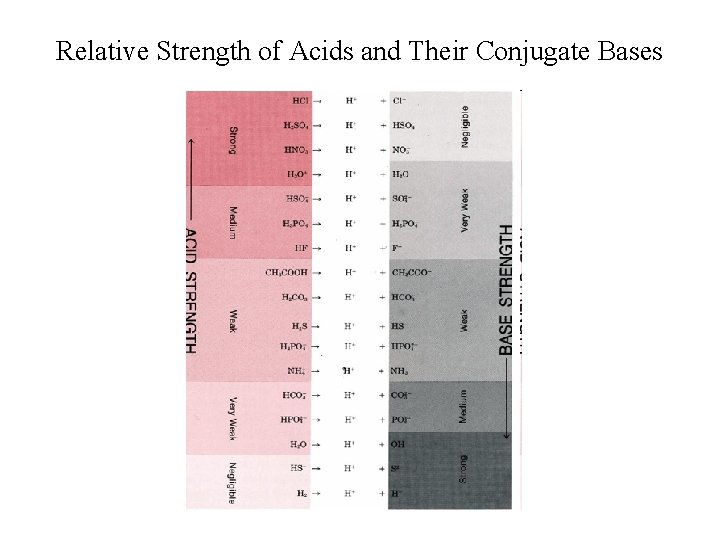
Relative Strength of Acids and Their Conjugate Bases

Acid-Base Properties of Water • Auto-ionization of water: 2 H 2 O H 3 O+(aq) + OH-(aq) Kw = [H 3 O+][OH-] = 1. 0 x 10 -14 at 25 o. C • Water ionizes to produce both H 3 O+ and OH-, thus it has both acid and base properties. Kw is called water ionization constant. • Pure water at 25 o. C: [H 3 O+] = [OH-] = 1. 0 x 10 -7 M
![Expressing Acidity in p. H Scale • p. H = -log[H+] (note: [H+] = Expressing Acidity in p. H Scale • p. H = -log[H+] (note: [H+] =](http://slidetodoc.com/presentation_image_h/86d9708d5f0ad9732991202c45810f8a/image-38.jpg)
Expressing Acidity in p. H Scale • p. H = -log[H+] (note: [H+] = [H 3 O+]) • p. OH = -log[OH-] • p. Kw = -log(Kw); p. Ka = -log(Ka); p. Kb = -log(Kb) • For water, Kw = [H 3 O+][OH-] = 1. 0 x 10 -14 • -log(Kw) = -log [H 3 O+] + (-log[OH-]) • p. Kw = p. H + p. OH = 14. 00 • At 25 o. C, p. OH = 14 – p. H
![Acidity and p. H Range • Acidic solutions: [H 3 O+] > 1. 0 Acidity and p. H Range • Acidic solutions: [H 3 O+] > 1. 0](http://slidetodoc.com/presentation_image_h/86d9708d5f0ad9732991202c45810f8a/image-39.jpg)
Acidity and p. H Range • Acidic solutions: [H 3 O+] > 1. 0 x 10 -7 M; p. H < 7; • Basic solutions: [OH-] > 1. 0 x 10 -7 M or [H 3 O+] < 1. 0 x 10 -7 M p. H > 7; • Neutral solutions: [H 3 O+] = [OH-] = 1. 0 x 10 -7 M; p. H = 7. 00
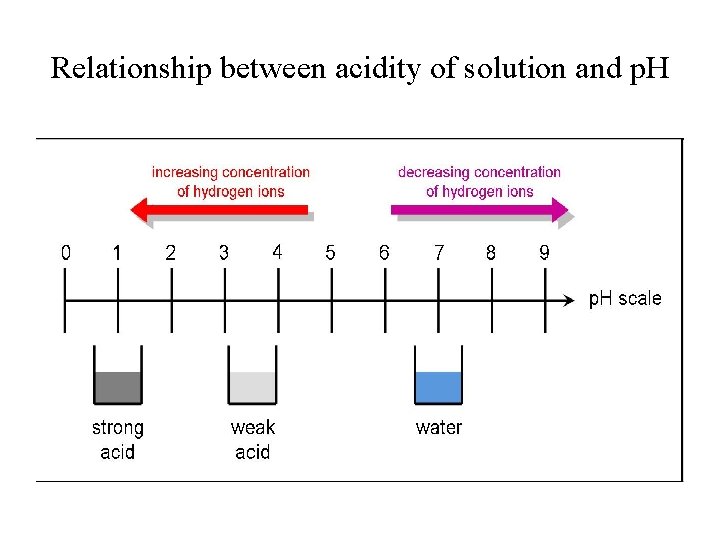
Relationship between acidity of solution and p. H
![[H 3 O+] and p. H of Strong Acids • Strong acids like HCl [H 3 O+] and p. H of Strong Acids • Strong acids like HCl](http://slidetodoc.com/presentation_image_h/86d9708d5f0ad9732991202c45810f8a/image-41.jpg)
