MODELS OF THE ATOM QUANTUM THEORY NIELS BOHR

MODELS OF THE ATOM & QUANTUM THEORY

NIELS BOHR (1913) • Previous research had concluded that light has a dual nature. • It could act as both particles and waves • Bohr proposed a model that explained why only certain frequencies of light were emitted from hydrogen.
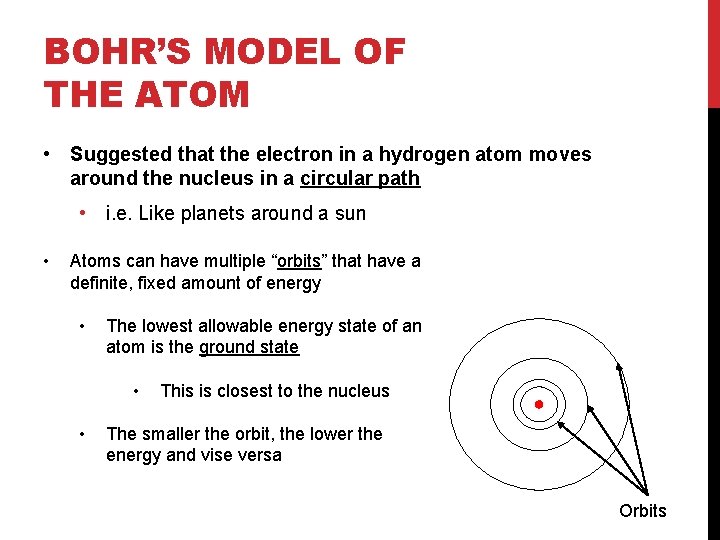
BOHR’S MODEL OF THE ATOM • Suggested that the electron in a hydrogen atom moves around the nucleus in a circular path • i. e. Like planets around a sun • Atoms can have multiple “orbits” that have a definite, fixed amount of energy • The lowest allowable energy state of an atom is the ground state • • This is closest to the nucleus The smaller the orbit, the lower the energy and vise versa Orbits
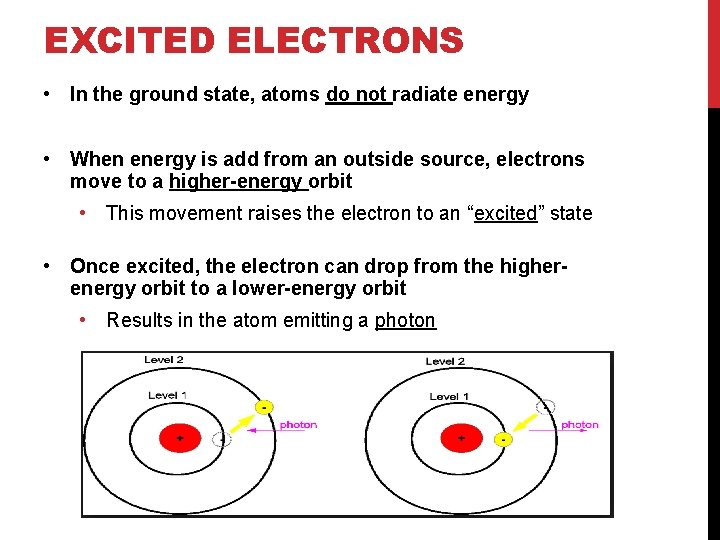
EXCITED ELECTRONS • In the ground state, atoms do not radiate energy • When energy is add from an outside source, electrons move to a higher-energy orbit • This movement raises the electron to an “excited” state • Once excited, the electron can drop from the higherenergy orbit to a lower-energy orbit • Results in the atom emitting a photon

LADDER ANALOGY • Like the rungs of the ladder on the right, the energy levels in an atom are not equally spaced • The higher the energy level occupied by an electron, the less energy it takes to move from the energy level to the next highest energy level

PROBLEM WITH BOHR’S MODEL • Bohr’s model had some limits • It could not explain the spectra of light emitted for any other element • It did not fully account for the chemical behavior of atoms

ELECTRONS AS WAVES • French scientist De Broglie pointed out the electron orbits in Bohr’s model were similar to the behavior of waves • Electrons, like waves, could be: • Diffracted (bent) • Bending a wave as it passes by the edge of an object • Interfere with each other • When waves overlap. Results in a reduction of energy in some areas and an increase of energy in others.

UNCERTAINTY PRINCIPLE • Werner Heisenberg – 1927 • Showed that it is impossible to take any measurement of an object without disturbing the object! • Detection of electrons • Detected by their interaction with photons • Have about the same mass as an electron • Found by “bumping” a photon into an electron • Doing so changes both wavelength of photon and the position and velocity of the electron • Heisenberg’s Uncertainty Principle: • It is fundamentally impossible to know precisely both the velocity and position of a particle at the same time.


