Modern Atomic Theory Chapter 11 The ELECTRON Wave













































![Element Lithium Configuration notation Orbital notation 1 s 22 s 1 [He]2 s 1 Element Lithium Configuration notation Orbital notation 1 s 22 s 1 [He]2 s 1](https://slidetodoc.com/presentation_image_h2/f11bc924aff8e70e2af90cd609ca117f/image-75.jpg)

- Slides: 78

Modern Atomic Theory Chapter 11

The ELECTRON: Wave – Particle Duality “No familiar conceptions can be woven around the electron. Something unknown is doing we don’t know what. ” -Sir Arthur Eddington The Nature of the Physical World (1934)

The Dilemma of the Atom �Electrons outside the nucleus are attracted to the protons in the nucleus �Charged particles moving in curved paths lose energy �What keeps the atom from collapsing?

Wave-Particle Duality JJ Thomson won the Nobel prize for describing the electron as a particle. His son, George Thomson won the Nobel prize for describing the wave-like nature of the electron. The electron is a particle! The electron is an energy wave!


Electromagnetic Radiation � Most subatomic particles behave as PARTICLES (photons) and obey the physics of waves.

The Wave-like Electron The electron propagates through space as an energy wave. To understand the atom, one must understand the behavior of electromagnetic waves. Louis de. Broglie

Electromagnetic radiation propagates through space as a wave moving at the speed of light. c = speed of light, a constant (3. 00 x 108 m/s) = frequency, in units of hertz (hz, sec-1) = wavelength, in meters

The energy (E ) of electromagnetic radiation is directly proportional to the frequency ( ) of the radiation. E = h E = Energy, in units of Joules (kg·m 2/s 2) h = Planck’s constant (6. 626 x 10 -34 J·s) = frequency, in units of hertz (hz, sec-1) Often times in calculations, you will use the following formula (remember c=vλ): E = hc λ

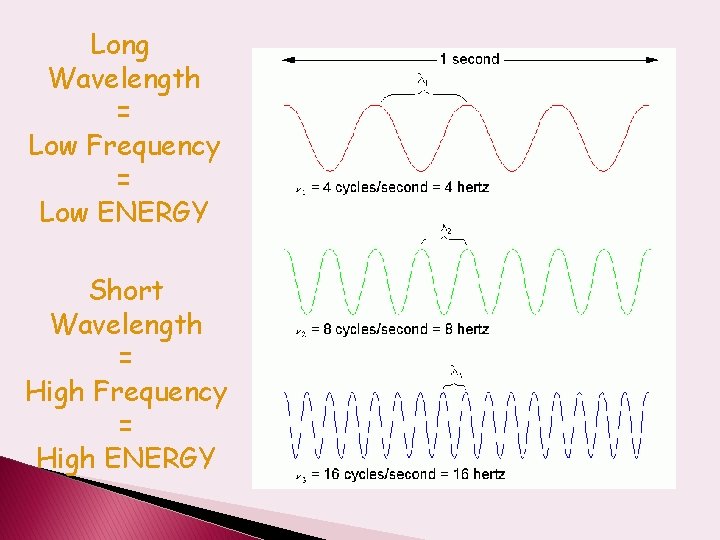
Long Wavelength = Low Frequency = Low ENERGY Short Wavelength = High Frequency = High ENERGY

Answering the Dilemma of the Atom �Treat electrons as waves �As the electron moves toward the nucleus, the wavelength shortens �Shorter wavelength = higher energy �Higher energy = greater distance from the nucleus

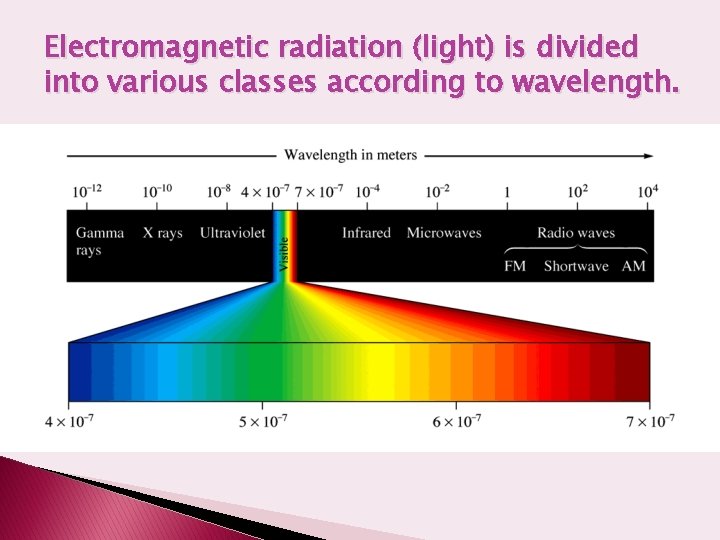
Electromagnetic radiation (light) is divided into various classes according to wavelength.

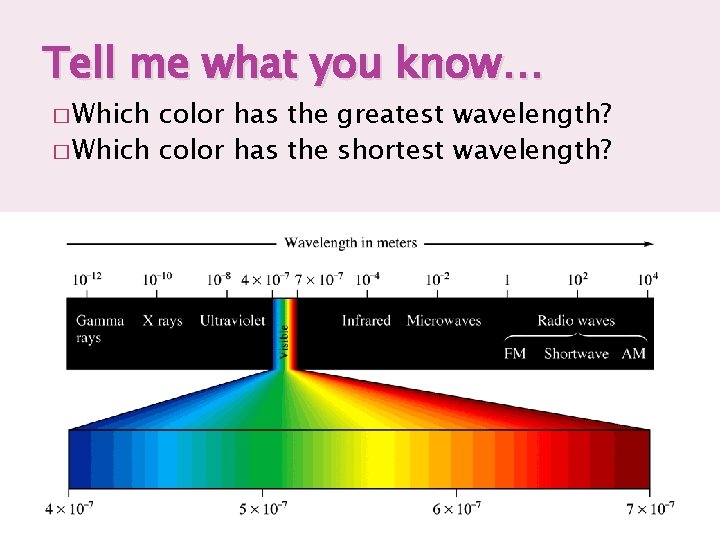
Tell me what you know… � Which color has the greatest wavelength? � Which color has the shortest wavelength?

