MODELS OF THE ATOM Beginning with Rutherford Section

MODELS OF THE ATOM Beginning with Rutherford

Section 7. 5 PROBLEMS WITH RUTHERFORD’S The Quantum Mechanical Model of the Atom MODEL It didn’t explain WHY metals and metal compounds give off characteristic colors when they are flame tested It didn’t explain why metals glow when heated – first red, orange yellow and then white It didn’t explain the CHEMICAL properties of elements Return to TOC

Section 7. 5 The Quantum Mechanical Model of the Atom BOHR’S THEORY Electrons are located at specific energy levels surrounding the nucleus Each rung on the ladder represents an energy level The higher the energy level – the farther it is from the nucleus Bohr thought the electrons moved in fixed ORBITS around the nucleus – we know this is not true today Return to TOC

Section 7. 5 The Quantum Mechanical Model of the Atom BOHR MODEL First model of the electron structure Gives levels where an electron is most likely to be found Incorrect today, but a key in understanding the atom 4 Return to TOC
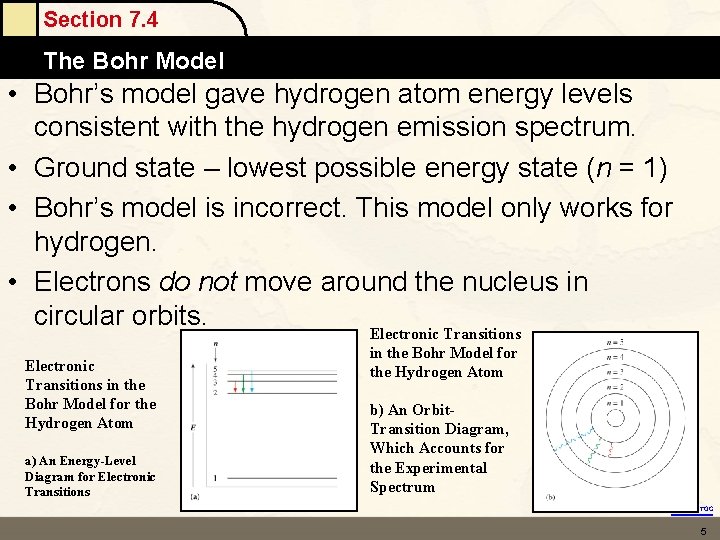
Section 7. 4 The Bohr Model • Bohr’s model gave hydrogen atom energy levels consistent with the hydrogen emission spectrum. • Ground state – lowest possible energy state (n = 1) • Bohr’s model is incorrect. This model only works for hydrogen. • Electrons do not move around the nucleus in circular orbits. Electronic Transitions in the Bohr Model for the Hydrogen Atom a) An Energy-Level Diagram for Electronic Transitions in the Bohr Model for the Hydrogen Atom b) An Orbit. Transition Diagram, Which Accounts for the Experimental Spectrum Return to TOC 5

Section 7. 5 The Quantum Mechanical Model of the Atom SCHRÖDINGER'S THEORY He agreed that electrons have a specific amount of energy He believed that the distance between rungs on the ladder were not consistent – they get closer together as you move higher up Quantum – the amount of energy needed to move from one energy level to another The electrons move in regions of probability around the nucleus called ORBITALS Return to TOC

Section 7. 5 The Quantum Mechanical Model of the Atom Quantum theory, also called wave mechanics, describes the arrangement and space occupied by electrons. Orbitals refers to the three-dimensional regions in space where there is a high probability of finding an electron around an atom. 7 Return to TOC

Section 7. 5 The Quantum Mechanical Model of the Atom CHARACTERISTICS OF ELECTRONS l Extremely small mass l Located outside the nucleus l Moving at extremely high speeds in a sphere l Have specific energy levels 8 Return to TOC