SCHRÖDINGER'S WAVE EQUATIONS • Remember the problem with Bohr’s model? • Only relevant to hydrogen… • Erwin Schrodinger – 1926 • Developed an equation that treated electrons in atoms as waves • His new model not only could be applied to hydrogen, but all the other elements as well!!! YAY • This new model, in which electrons are treated as waves is called: • The Quantum Mechanical Model of the Atom • Other names: Wave Mechanical Model or Charge Cloud Model
SCHRÖDINGER SOUND FAMILIAR?

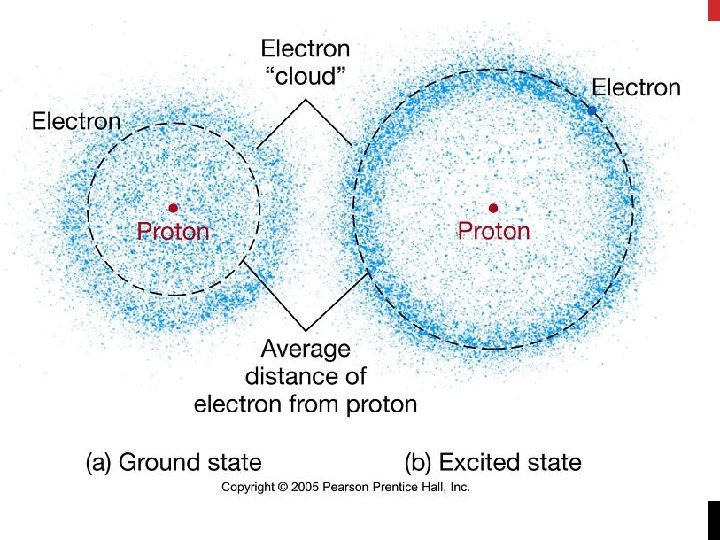
LOCATION OF ELECTRONS • Electrons described as waves only have a certain probability of being found within a particular volume of space around the nucleus • Schrödinger’s wave function predicts a 3 D region around the nucleus that described the electron’s probable location • Atomic Orbital: • Description of the 3 D region around the nucleus (looks like a fuzzy cloud) • Clouds show the region of probable electron locations • Size and shape of cloud depends on the energy of the electrons that occupy them

