Modern Atomic Theory The Electronic Structure of the














- Slides: 23

Modern Atomic Theory The Electronic Structure of the Atom

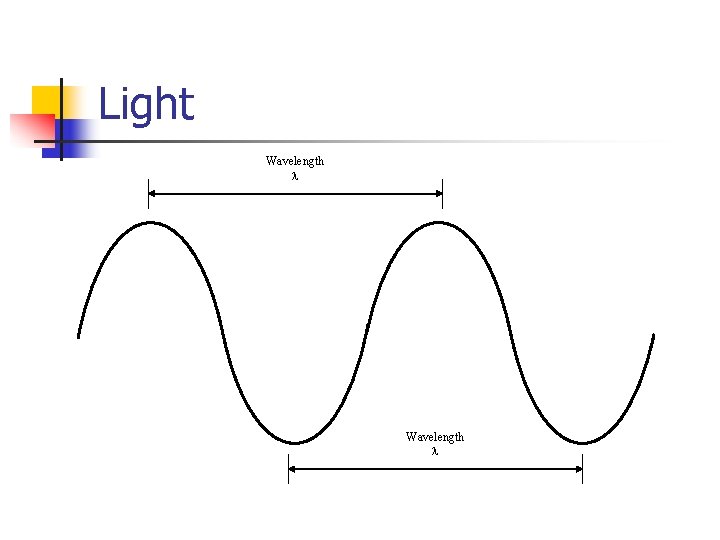
Light Wavelength

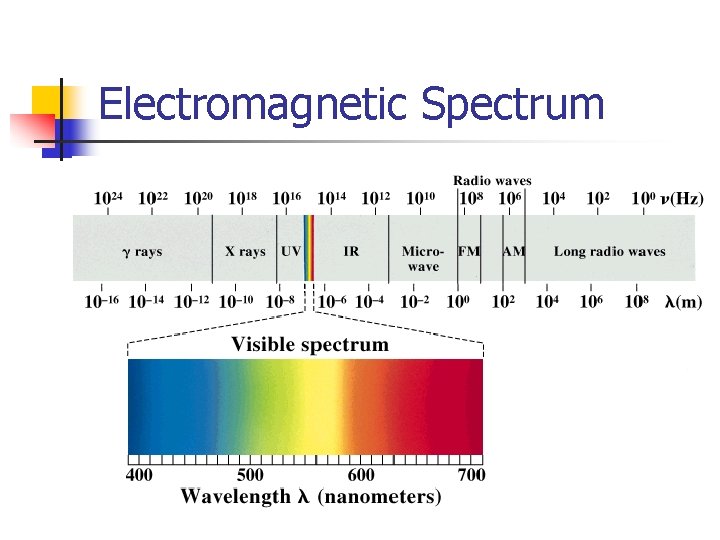
Light Waves n The speed of light is constant, and equals 3. 0 × 108 m/s c = λf n The energy of light depends on the frequency E = hf h = 6. 63 × 10 -34 J·s

Electromagnetic Spectrum

A Continuous Spectrum

Line Spectra

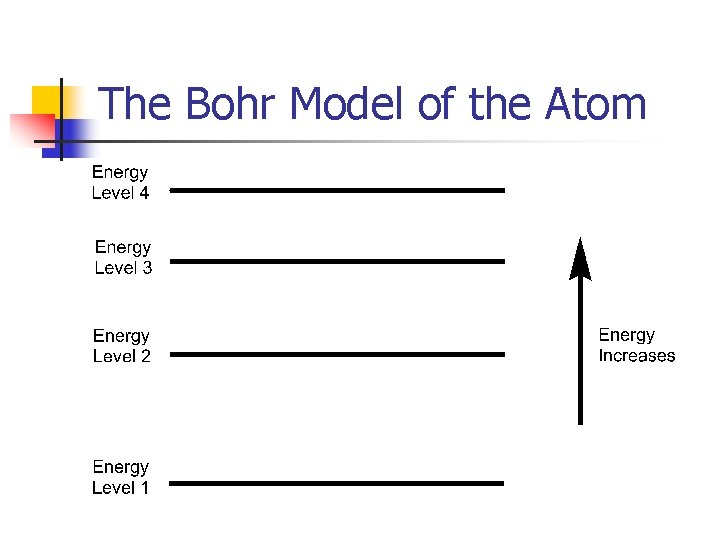
The Bohr Model of the Atom n electrons can only have certain energies; we say that the energies of an atom’s electrons are quantized or restricted to particular values