Section 7. 5 The Quantum Mechanical Model of the Atom ENERGY OF ELECTRONS l When atoms are heated, bright lines appear called line spectra l Electrons in atoms arranged in discrete levels. l An electron absorbs energy to “jump” to a higher energy level. l When an electron falls to a lower energy level, energy is emitted. 9 Return to TOC
Section 7. 5 The Quantum Mechanical Model of the Atom LOSS AND GAIN OF ENERGY G a I n L o s s 10 Return to TOC

Section 7. 5 The Quantum Mechanical Model of the Atom LEARNING CHECK Answer with 1) Energy absorbed 2) Energy emitted 3) No change in energy A. What energy change takes place when an electron in a hydrogen atom moves from the first (n=1) to the second shell (n=2)? B. What energy change takes place when the electron moves from the third shell to the second shell? 11 Return to TOC

Section 7. 5 The Quantum Mechanical Model of the Atom SOLUTION A. 1) Energy absorbed B. 2) Energy emitted 12 Return to TOC

Section 7. 5 The Quantum Mechanical Model of the Atom • We do not know the detailed pathway of an electron. • The electrons move in regions of probability around the nucleus called ORBITALS RELATIVE ORBITAL SIZE Difficult to define precisely. Orbital is a wave function. Picture an orbital as a three-dimensional electron density map. Hydrogen 1 s orbital: § Radius of the sphere that encloses 90% of the total electron probability. 13 Return to TOC

Section 7. 5 The Mechanical Model of the Atom THEQuantum ELECTRONS MOVE IN REGIONS OF PROBABILITY AROUND THE NUCLEUS CALLED ORBITALS DEFINING THESE ORBITALS: Quantum Numbers are used to define: The energy of the electron The electron’s relative distance from the nucleus The size and shape of the ORBITAL The pairings of the electrons Return to TOC

Section 7. 5 The Quantum Mechanical Model of the Atom QUANTUM NUMBERS Principle Quantum Number (n) – define the energy of the electron n=1 is closest to the nucleus – low energy n=2 is farther than n=1, slightly more energy n=3 is farther than n=1 and n=2, still increasing in energy n=4 …. . Remember – The difference in energy between energy levels decreases as “n” increases Return to TOC

Section 7. 5 The Quantum Mechanical Model of the Atom SUBLEVELS Within each principle energy level (n) – there are sublevel(s). The larger the value of ‘n’, the more sublevels you can have. Sublevels – named by their shape s – sphere p – pear d- dumbbell f - fundamental Return to TOC