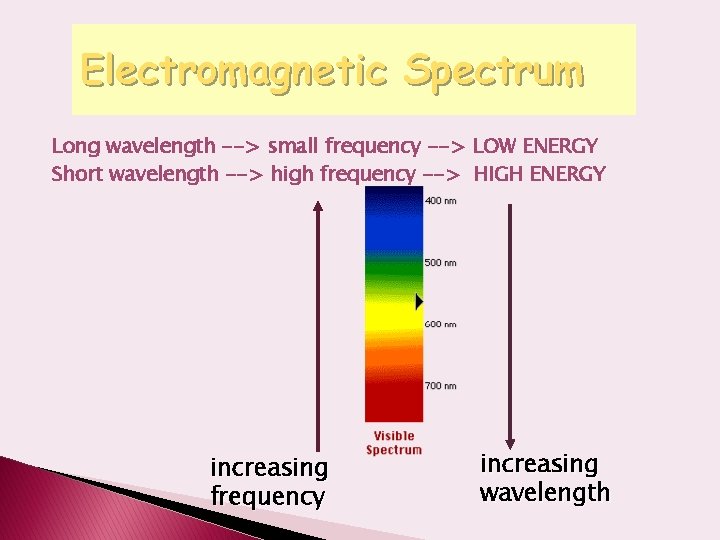
Electromagnetic Spectrum Long wavelength --> small frequency --> LOW ENERGY Short wavelength --> high frequency --> HIGH ENERGY increasing frequency increasing wavelength

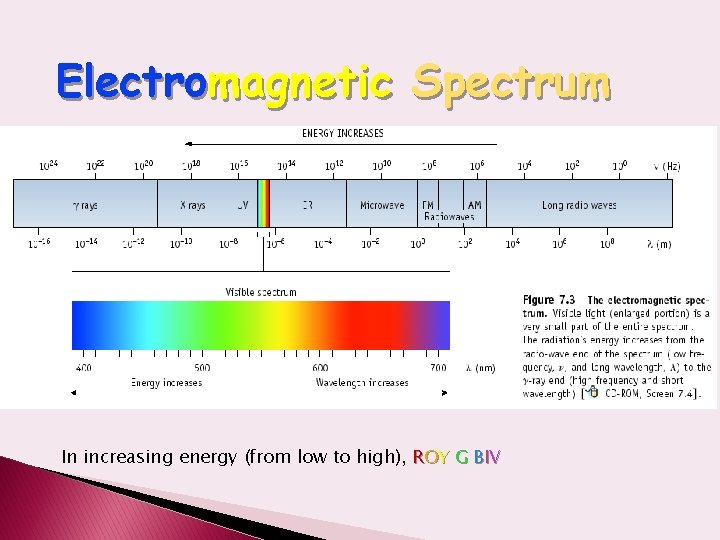
Electromagnetic Spectrum In increasing energy (from low to high), ROY G BIV

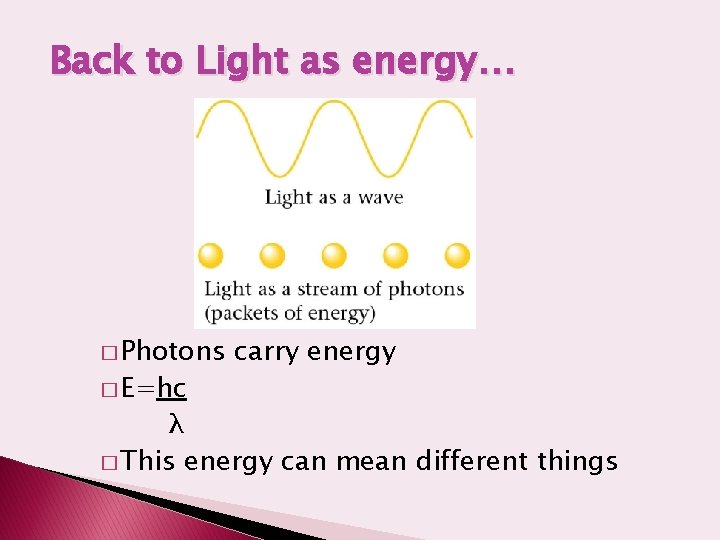
Back to Light as energy… � Photons � E=hc carry energy λ � This energy can mean different things

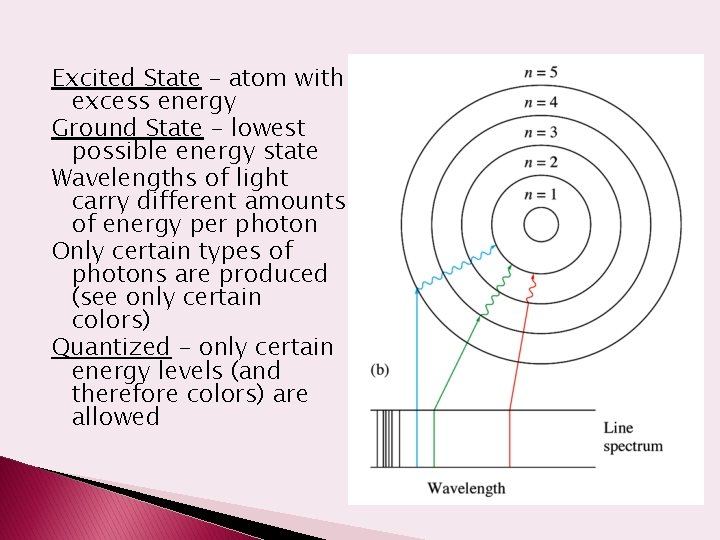
Excited State – atom with excess energy Ground State – lowest possible energy state Wavelengths of light carry different amounts of energy per photon Only certain types of photons are produced (see only certain colors) Quantized – only certain energy levels (and therefore colors) are allowed

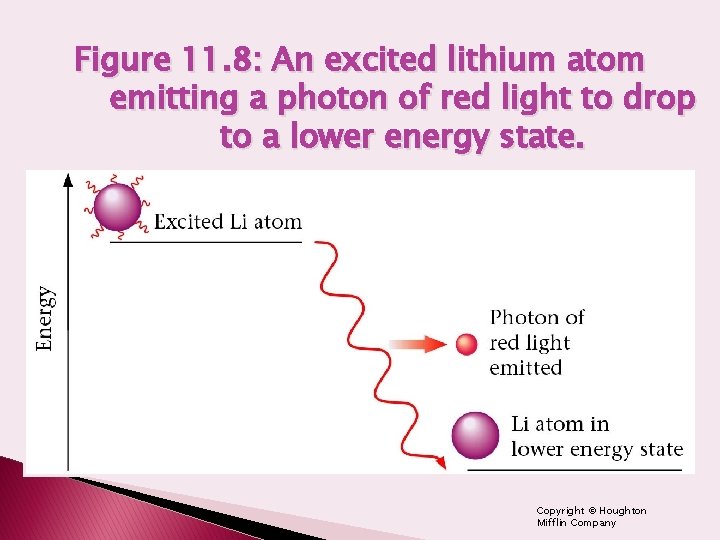
Emission of Energy by Atoms � When atoms receive energy from some source, they become excited, and they can release this energy by emitting light by releasing a photon. � The energy of the photon corresponds exactly to the energy change experienced by the emitting atom � Red=lower energy=longer wavelength � Violet=higher energy=shorter wavelength

Some Definitions � Ground State: Lowest possible energy state of an atom � Excited States: Possible Higher energy states. Only certain states are allowed for certain atoms.