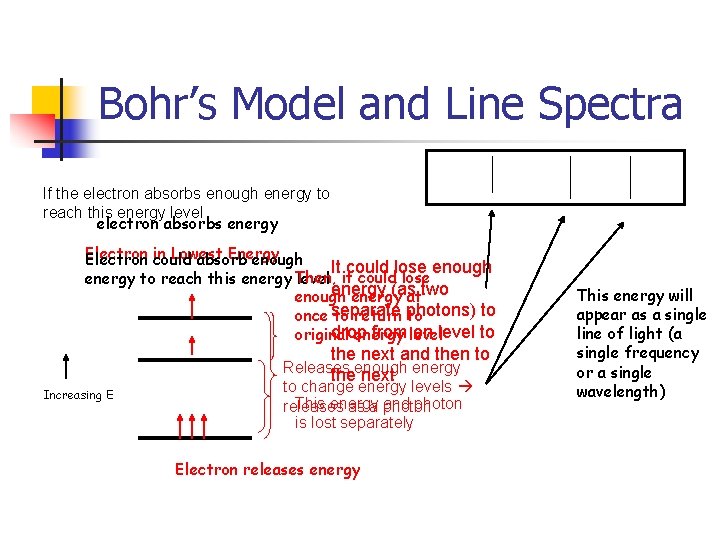
Light and the Atom n n n An atom can absorb energy to promote an electron to a higher energy state When the electron drops to a lower energy state it releases energy as light The energy of the light released equals the energy difference between the two electron energy levels

The Bohr Model of the Atom

Bohr’s Model and Line Spectra If the electron absorbs enough energy to reach this energy level electron absorbs energy Electron Lowest Energy Electron in could absorb enough It could lose enough energy to reach this energy Then, level it could lose energy (as enough energy attwo to once separate to return photons) to drop from level on level to original energy the next and then to Releases theenough next energy Increasing E to change energy levels This energy and photon releases as a photon is lost separately Electron releases energy This energy will appear as a single line of light (a single frequency or a single wavelength)

The Atom n n The electron energy levels are NOT circular orbits around the nucleus, but probability clouds called orbitals. An orbital is the region in space where we are most likely to find the electron

The Atom n n The atom contains shells; n = 1, n = 2, n = 3, etc. The higher n, the larger the shell. Each shell is divided into subshells; there are four kinds of subshell n s, p, d, f

The Atom n n n Each subshell consists of orbitals Orbitals in s subshells are called sorbitals, orbitals in p-subshells are called p-orbitals, etc. An orbital can accommodate one or two electrons, but no more.

