MODERN ATOMIC THEORY Niels Bohr In 1913 Niels

MODERN ATOMIC THEORY

Niels Bohr • In 1913, Niels Bohr, a Danish physicist, suggested that the energy of each electron was related to the electron’s path around the nucleus.

Niels Bohr • He stated that they must gain energy to move to a higher energy level or lose energy to move to a lower energy level.

• In 1925, a new model of the atom was proposed and stated that electrons behave more like waves on a vibrating string than like particles. • Determining the exact location of an electron, speed, and direction of an electron is impossible.

Calculations • Scientist based their calculations on “chance” when studying electron orbital's.
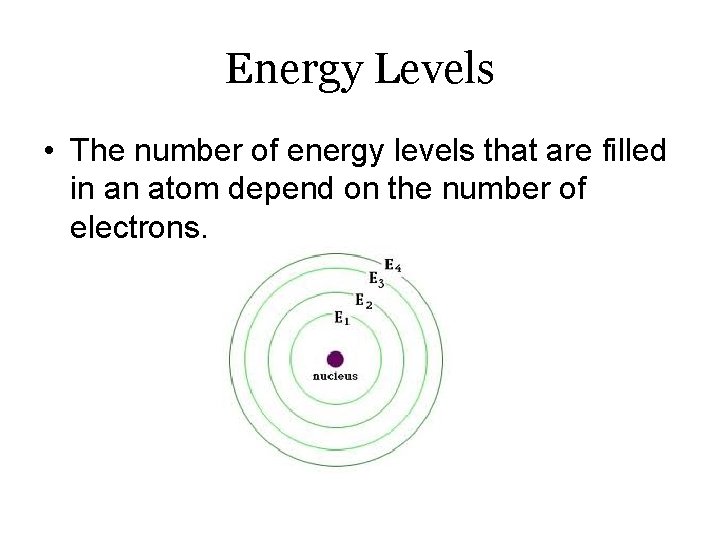
Energy Levels • The number of energy levels that are filled in an atom depend on the number of electrons.

Energy Levels • The electrons in the outer energy level of an atom are called valence electrons. • These determine the chemical properties of an atom.
Orbitals • There are four kinds of orbitals:

S Orbital • Shaped like a sphere and only has one orientation: • Has the lowest energy and can hold two electrons.
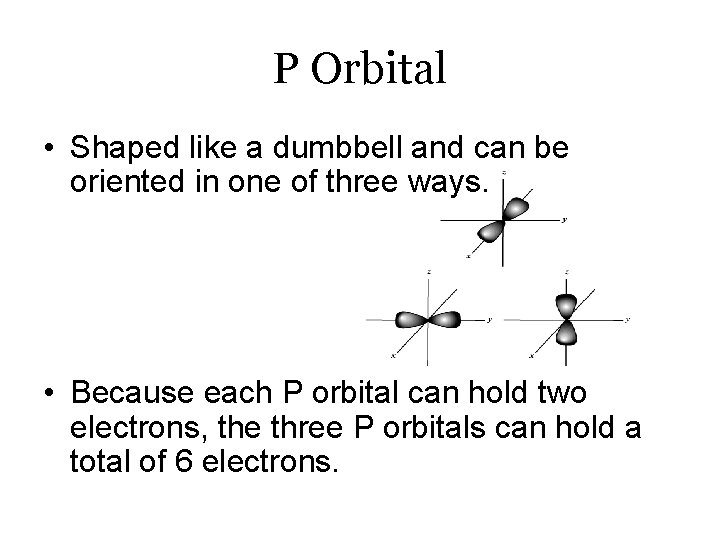
P Orbital • Shaped like a dumbbell and can be oriented in one of three ways. • Because each P orbital can hold two electrons, the three P orbitals can hold a total of 6 electrons.
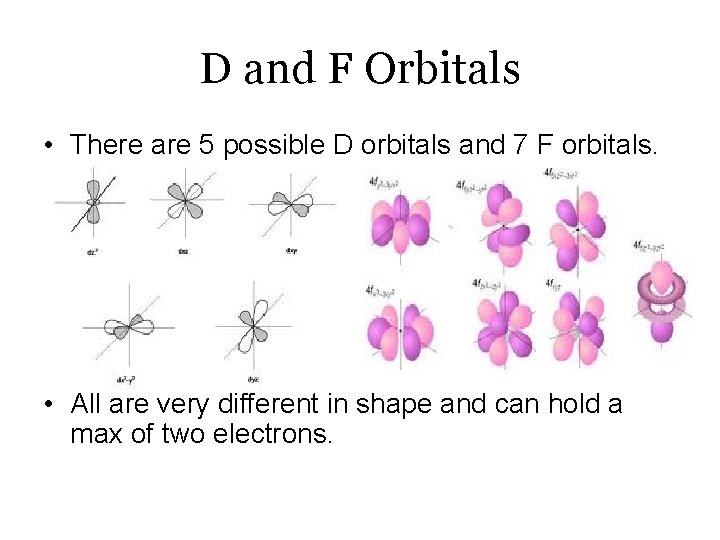
D and F Orbitals • There are 5 possible D orbitals and 7 F orbitals. • All are very different in shape and can hold a max of two electrons.