Section 7. 5 The Quantum Mechanical Model of the Atom TWO REPRESENTATIONS FOR AN S ORBITAL Figure 3. 16, pg. 77 Investigating Chemistry, 2 nd Edition © 2009 W. H. Freeman & Company Return to TOC
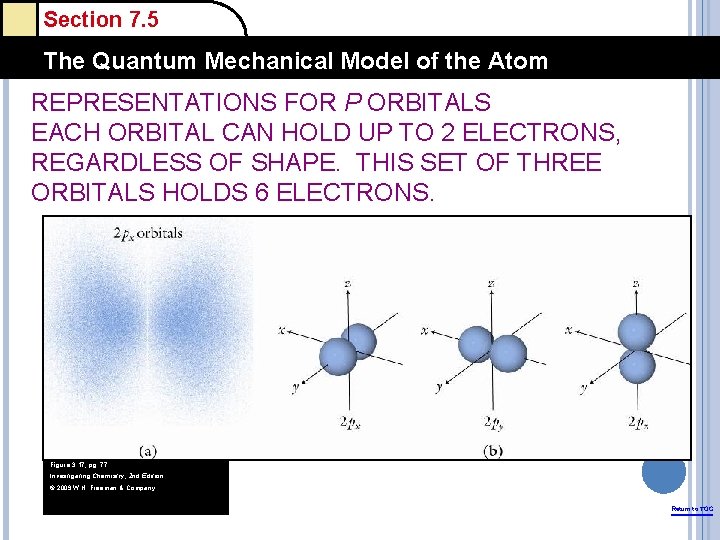
Section 7. 5 The Quantum Mechanical Model of the Atom REPRESENTATIONS FOR P ORBITALS EACH ORBITAL CAN HOLD UP TO 2 ELECTRONS, REGARDLESS OF SHAPE. THIS SET OF THREE ORBITALS HOLDS 6 ELECTRONS. Figure 3. 17, pg. 77 Investigating Chemistry, 2 nd Edition © 2009 W. H. Freeman & Company Return to TOC
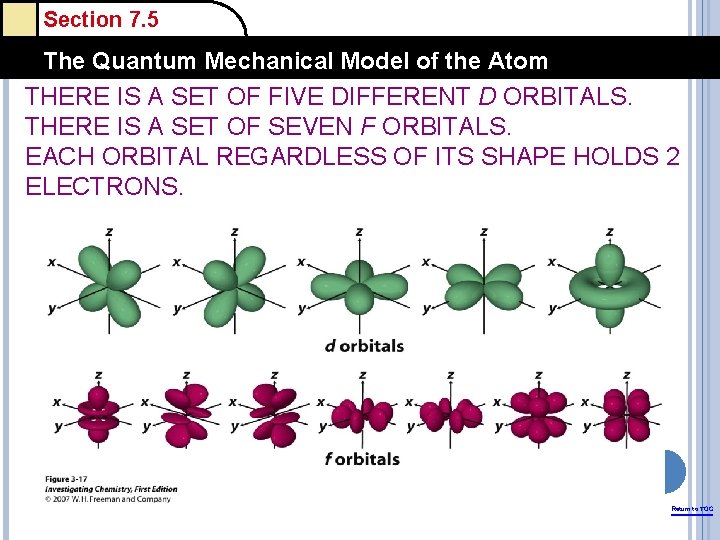
Section 7. 5 The Quantum Mechanical Model of the Atom THERE IS A SET OF FIVE DIFFERENT D ORBITALS. THERE IS A SET OF SEVEN F ORBITALS. EACH ORBITAL REGARDLESS OF ITS SHAPE HOLDS 2 ELECTRONS. Return to TOC