ACTIVITY Probability of finding an electron around the nucleus activity

ELECTRONS AROUND THE NUCLEUS • Orbitals can differ based on size and shape • There are four types of orbitals • S, P, D and F • Each orbital can hold two electrons 1 s • An S orbital is shaped like a sphere • A 1 s orbital is smaller than a 2 s orbital 2 s
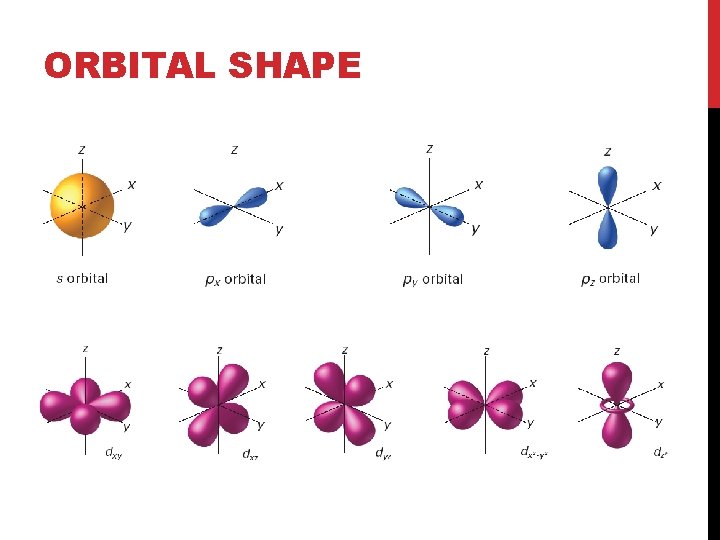
ORBITAL SHAPE
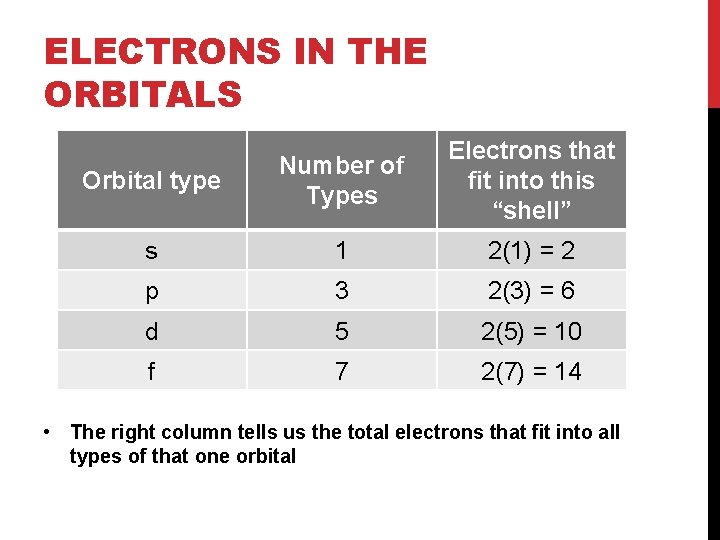
ELECTRONS IN THE ORBITALS Orbital type Number of Types Electrons that fit into this “shell” s 1 2(1) = 2 p 3 2(3) = 6 d 5 2(5) = 10 f 7 2(7) = 14 • The right column tells us the total electrons that fit into all types of that one orbital
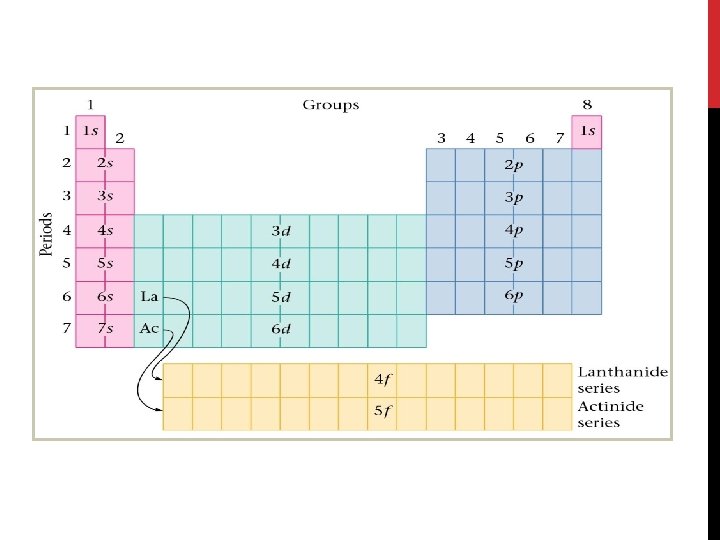
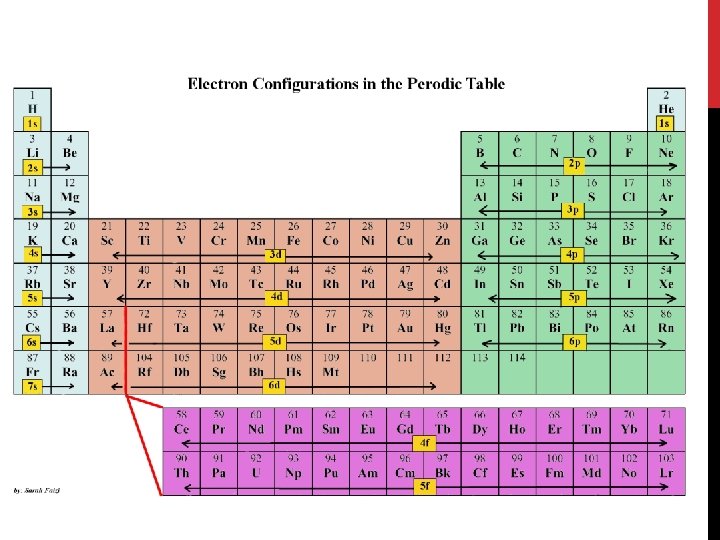