Figure 11. 8: An excited lithium atom emitting a photon of red light to drop to a lower energy state. Copyright © Houghton Mifflin Company

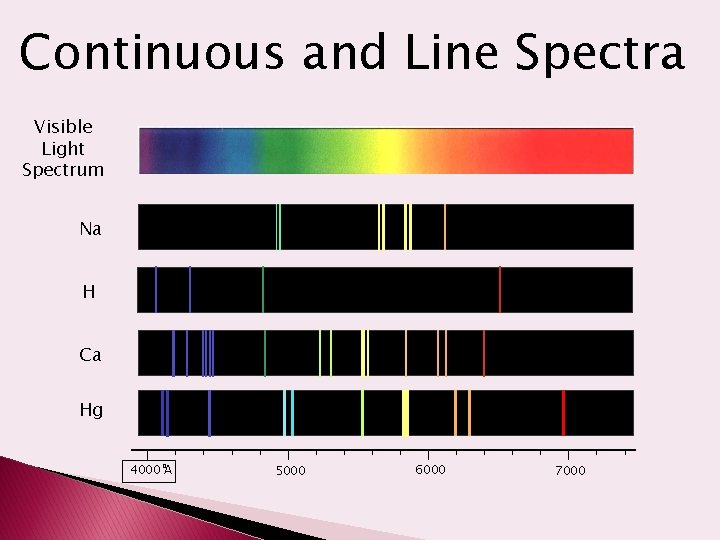
Flame Tests Many elements give off characteristic light which can be used to help identify them. strontium sodium lithium potassium copper


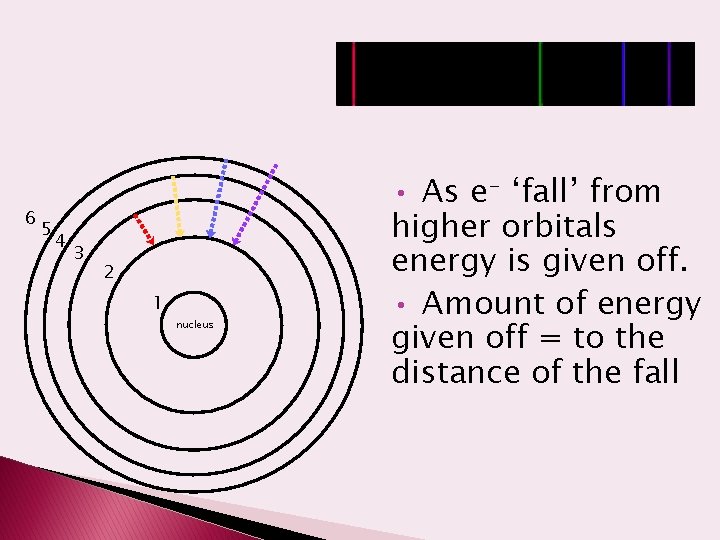
Emission Spectra � Atoms in the excited state are unstable. The electrons will fall back to the ground state. � When an electron moves from a higher energy level to a lower energy level, energy is released and light is emitted. � This energy is emitted in a form of light. The energy (color) of the light depends on how far the electron falls.

6 5 4 3 2 1 nucleus • As e- ‘fall’ from higher orbitals energy is given off. • Amount of energy given off = to the distance of the fall

‘Atomic Fingerprints’ • Each element has a unique atomic line emission spectrum. • Used to identify the element

Continuous and Line Spectra Visible Light Spectrum Na H Ca Hg o 4000 A 5000 6000 7000

Flame Emission Spectra Photographs of flame tests of burning wooden splints soaked in different salts. methane gas wooden splint sodium ion calcium ion copper ion strontium ion


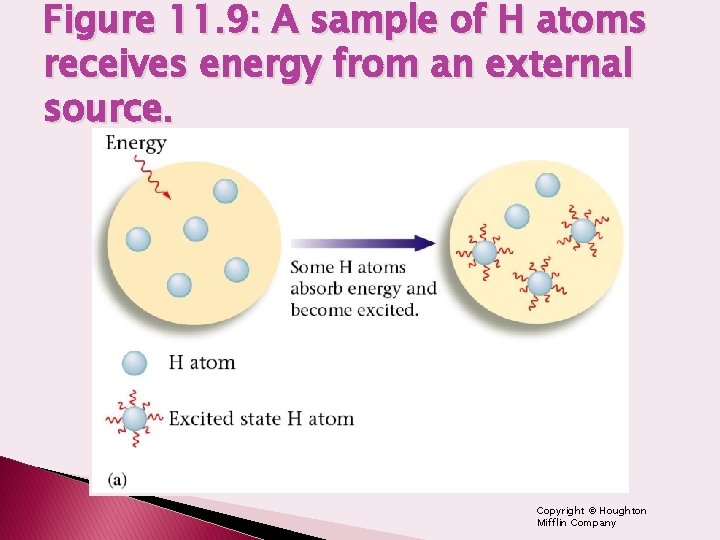
Let’s look more at hydrogen…

Figure 11. 9: A sample of H atoms receives energy from an external source. Copyright © Houghton Mifflin Company

Figure 11. 9: The excited atoms release energy by emitting photons. Excited atom can release some or all of its energy by emitting a photon (electromagnetic radiation “particle”) Copyright © Houghton Mifflin Company

Figure 11. 10: An excited H atom returns to a lower energy level. Energy contained in photon = change in energy of atom Copyright © Houghton Mifflin Company

Figure 11. 11: Colors and wavelengths of photons in the visible region. ØVisible light photons emitted by Hydrogen – always the same ØBecause only certain photons are emitted, only certain energy changes are occurring ØHydrogen atom has certain discrete energy levels ØEnergy levels of Hydrogen are quantized – only certain values allowed Copyright © Houghton Mifflin Company

Figure 11. 12: The color of the photon emitted depends on the energy change that produces it. Copyright © Houghton Mifflin Company

Figure 11. 13: Each photon emitted corresponds to a particular energy change. Copyright © Houghton Mifflin Company

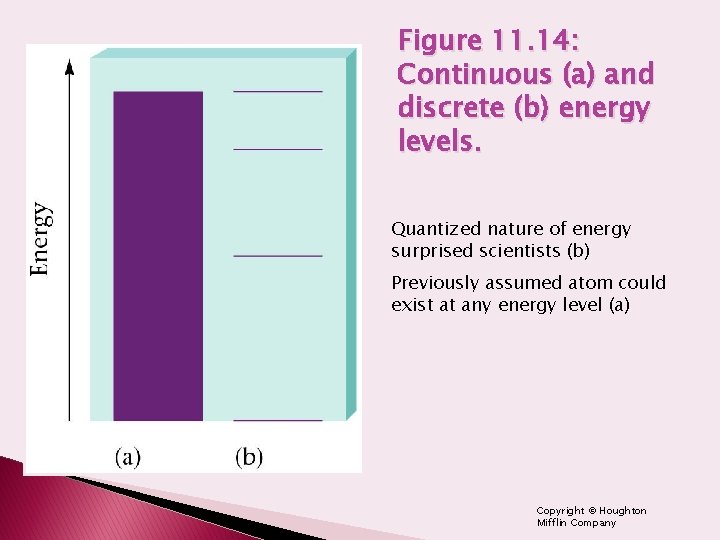
Figure 11. 14: Continuous (a) and discrete (b) energy levels. Quantized nature of energy surprised scientists (b) Previously assumed atom could exist at any energy level (a) Copyright © Houghton Mifflin Company