Section 7. 7 Orbital Shapes and Energies 1 s Orbital Return to TOC 20
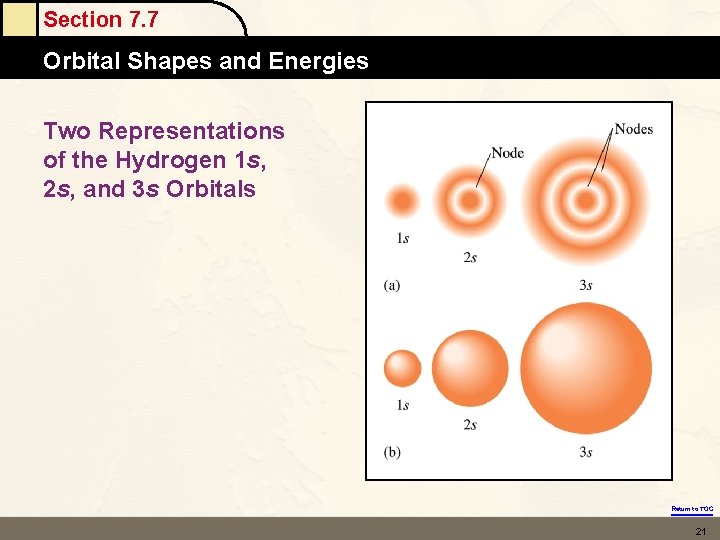
Section 7. 7 Orbital Shapes and Energies Two Representations of the Hydrogen 1 s, 2 s, and 3 s Orbitals Return to TOC 21
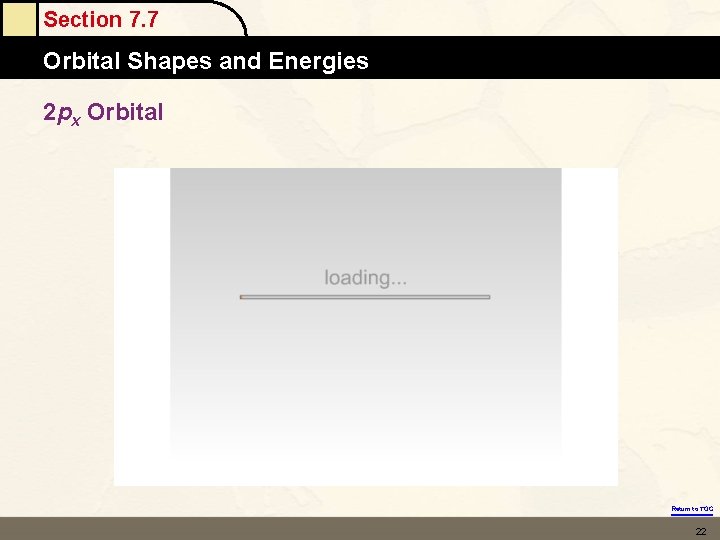
Section 7. 7 Orbital Shapes and Energies 2 px Orbital Return to TOC 22
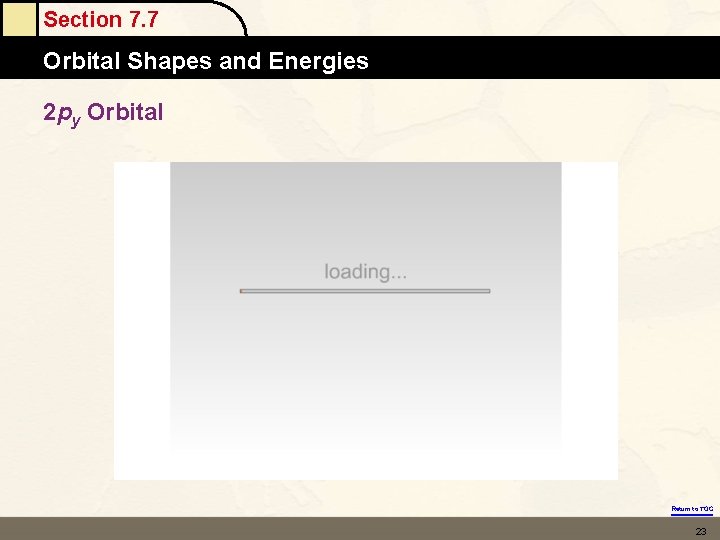
Section 7. 7 Orbital Shapes and Energies 2 py Orbital Return to TOC 23
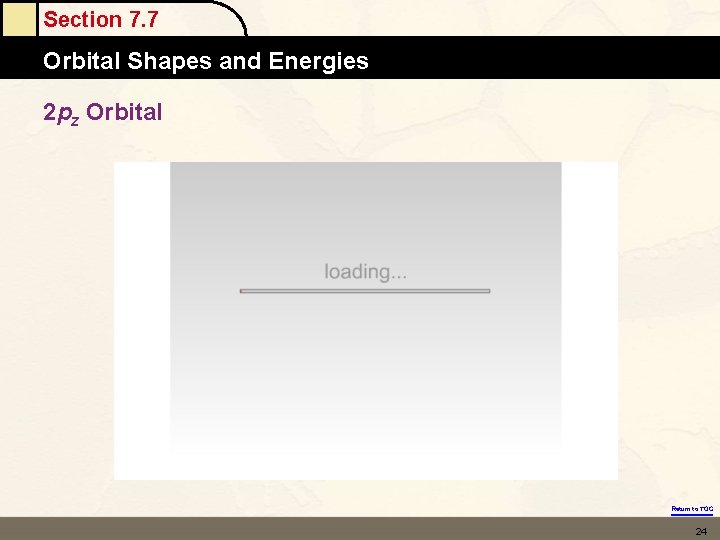
Section 7. 7 Orbital Shapes and Energies 2 pz Orbital Return to TOC 24
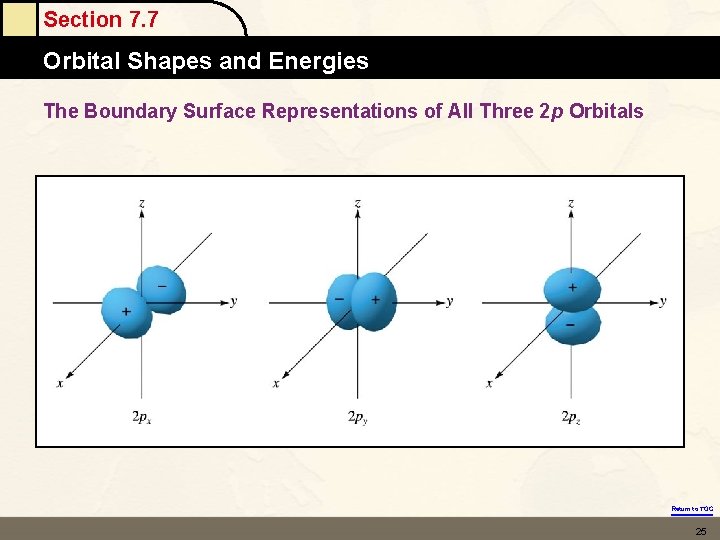
Section 7. 7 Orbital Shapes and Energies The Boundary Surface Representations of All Three 2 p Orbitals Return to TOC 25