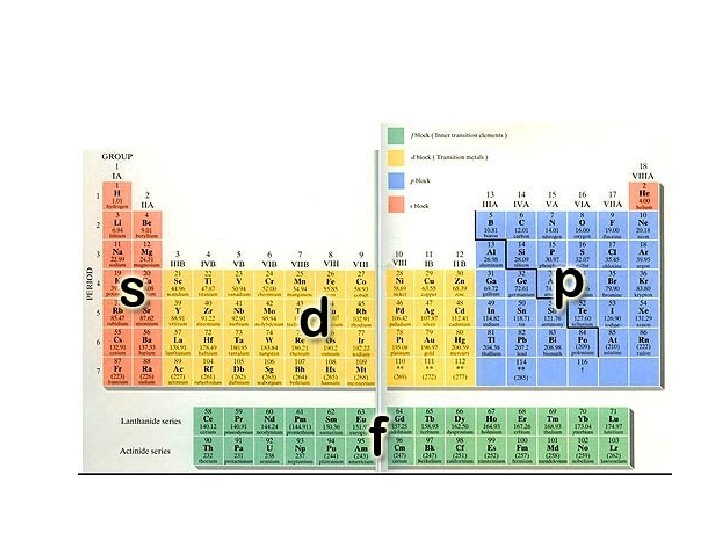
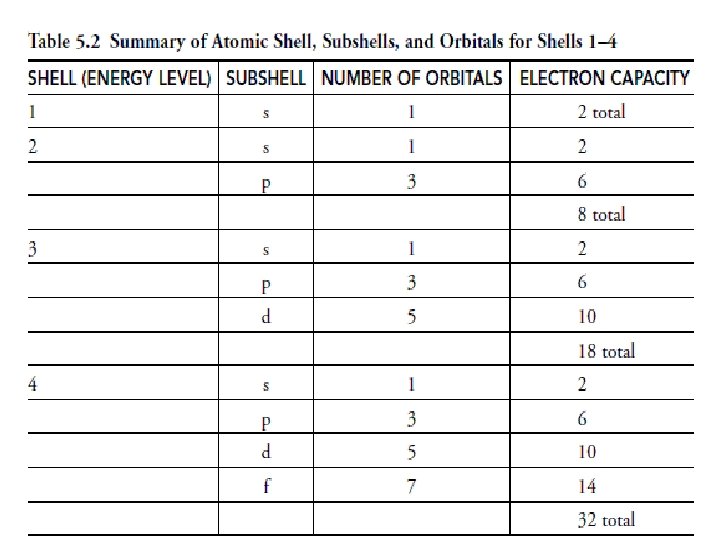
Orbital Levels • Orbitals determine the number of electrons that each level can hold.

Electron Transitions • Electrons jump between energy levels when an atom gains or loses electrons. • The lowest state of energy is called ground state. • This is usually the state that electrons are in at normal temperatures.
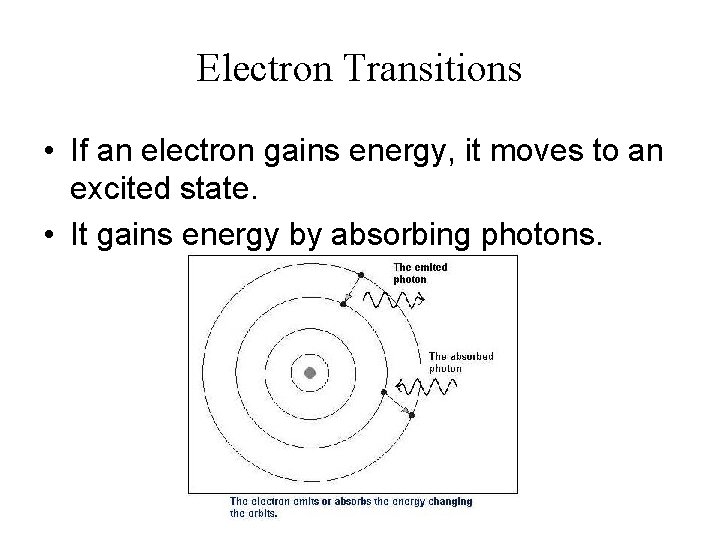
Electron Transitions • If an electron gains energy, it moves to an excited state. • It gains energy by absorbing photons.
- Slides: 16