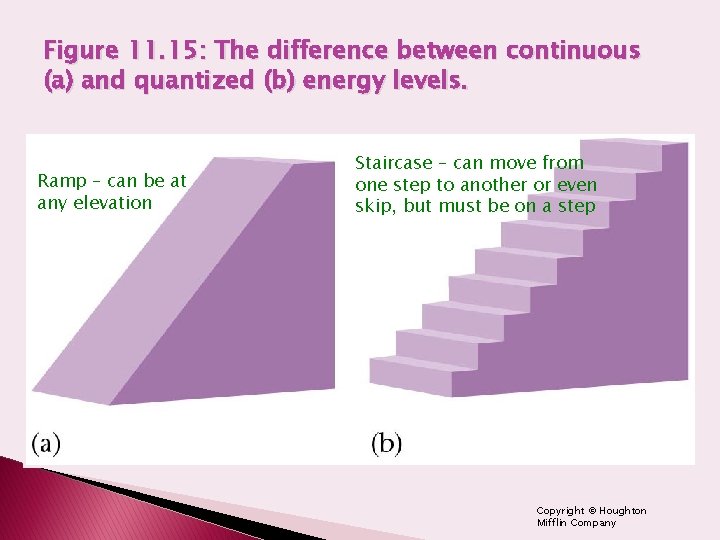
Figure 11. 15: The difference between continuous (a) and quantized (b) energy levels. Ramp – can be at any elevation Staircase – can move from one step to another or even skip, but must be on a step Copyright © Houghton Mifflin Company

Let’s look at Old Atom Models and Compare them to Newer Ones � OLD ◦ Rutherford ◦ Bohr � NEW ◦ de Broglie ◦ Schrodinger

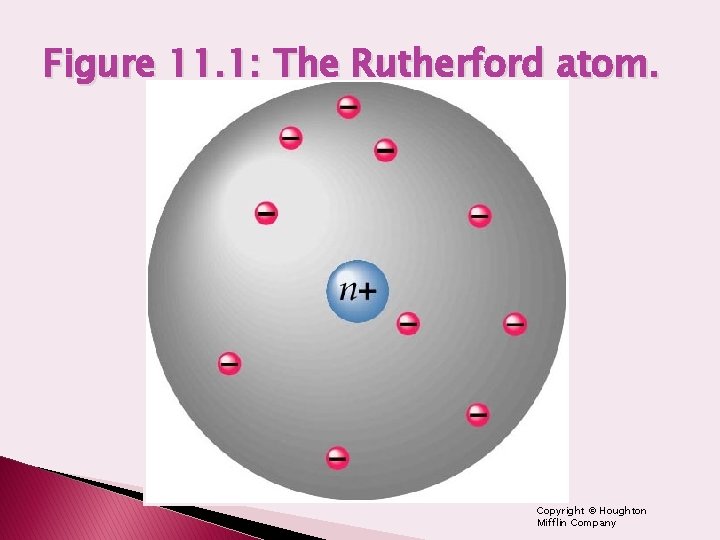
Figure 11. 1: The Rutherford atom. Copyright © Houghton Mifflin Company

Rutherford Atom Review � Alpha particle/Gold foil experiment � Nuclear Atom ◦ Nucleus composed of protons & neutrons ◦ Nucleus small compared to atomic size ◦ Electrons account for rest of atom � Unanswered questions: ◦ What are electrons doing? – How are they arranged & how do they move? ◦ Thought electrons revolved around nucleus like planets orbit the sun ◦ Couldn’t explain why electrons aren’t attracted to protons causing atom to collapse Copyright © Houghton Mifflin Company

Limitations of Rutherford’s Atomic Model • It explained only a few simple properties of atoms. • It could not explain the chemical properties of elements. For example, Rutherford’s model could not explain why an object such as the iron scroll shown here first glows dull red, then yellow, and then white when heated to higher and higher temperatures.

The Bohr Model � In 1913, Niels Bohr (1885– 1962), a young Danish physicist and a student of Rutherford, developed a new atomic model. • He changed Rutherford’s model to incorporate newer discoveries about how the energy of an atom changes when the atom absorbs or emits light.

The Bohr Model �According to Bohr’s atomic model, electrons move in definite orbits around the nucleus, much like planets circle the sun. �These orbits, or energy levels, are located at certain distances from the nucleus. � The energy levels are also called “electron shells”.

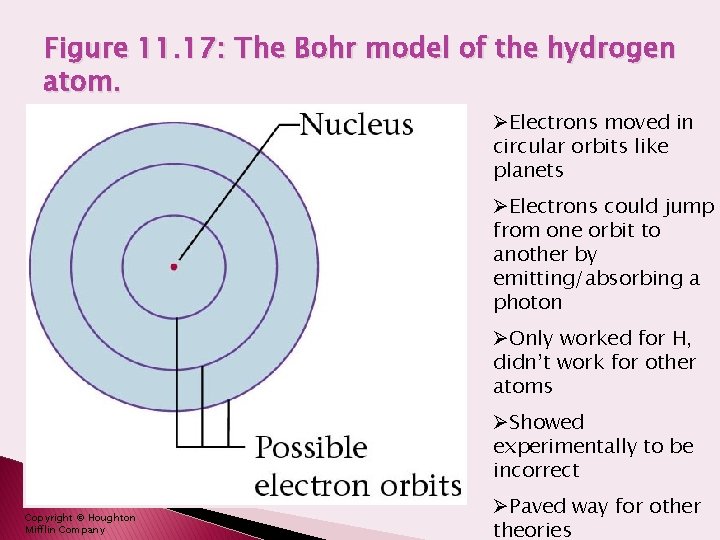
Figure 11. 17: The Bohr model of the hydrogen atom. ØElectrons moved in circular orbits like planets ØElectrons could jump from one orbit to another by emitting/absorbing a photon ØOnly worked for H, didn’t work for other atoms ØShowed experimentally to be incorrect Copyright © Houghton Mifflin Company ØPaved way for other theories