Section 7. 7 Orbital Shapes and Energies 3 dx -y Orbital 2 2 Return to TOC 26
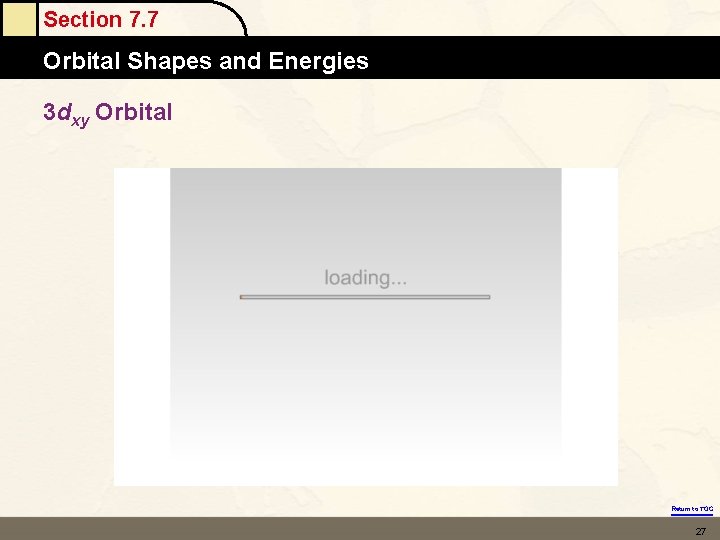
Section 7. 7 Orbital Shapes and Energies 3 dxy Orbital Return to TOC 27
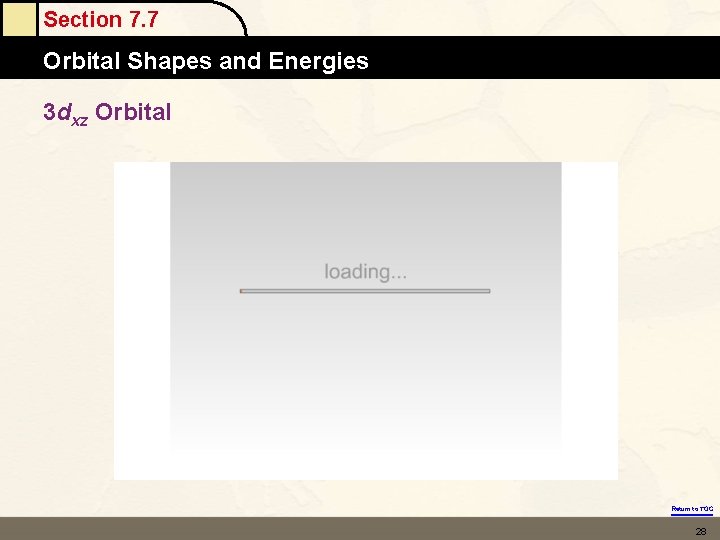
Section 7. 7 Orbital Shapes and Energies 3 dxz Orbital Return to TOC 28
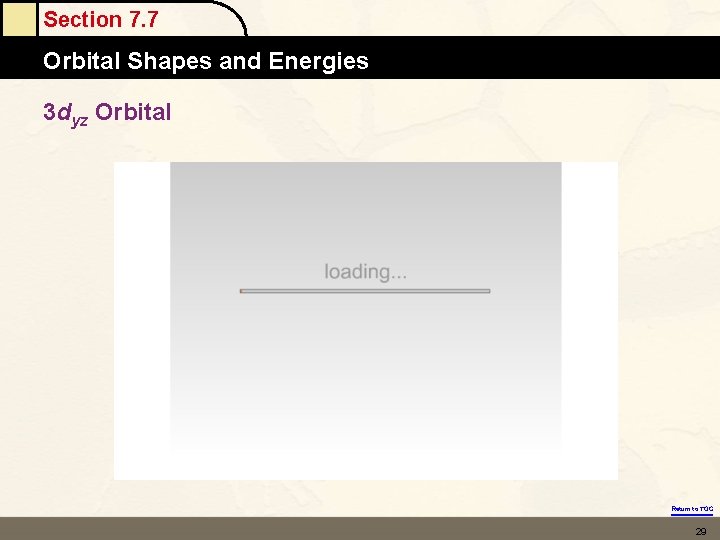
Section 7. 7 Orbital Shapes and Energies 3 dyz Orbital Return to TOC 29