RULES OF ARRANGEMENT • Three rules, or principles define how electrons can be arranged in an atom’s orbitals 1. Aufbau Principle 2. Pauli Exclusion Principle 3. Hund’s Rule
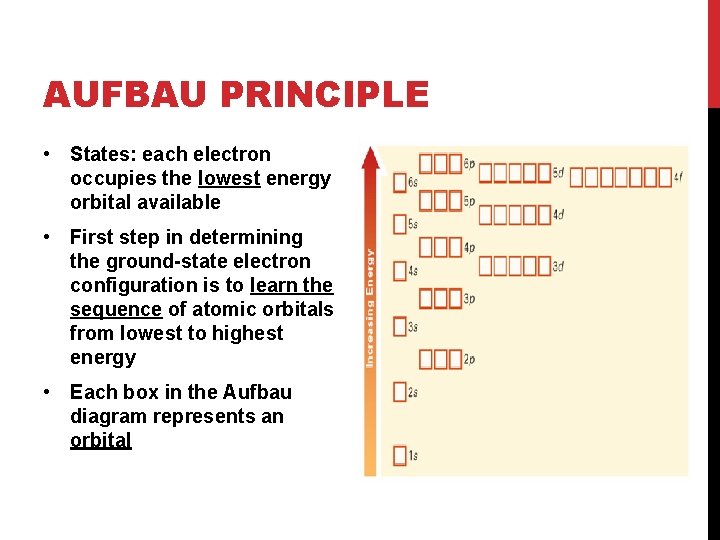
AUFBAU PRINCIPLE • States: each electron occupies the lowest energy orbital available • First step in determining the ground-state electron configuration is to learn the sequence of atomic orbitals from lowest to highest energy • Each box in the Aufbau diagram represents an orbital

ELECTRON CONFIGURATION Based on Aufbau’s Principle, the order of the orbitals should be: 1 s – 2 p – 3 s – 3 p – 4 s – 3 d – 4 p – 5 s – 4 d – 5 p – 6 s… All elements follow this rule with few exceptions

PAULI EXCLUSION PRINCIPLE • Electrons in orbitals can be represented by arrows in boxes • Each electron has an associated spin • • Arrow pointing up represents electron spinning in one direction • Arrow point down represents electron spinning in opposite direction • Empty box represents an unoccupied orbital Pauli Exclusion Principle: • A maximum of two electrons can occupy a single atomic orbital, but only if the electrons have opposite spins = 0 e- = 1 e- = 2 e-

HUND’S RULE • Remember that negatively charged electrons repel each other • Hund’s Rule: • Single electrons with the same spin must occupy each equal-energy orbital before additional electrons with opposite spins can occupy the same orbital

ELECTRON CONFIGURATION • The electron configuration on an atom is a shorthand method of writing the location of electrons by sublevel • • • The first number describes the principle energy level The letter represents the sublevel The sublevel is followed by a superscript with the number of electrons in the sublevel • If the 2 p sublevel contains 2 electrons, it is written 2 p 2

WRITING ELECTRON CONFIGURATIONS First, determine how many electrons are in the atom • Iron has 26 electrons Arrange the energy sublevels to increasing energy • 1 s 2 s 2 p 3 s 3 p 4 s 3 d… Fill each sublevel with electrons until you have used all the electrons in the atom • Fe: 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 6 The sum of the superscripts equals the atomic number of iron (26)

EXAMPLE Phosphorus, an element used in matches, has an atomic number of 15. Write the electron configuration of a phosphorus atom. Answer: • 1 s 2 2 p 6 3 s 2 3 p 3

PRACTICE ELECTRON CONFIGURATIONS Write the complete electron configurations for: • • Be F S Ca Things to double check: 1. All superscripts add up to total electrons 2. Orbital order follows the Aufbau Principle
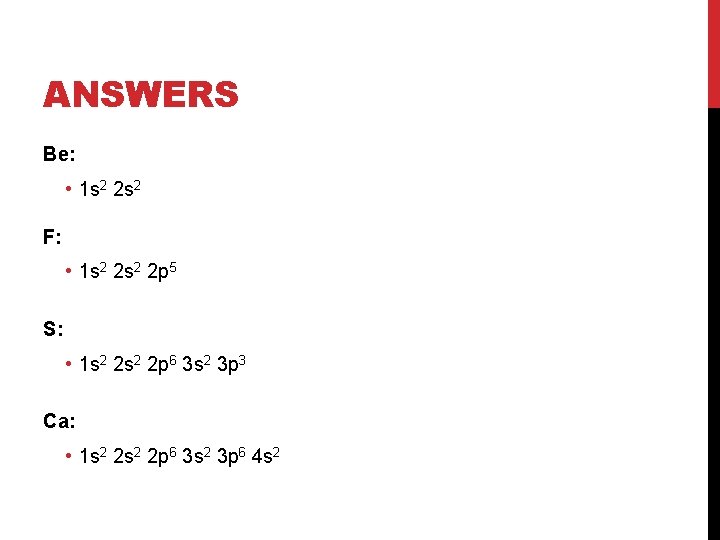
ANSWERS Be: • 1 s 2 2 s 2 F: • 1 s 2 2 p 5 S: • 1 s 2 2 p 6 3 s 2 3 p 3 Ca: • 1 s 2 2 p 6 3 s 2 3 p 6 4 s 2
- Slides: 29