s-orbitals 1 s 2 s

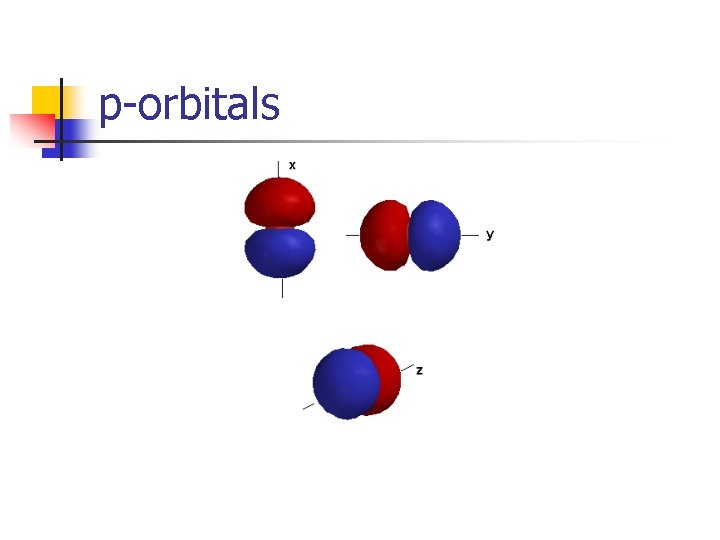
p-orbitals

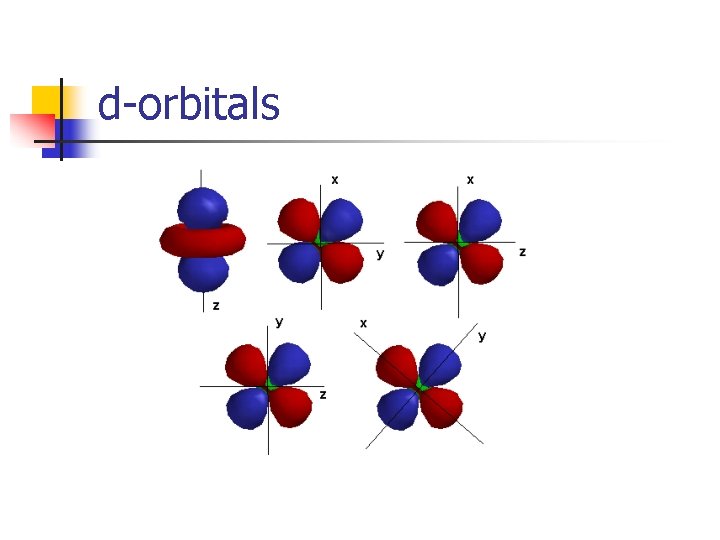
d-orbitals

An electron configuration shows the distribution of electrons among the subshells of an atom. 6 s 5 s 4 s 3 s 2 s 1 s 6 p 5 p 4 p 3 p 2 p 6 d 6 f 5 d 5 f 4 d 4 f 3 d

Examples – e 1 - configurations H 1 s 1 Li 1 s 22 s 1 O 1 s 22 p 4 Ni 1 s 22 p 63 s 23 p 64 s 23 d 8

The electrons in the outer most shell are called the valence electrons. Electrons in the inner shells are core electrons. F 1 s 22 p 5 valence electrons P 1 s 22 p 63 s 23 p 3 valence electrons

Give the electron configuration for the following: n Li 1 s 22 s 1 n Na 1 s 22 p 63 s 1 n K 1 s 22 p 63 s 23 p 64 s 1 Atoms in a group have the same sort of valence electron configuration.

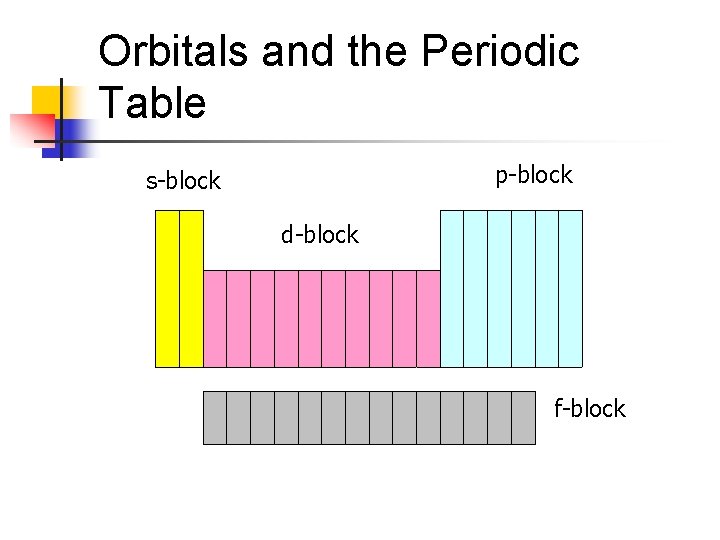
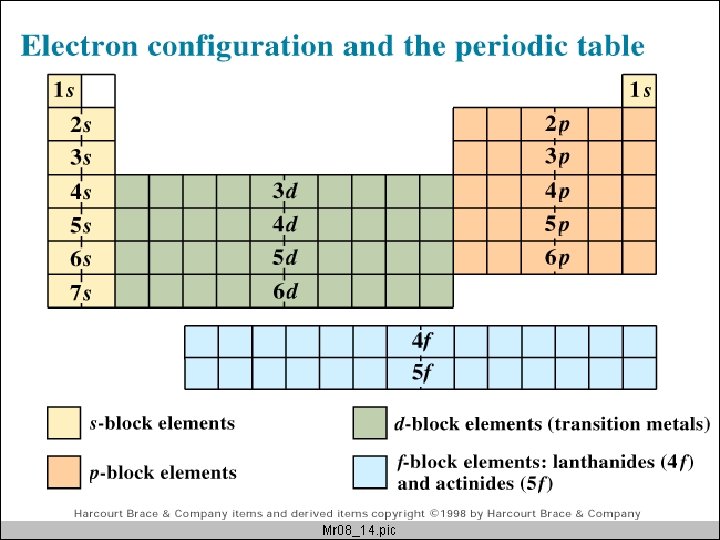
Orbitals and the Periodic Table p-block s-block d-block f-block