Section 7. 7 Orbital Shapes and Energies Orbital Return to TOC 30
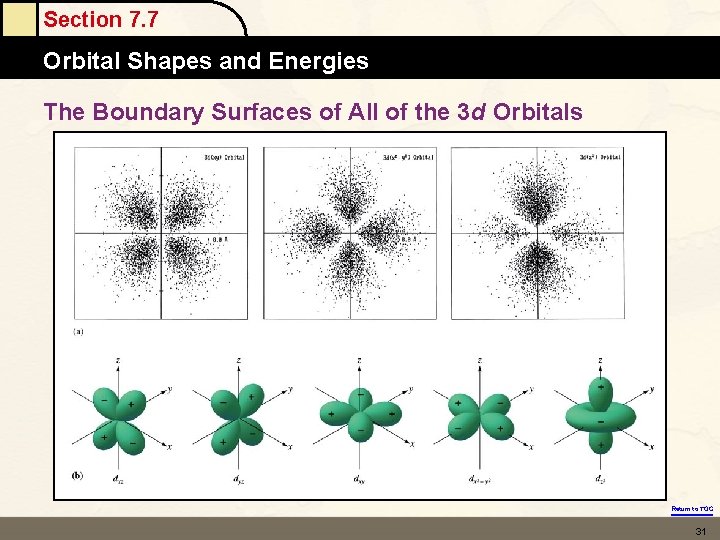
Section 7. 7 Orbital Shapes and Energies The Boundary Surfaces of All of the 3 d Orbitals Return to TOC 31
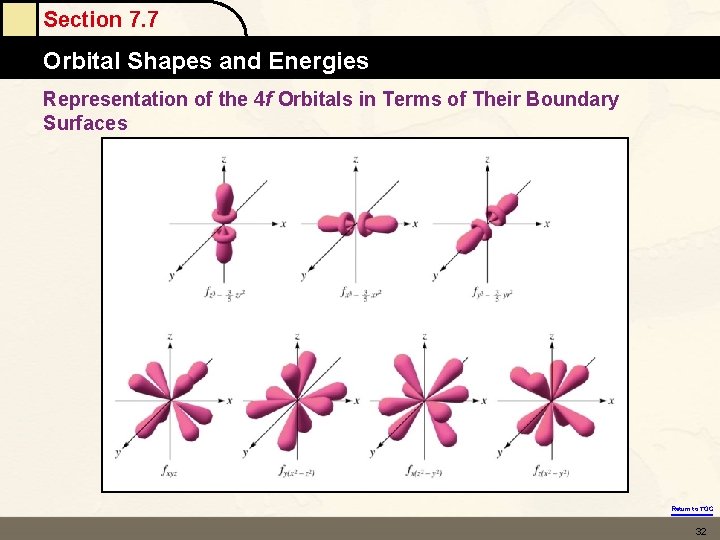
Section 7. 7 Orbital Shapes and Energies Representation of the 4 f Orbitals in Terms of Their Boundary Surfaces Return to TOC 32