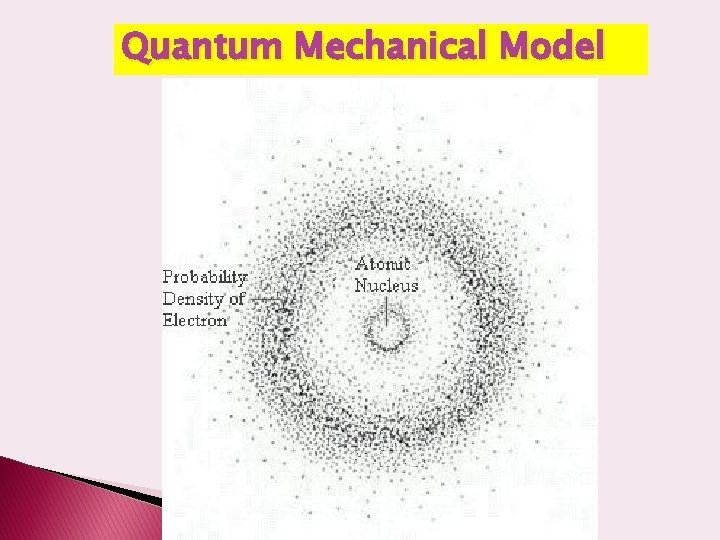
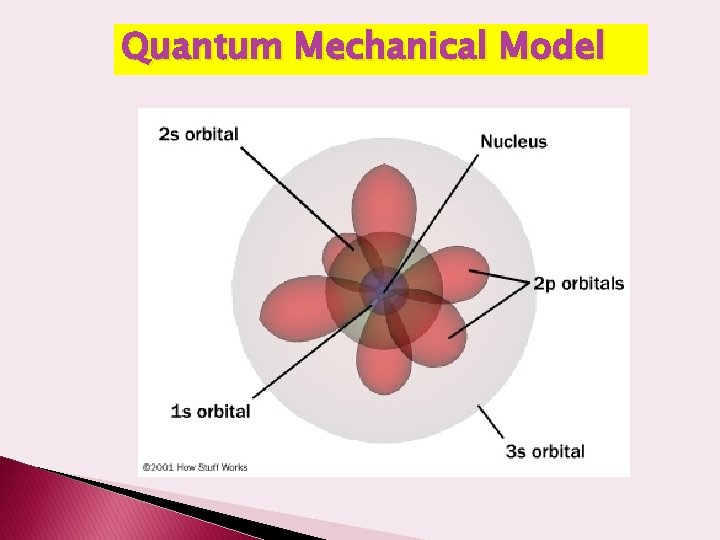
The Modern Model of the Atom � Accredited to Schrodinger, � According to the wave mechanic model, electrons do not move about an atom in a definite path, like the planets around the sun. � Electrons are NOT in fixed paths � Electrons are in orbitals which are nothing like Bohr’s orbits � The electrons move constantly throughout the energy levels forming an electron cloud

Electron Cloud • The space in which electrons are most likely to be found. • The cloud is more dense where the probability of finding the electron is high. • We can’t know exactly where an electron is (Heisenberg Uncertainty Principle) Electron cloud

Heisenberg Uncertainty Principle “One cannot simultaneously determine both the position and momentum of an electron. ” You can find out where the electron is, but not where it is going. OR… Werner Heisenberg You can find out where the electron is going, but not where it is!

Figure 11. 18: A representation of the photo of the firefly experiment (lightning bugs). Shows probability (or likelihood) of where firefly will be found Usually near the center, but can be found in any of the shaded areas at any time Copyright © Houghton Mifflin Company

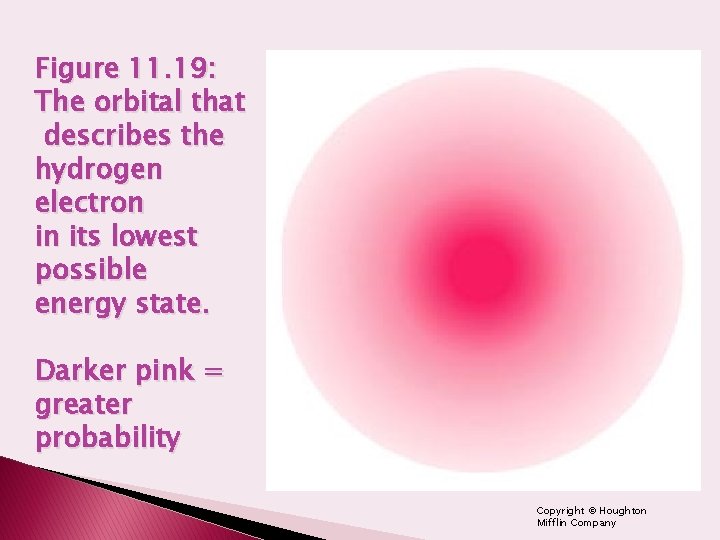
Figure 11. 19: The orbital that describes the hydrogen electron in its lowest possible energy state. Darker pink = greater probability Copyright © Houghton Mifflin Company

Drawbacks of wave mechanical model: � Gives no information about when the electron occupies a certain point in space or how it moves � We will probably never know the details of electron motion � Confident that Bohr model is incorrect 11 -50 Copyright © Houghton Mifflin Company

Quantum Mechanical Model

Quantum Mechanical Model

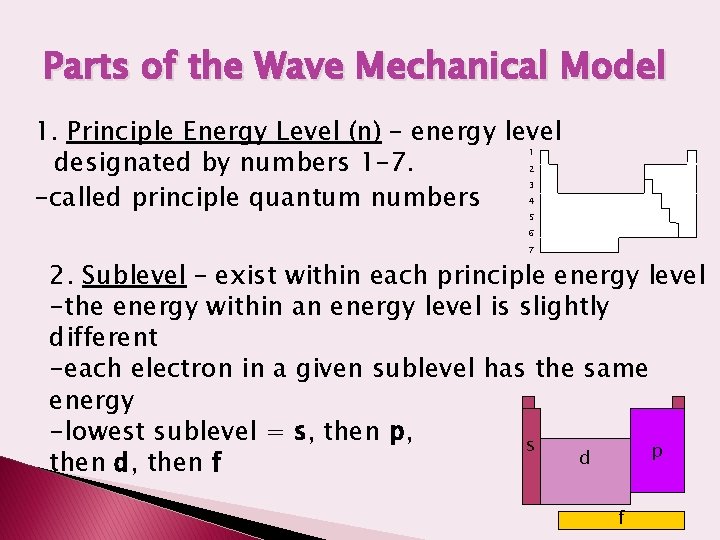
Parts of the Wave Mechanical Model 1. Principle Energy Level (n) – energy level designated by numbers 1 -7. -called principle quantum numbers 1 2 3 4 5 6 7 2. Sublevel – exist within each principle energy level -the energy within an energy level is slightly different -each electron in a given sublevel has the same energy -lowest sublevel = s, then p, s p d then d, then f f

Parts of the Wave Mechanical Model cont. 3. Orbital – region within a sublevel or energy level where electrons can be found s sublevel – 1 orbital p sublevel – 3 orbitals d sublevel – 5 orbitals f sublevel – 7 orbitals - ** No more than two electrons can occupy an orbital** -an orbital can be empty, half-filled, filled