[H 3 O+] and p. H of Strong Acids • Strong acids like HCl and HCl. O 4 ionize completely in aqueous solution: HCl(aq) + H 2 O H 3 O+(aq) + Cl-(aq); HCl. O 4(aq) + H 2 O H 3 O+(aq) + Cl. O 4 -(aq); • In solutions of strong monoprotic acids HA, such as HCl and HCl. O 4, [H 3 O+] = [HA]0 • For example, in 0. 10 M HCl, [H 3 O+] = 0. 10 M, and p. H = -log(0. 10) = 1. 00
![[OH-] and p. H of Strong Bases • Like strong acids, strong bases also [OH-] and p. H of Strong Bases • Like strong acids, strong bases also](http://slidetodoc.com/presentation_image_h/86d9708d5f0ad9732991202c45810f8a/image-42.jpg)
[OH-] and p. H of Strong Bases • Like strong acids, strong bases also ionize completely in aqueous solution. • Examples: Na. OH(aq) Na+(aq) + OH-(aq); Ba(OH)2(aq) Ba 2+(aq) + 2 OH-(aq); • In a base solution such as 0. 050 M Na. OH, [OH-] = [Na. OH]0 = 0. 050 M; p. OH = -log(0. 050 M) = 1. 30; p. H = 14. 00 - 1. 30 = 12. 70 • In a base solution such as 0. 050 M Ba(OH)2, [OH-] = 2 x [Ba(OH)2]0 = 0. 10 M; p. OH = -log(0. 10) = 1. 00; p. H = 14. 00 - 1. 00 = 13. 00
![[H 3 O+] and p. H of Weak Acids • In weak acid solutions, [H 3 O+] and p. H of Weak Acids • In weak acid solutions,](http://slidetodoc.com/presentation_image_h/86d9708d5f0ad9732991202c45810f8a/image-43.jpg)
[H 3 O+] and p. H of Weak Acids • In weak acid solutions, [H 3 O+] < [HA]0; • [H 3 O+] and p. H can be calculated from the initial concentration of the acid and its Ka value. • For example, in 0. 100 M acetic acid, CH 3 COOH, with Ka = 1. 8 x 10 -5, [H 3 O+] and p. H can be calculated using the “ICE” table.