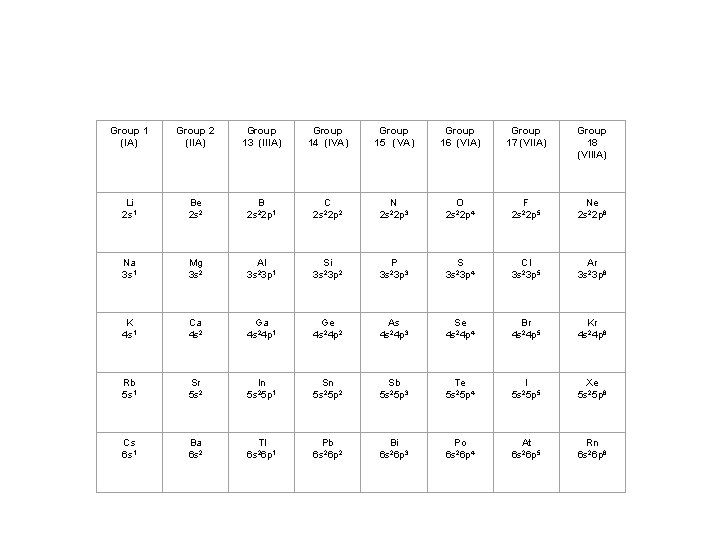
Group 1 (IA) Group 2 (IIA) Group 13 (IIIA) Group 14 (IVA) Group 15 (VA) Group 16 (VIA) Group 17 (VIIA) Group 18 (VIIIA) Li 2 s 1 Be 2 s 2 B 2 s 22 p 1 C 2 s 22 p 2 N 2 s 22 p 3 O 2 s 22 p 4 F 2 s 22 p 5 Ne 2 s 22 p 6 Na 3 s 1 Mg 3 s 2 Al 3 s 23 p 1 Si 3 s 23 p 2 P 3 s 23 p 3 S 3 s 23 p 4 Cl 3 s 23 p 5 Ar 3 s 23 p 6 K 4 s 1 Ca 4 s 2 Ga 4 s 24 p 1 Ge 4 s 24 p 2 As 4 s 24 p 3 Se 4 s 24 p 4 Br 4 s 24 p 5 Kr 4 s 24 p 6 Rb 5 s 1 Sr 5 s 2 In 5 s 25 p 1 Sn 5 s 25 p 2 Sb 5 s 25 p 3 Te 5 s 25 p 4 I 5 s 25 p 5 Xe 5 s 25 p 6 Cs 6 s 1 Ba 6 s 2 Tl 6 s 26 p 1 Pb 6 s 26 p 2 Bi 6 s 26 p 3 Po 6 s 26 p 4 At 6 s 26 p 5 Rn 6 s 26 p 6