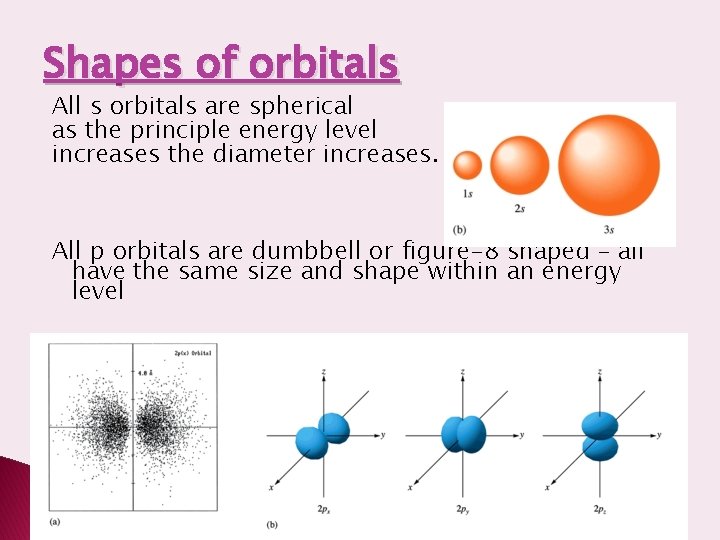
Shapes of orbitals All s orbitals are spherical as the principle energy level increases the diameter increases. All p orbitals are dumbbell or figure-8 shaped – all have the same size and shape within an energy level

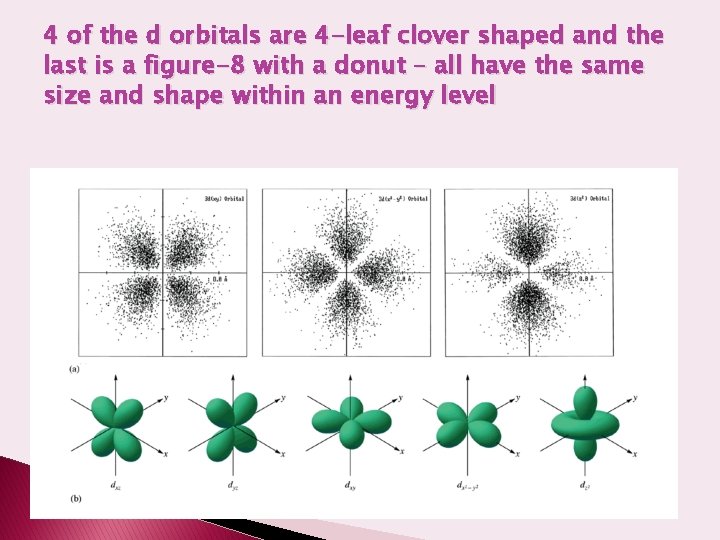
4 of the d orbitals are 4 -leaf clover shaped and the last is a figure-8 with a donut – all have the same size and shape within an energy level

f orbitals are complicated!!!!!

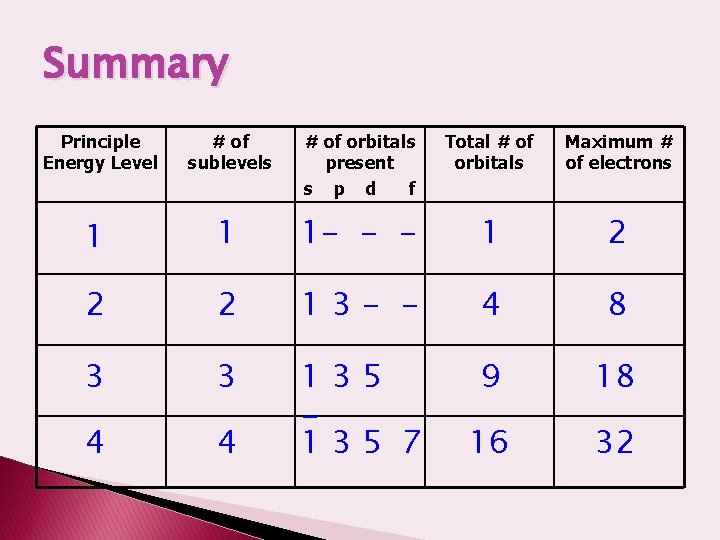
Summary Principle Energy Level # of sublevels # of orbitals present s p d f Total # of orbitals Maximum # of electrons 1 1 1 - - - 1 2 2 2 13 - - 4 8 3 3 9 18 4 4 135 7 16 32

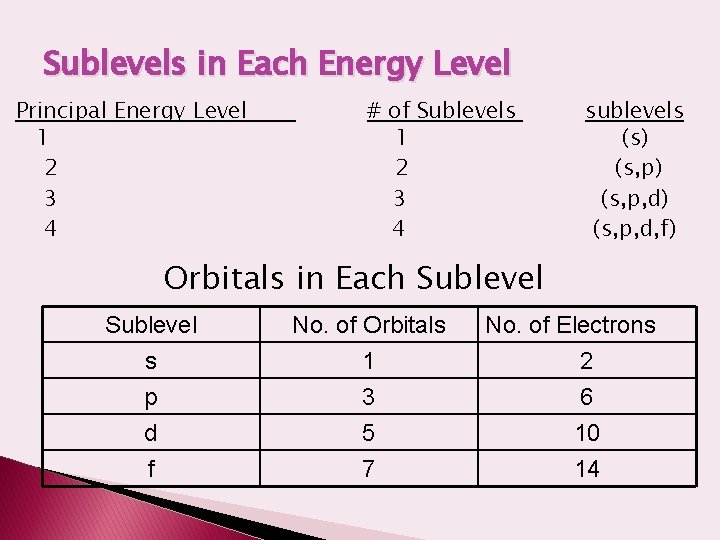
Sublevels in Each Energy Level Principal Energy Level 1 2 3 4 # of Sublevels 1 2 3 4 sublevels (s) (s, p, d) (s, p, d, f) Orbitals in Each Sublevel s p d No. of Orbitals 1 3 5 f 7 No. of Electrons 2 6 10 14