Section 7. 5 The Quantum Mechanical Model of the Atom SUBLEVELS Principle Energy Level n= 1 n=2 n=3 Sublevel s s and p and d n=4 s, p, d, and f NOTICE: The value of ‘n’ tells you how many sublevels are present in that energy level Return to TOC

Section 7. 6 Quantum Numbers • Principal quantum number (n) – size and energy of the orbital. • Angular momentum quantum number (l) – shape of atomic orbitals (sometimes called a subshell). • Magnetic quantum number (ml) – orientation of the orbital in space relative to the other orbitals in the atom. Return to TOC 34

QUANTUM NUMBERS FOR THE FIRST FOUR LEVELS OF ORBITALS IN THE HYDROGEN ATOM 35

Section 7. 6 Quantum Numbers Exercise For principal quantum level n = 3, determine the number of allowed subshells (different values of l), and give the designation of each. (hint refer to previous chart) # of allowed subshells = 3 l = 0, 3 s l = 1, 3 p l = 2, 3 d Return to TOC 36

Section 7. 6 Quantum Numbers Exercise For l = 2, determine the magnetic quantum numbers (ml) and the number of orbitals. (note refer to previous chart) magnetic quantum numbers = – 2, – 1, 0, 1, 2 number of orbitals = 5 Return to TOC 37