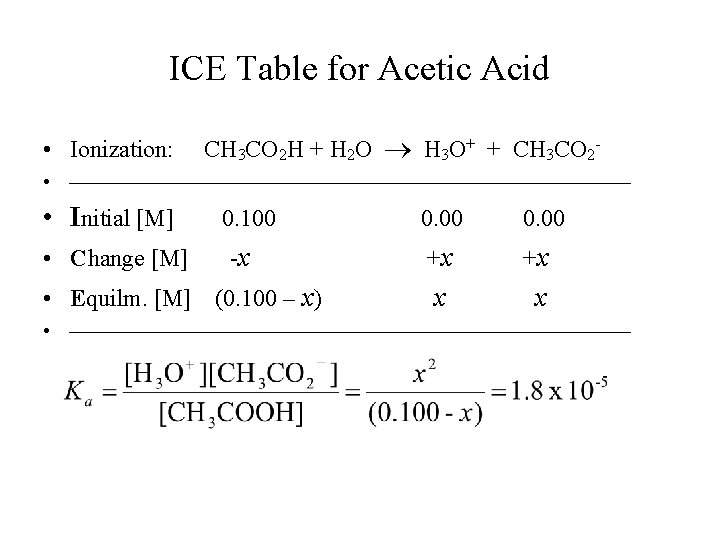
ICE Table for Acetic Acid • Ionization: CH 3 CO 2 H + H 2 O H 3 O+ + CH 3 CO 2 - • • Initial [M] • Change [M] 0. 100 -x • Equilm. [M] (0. 100 – x) 0. 00 +x +x x x •
![Calculating [H 3 O+] from initial concentration and Ka by approximation method. • Solving Calculating [H 3 O+] from initial concentration and Ka by approximation method. • Solving](http://slidetodoc.com/presentation_image_h/86d9708d5f0ad9732991202c45810f8a/image-45.jpg)
Calculating [H 3 O+] from initial concentration and Ka by approximation method. • Solving for x in the expression for Ka:
![[OH-] and p. H of a Weak Base • In a weak base, [OH-] [OH-] and p. H of a Weak Base • In a weak base, [OH-]](http://slidetodoc.com/presentation_image_h/86d9708d5f0ad9732991202c45810f8a/image-46.jpg)
[OH-] and p. H of a Weak Base • In a weak base, [OH-] < [Base]0; • [OH-] and p. H can be calculated from the initial concentration of the base and its Kb value. • For example, in 0. 100 M ammonia, NH 3, with Kb = 1. 8 x 10 -5, [OH-] and p. H can be calculated using the following “ICE” table.
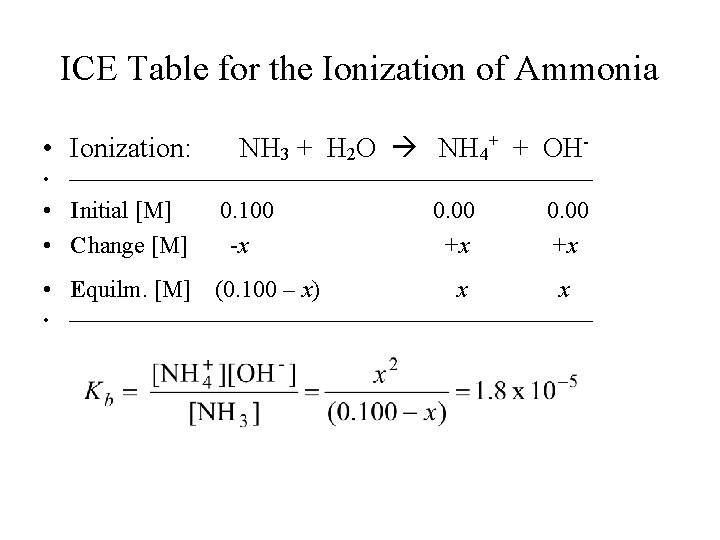
ICE Table for the Ionization of Ammonia • Ionization: • NH 3 + H 2 O NH 4+ + OH- • Initial [M] • Change [M] 0. 100 -x • Equilm. [M] (0. 100 – x) • 0. 00 +x x
![Calculate [OH-] from initial concentration and Kb by approximation method. • Solving for x Calculate [OH-] from initial concentration and Kb by approximation method. • Solving for x](http://slidetodoc.com/presentation_image_h/86d9708d5f0ad9732991202c45810f8a/image-48.jpg)
Calculate [OH-] from initial concentration and Kb by approximation method. • Solving for x in the expression for Kb:
- Slides: 48