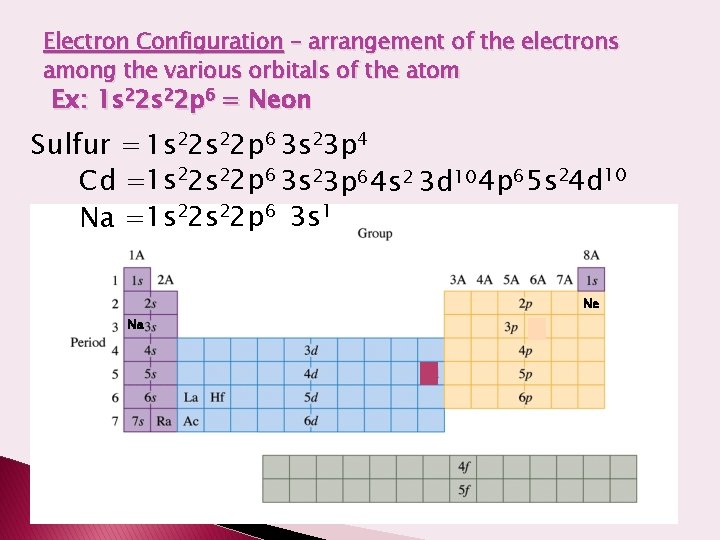
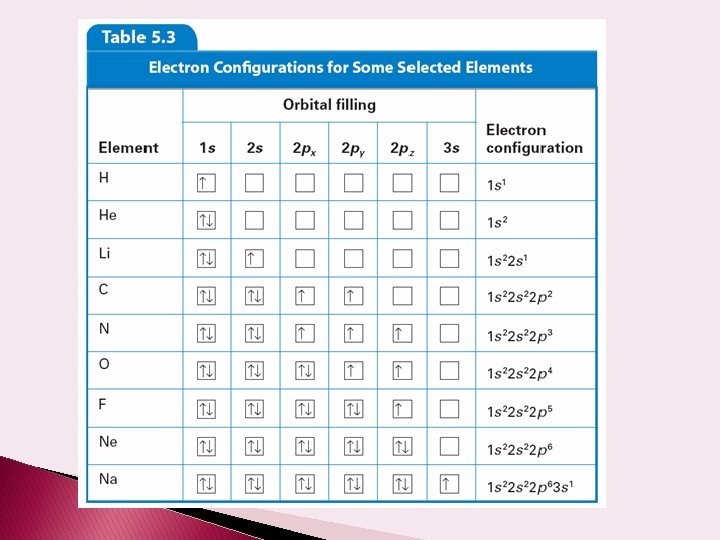
Electron Configuration – arrangement of the electrons among the various orbitals of the atom Ex: 1 s 22 p 6 = Neon Sulfur = 1 s 22 p 6 3 s 23 p 4 Cd =1 s 22 p 6 3 s 23 p 6 4 s 2 3 d 10 4 p 6 5 s 24 d 10 Na =1 s 22 p 6 3 s 1 Ne Na

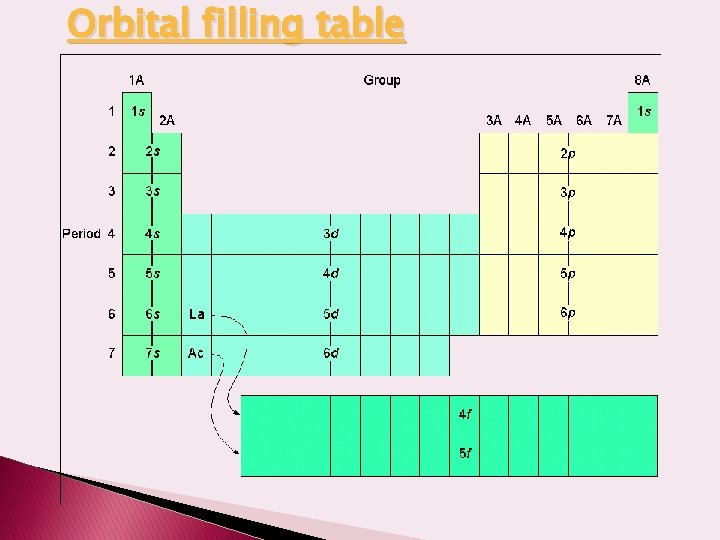
Orbital filling table

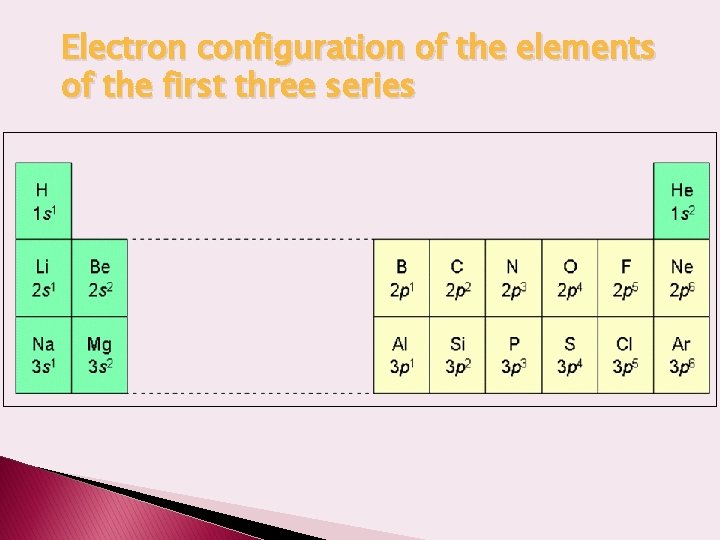
Electron configuration of the elements of the first three series

*Examples: Write the electron configuration using sublevels. 1. Hydrogen: ___________ 2. Carbon: _____________ 3. Phosphorous: ___________________ 4. Potassium: ____________________ 5. Calcium: ______________________ 6. Iodine: _____________________ **NOTE 1: Superscripts should ALWAYS add up to the # of electrons the atom has! (This is a good way of checking yourself!!!** **NOTE 2: When electrons are in an excited state, they will jump to a higher energy level before all the orbitals in the lower energy level are completely filled.

Sample Problems Which is the electron configuration of an atom in the excited state? a. 1 s 22 p 2 b. 1 s 22 p 1 c. 1 s 22 p 53 s 2 d. 1 s 22 p 63 s 1

An atom in the excited state can have an electron configuration of? a. 1 s 22 s 2 b. 1 s 22 p 1 c. 1 s 22 p 5 d. 1 s 22 p 6 Which electron configuration represents a potassium atom in the excited state? a. 1 s 22 p 63 s 23 p 3 b. 1 s 22 p 63 s 13 p 4 c. 1 s 22 p 63 s 23 p 64 s 1 d. 1 s 22 p 63 s 23 p 54 s 2

Electron Spin – motion that resembles earth rotating on its axis– clockwise or counterclockwise Pauli Exclusion Principle – two electrons in the same orbital must have opposite spins Hund’s Rule – All orbitals within a sublevel must contain at least one electron before any orbital can have two Orbital Diagram – describes the placement of electrons in orbitals � use arrows to represent electrons with spin � line represents orbital (s=1, p=3, d=5, f=7) ____ full ____ half-full ____ empty