Section 7. 7 Orbital Shapes and Energies Locating these on the Periodic Table Principle Energy Level (n) – is the period in the periodic table The Sublevels are located in specific regions – Color these together Return to TOC
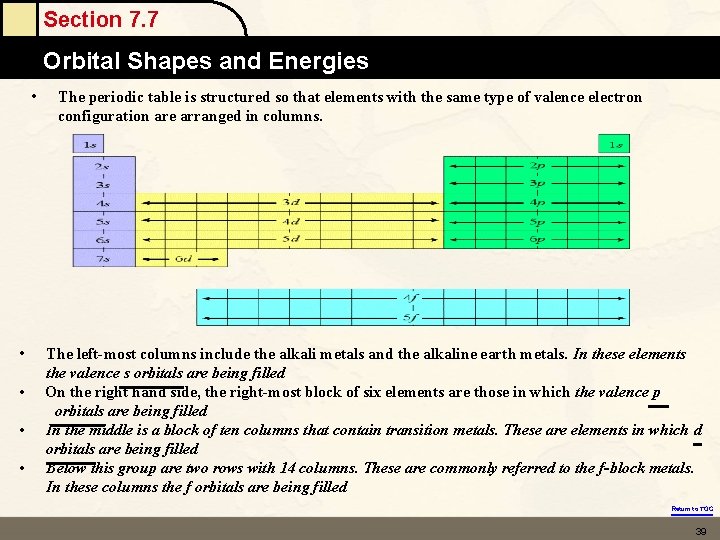
Section 7. 7 Orbital Shapes and Energies • • • The periodic table is structured so that elements with the same type of valence electron configuration are arranged in columns. The left-most columns include the alkali metals and the alkaline earth metals. In these elements the valence s orbitals are being filled On the right hand side, the right-most block of six elements are those in which the valence p orbitals are being filled In the middle is a block of ten columns that contain transition metals. These are elements in which d orbitals are being filled Below this group are two rows with 14 columns. These are commonly referred to the f-block metals. In these columns the f orbitals are being filled Return to TOC 39
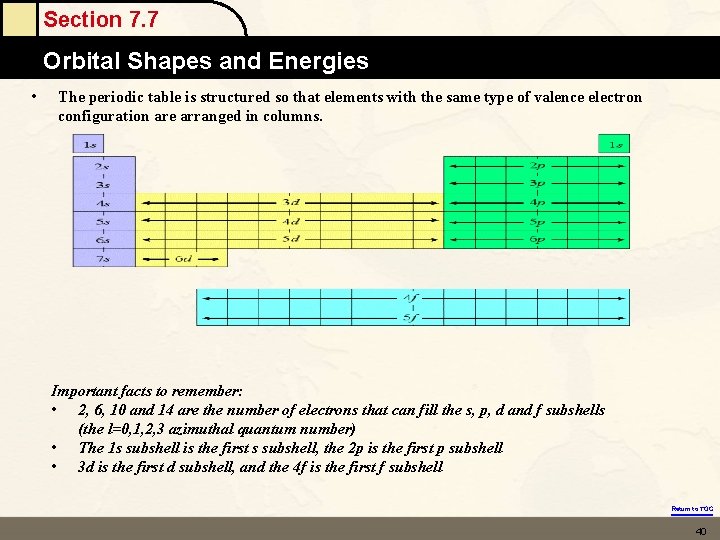
Section 7. 7 Orbital Shapes and Energies • The periodic table is structured so that elements with the same type of valence electron configuration are arranged in columns. Important facts to remember: • 2, 6, 10 and 14 are the number of electrons that can fill the s, p, d and f subshells (the l=0, 1, 2, 3 azimuthal quantum number) • The 1 s subshell is the first s subshell, the 2 p is the first p subshell • 3 d is the first d subshell, and the 4 f is the first f subshell Return to TOC 40

Section 7. 7 Orbital Shapes and Energies Naming the sublevels 1 s 2 s 3 s 4 s 2 p 3 p 4 p 3 d 4 d 4 f Return to TOC

Section 7. 7 Orbital Shapes and Energies Orbitals are regions of probability – each orbital can hold a maximum of 2 e. The ‘s’ sublevel has 1 orbital The ‘p’ sublevel has 3 orbitals The ‘d’ sublevel has 5 orbitals The ‘f’ sublevel has 7 orbitals Return to TOC
Section 7. 7 Orbital Shapes and Energies Orbitals Do you have to memorize this? NO Look at the sublevel regions that you colored in on your periodic table. Return to TOC
Section 7. 7 Orbital Shapes and Energies Orbitals Count how many electrons are in the ‘s’ sublevel 2 This means that since there are two electrons, and each orbital can hold two electrons, that there is only ONE orbital. Return to TOC
Section 7. 7 Orbital Shapes and Energies Orbitals Count how many electrons are in the ‘p’ sublevel 6 This means that since there are six electrons, and each orbital can hold two electrons, that there are THREE orbitals. Return to TOC
Section 7. 7 Orbital Shapes and Energies Orbitals Count how many electrons are in the ‘d’ sublevel 10 This means that since there are ten electrons, and each orbital can hold two electrons, that there are FIVE orbitals. Return to TOC

ORBITALS Count how many electrons are in the ‘f’ sublevel 14 This means that since there are fourteen electrons, and each orbital can hold two electrons, that there are SEVEN orbitals.
- Slides: 47