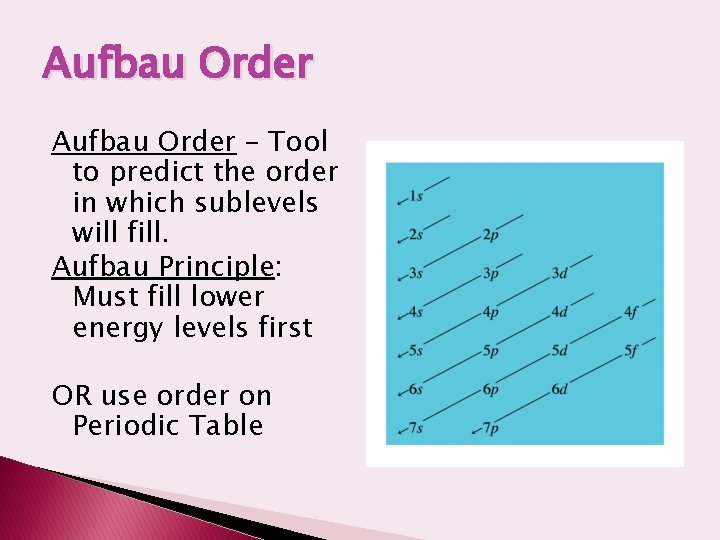
Aufbau Order – Tool to predict the order in which sublevels will fill. Aufbau Principle: Must fill lower energy levels first OR use order on Periodic Table


Orbital Diagrams Neon = 1 s__ 2 p__ __ __ Carbon = 1 s__ 2 p__ __ __ Zinc =1 s__ 2 p__ __ __ 3 s__ 3 p__ __ __ 4 s__ 3 d__ __ __ Gallium =1 s__ 2 p__ __ __ 3 s__ 3 p__ __ __ 4 s__ 3 d__ __ __ 4 p__ __ __

Atomic # (= # of electrons) Symbol Orbital Filling Diagram Electron Configuration 10 Ne

Atomic # (= # of electrons) Symbol Orbital Filling Diagram Electron Configuration 5 B

Atomic # (= # of electrons) Symbol Orbital Filling Diagram Electron Configuration 7 N

Atomic # (= # of electrons) Symbol Orbital Filling Diagram Electron Configuration 8 O

� Noble Gas Configuration – Shorthand configuration that substitutes a noble gas for electrons. Ex: Na = 1 s 22 p 63 s 1 or [Ne]3 s 1 Sn = 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 105 p 2 or [Kr]5 s 24 d 105 p 2 � Valence Electrons – Electrons in the outermost (highest) principle energy level in an atom, electrons use in bonding (outside of brackets) � Core Electrons – innermost electrons – not involved in bonding (inside brackets) � Valence Configuration – shows just the valence electrons 3 rd Shell/1 valence electron Ex: Na = 3 s 1 Sn = 5 s 25 p 2 5 th Shell/4 valence electrons
![Element Lithium Configuration notation Orbital notation 1 s 22 s 1 He2 s 1 Element Lithium Configuration notation Orbital notation 1 s 22 s 1 [He]2 s 1](https://slidetodoc.com/presentation_image_h2/f11bc924aff8e70e2af90cd609ca117f/image-75.jpg)
Element Lithium Configuration notation Orbital notation 1 s 22 s 1 [He]2 s 1 ____ 1 s Beryllium ____ 2 p ____ 2 s ____ 2 p ____ [He]2 s 2 p 2 ____ 2 s ____ 2 p ____ 1 s 22 p 3 [He]2 s 2 p 3 ____ 2 s ____ 2 p ____ 1 s 22 p 4 [He]2 s 2 p 4 ____ 2 s ____ 2 p ____ 1 s 22 p 5 [He]2 s 2 p 5 ____ 1 s Neon ____ 2 s 1 s 22 p 2 ____ 1 s Fluorine ____ [He]2 s 2 p 1 ____ 1 s Oxygen ____ 2 p 1 s 22 p 1 ____ 1 s Nitrogen ____ [He]2 s 2 ____ 1 s Carbon ____ 2 s 1 s 22 s 2 ____ 1 s Boron Noble gas notation ____ 2 s ____ 2 p ____ 1 s 22 p 6 [He]2 s 2 p 6 ____ 1 s ____ 2 s ____ 2 p ____

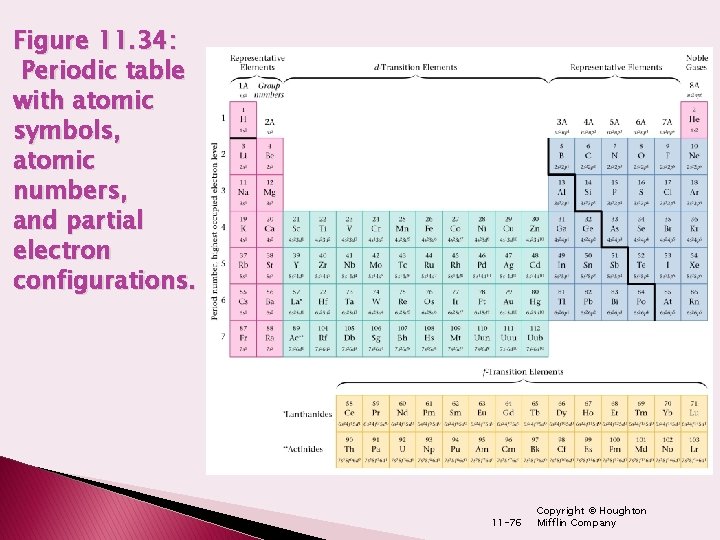
Figure 11. 34: Periodic table with atomic symbols, atomic numbers, and partial electron configurations. 11 -76 Copyright © Houghton Mifflin Company

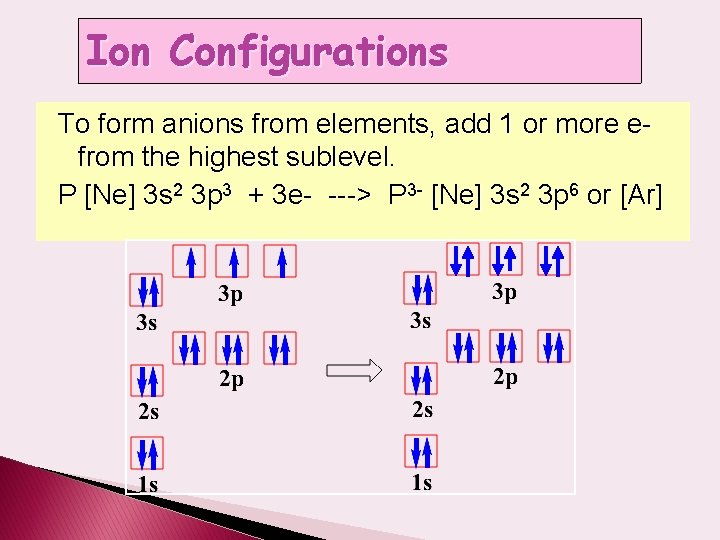
Ion Configurations To form anions from elements, add 1 or more efrom the highest sublevel. P [Ne] 3 s 2 3 p 3 + 3 e- ---> P 3 - [Ne] 3 s 2 3 p 6 or [Ar]

Ions � Cations: Take away that many electrons as the number on the charge (+1 means take away highest energy electron in configuration) � Anions: Add that many electrons as the number on the charge. Abide by Aufbau’s Principle